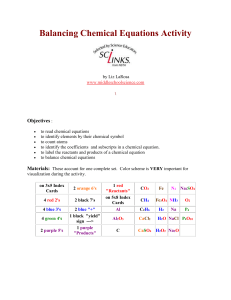
Balancing Chemical Equations Activity by Liz LaRosa www
... few days. Once done, you can laminate them and have them forever! The materials account for one complete set which is good for 2-3 students to use. Print activity cards on card stock instead of making index cards for quicker set up. The color coding is very important for visualization. It is easier ...
... few days. Once done, you can laminate them and have them forever! The materials account for one complete set which is good for 2-3 students to use. Print activity cards on card stock instead of making index cards for quicker set up. The color coding is very important for visualization. It is easier ...
oxidation, reduction, redox potential, citric acid cycle, respiratory
... phosphorylation) and electrons reduce oxygen atoms to O2-. Oxygen anoints react with protons in the mitochondrial matrix and form H2O (metabolic water). Oxidative phosphorylation rate depends on need of ATP and sufficiency of reduced coenzyme, O2, ADP and Pi are essential for this reaction. The resp ...
... phosphorylation) and electrons reduce oxygen atoms to O2-. Oxygen anoints react with protons in the mitochondrial matrix and form H2O (metabolic water). Oxidative phosphorylation rate depends on need of ATP and sufficiency of reduced coenzyme, O2, ADP and Pi are essential for this reaction. The resp ...
What is hydrogen peroxide?
... substance, said to be capable of turning base metals , especially lead , into gold; it was also sometimes believed to be an elixir of life , useful for rejuvenation and possibly ...
... substance, said to be capable of turning base metals , especially lead , into gold; it was also sometimes believed to be an elixir of life , useful for rejuvenation and possibly ...
Energetics of alpha-helix formation and packing
... In the context of an alpha helix. Use your model of an alpha helix to see which pf the three staggered conformations around the C-alpha-to-C-beta bond a side chain can adopt without causing bad steric clashes. There are only two conformations. If the amino acid is not beta branched, this means the m ...
... In the context of an alpha helix. Use your model of an alpha helix to see which pf the three staggered conformations around the C-alpha-to-C-beta bond a side chain can adopt without causing bad steric clashes. There are only two conformations. If the amino acid is not beta branched, this means the m ...
Welcome to AP Chemistry! I am glad that you decided to take on the
... This assignment will be collected at the second week of school in September. There will be a test on this material during the second week. Refer to related material in your notes from last ...
... This assignment will be collected at the second week of school in September. There will be a test on this material during the second week. Refer to related material in your notes from last ...
Preparation and Properties of Hydrogen
... Hydrogen (H2) is a diatomic gas (two atoms) that is tasteless, colourless, and odourless. The element hydrogen (H) has the lowest atomic weight (1.008 amu), and is the least dense of any known substance. Because of hydrogen's low density (1/14 th the density of air), balloons filled with hydrogen wi ...
... Hydrogen (H2) is a diatomic gas (two atoms) that is tasteless, colourless, and odourless. The element hydrogen (H) has the lowest atomic weight (1.008 amu), and is the least dense of any known substance. Because of hydrogen's low density (1/14 th the density of air), balloons filled with hydrogen wi ...
(ΔG) Hydrogen Bonds
... Entropy is a measure of disorder in a system. It decreases with increasing order. If -T ΔS is negative as in the above table then ΔS must be positive. Rationalization: Because the non-polar group can not hydrogen bond to water, the water molecules at the surface of the non-polar molecule have fewer ...
... Entropy is a measure of disorder in a system. It decreases with increasing order. If -T ΔS is negative as in the above table then ΔS must be positive. Rationalization: Because the non-polar group can not hydrogen bond to water, the water molecules at the surface of the non-polar molecule have fewer ...
Bohr Atom, Atomic spectra
... and we are continually bombarded by them They rain down continuously (will cover later), and some of them lose energy and slow down near an atom. Will its orbit? A. Further away B. Nearer to nucleus C. Accelerate inward, destroy the nucleus and all life as we know it ! ...
... and we are continually bombarded by them They rain down continuously (will cover later), and some of them lose energy and slow down near an atom. Will its orbit? A. Further away B. Nearer to nucleus C. Accelerate inward, destroy the nucleus and all life as we know it ! ...
chemical*equations
... “Success'is'not',inal,'failure' is'not'fatal:'it'is'the'courage' to'continue'that'counts.” ''7Winston'Churchill ...
... “Success'is'not',inal,'failure' is'not'fatal:'it'is'the'courage' to'continue'that'counts.” ''7Winston'Churchill ...
Problem Set 1 - Andrew.cmu.edu
... in the ammonium ion, NH4+. Nitrogen has 7 electrons. Two of these are in the 1st shell and the remaining five are in the second shell. The second shell contains four orbitals, three of these will have a single electron (that’s why nitrogen forms three bonds). The fourth is full, with two electrons. ...
... in the ammonium ion, NH4+. Nitrogen has 7 electrons. Two of these are in the 1st shell and the remaining five are in the second shell. The second shell contains four orbitals, three of these will have a single electron (that’s why nitrogen forms three bonds). The fourth is full, with two electrons. ...
... alternative energy sources are becoming exceedingly important. Hydrogen is a promising alternative fuel because of its clean, renewable and high energy content of 122 kJ g – 1 which is 2.75 times greater than the hydrocarbon fuels [1]. For hydrogen production, the biological process is the most attr ...
Enzymes and Temperature
... Small changed in pH either side of the optimum will only slow the rate of reaction. This is because the shape of the active site is disrupted, but ...
... Small changed in pH either side of the optimum will only slow the rate of reaction. This is because the shape of the active site is disrupted, but ...
Vocabulary CHEM121
... All the elements in this group exist as homonuclear diatomic molecules: H2, F2, Cl2, Br2, I2. Only the elements in air (Oxygen*, O2 and Nitrogen*, N2) do the same. When the halogens form monatomic anions (negative ions), they have a charge of -1. The suffix “-ide” is used to indicate when this is th ...
... All the elements in this group exist as homonuclear diatomic molecules: H2, F2, Cl2, Br2, I2. Only the elements in air (Oxygen*, O2 and Nitrogen*, N2) do the same. When the halogens form monatomic anions (negative ions), they have a charge of -1. The suffix “-ide” is used to indicate when this is th ...
Medical Microbiology Lecture 5 Third class/ Dentistry College The
... 1- Facultative anaerobes. These bacteria can oxidize nutrient substrates by means of both respiration and fermentation. 2- Obligate aerobes. These bacteria can only reproduce in the presence of O2. Aerobic organisms possess cytochromes and cytochrome oxidase, which are involved in the process of oxi ...
... 1- Facultative anaerobes. These bacteria can oxidize nutrient substrates by means of both respiration and fermentation. 2- Obligate aerobes. These bacteria can only reproduce in the presence of O2. Aerobic organisms possess cytochromes and cytochrome oxidase, which are involved in the process of oxi ...
Document
... Complete the table that shows the reaction, if any, of the oxides with acid and alkali. Indicate a reaction with "R" and no reaction with "NR". ...
... Complete the table that shows the reaction, if any, of the oxides with acid and alkali. Indicate a reaction with "R" and no reaction with "NR". ...
The Atom
... Nils Bohr suggested that emitted or absorbed radiation corresponds to changes in the orbits of electrons about the nucleus. Moreover, the electrons can not orbit at any distance from the nucleus, but exist only in specific “allowed” orbits. To explain the allowed orbits, Bohr postulated that electro ...
... Nils Bohr suggested that emitted or absorbed radiation corresponds to changes in the orbits of electrons about the nucleus. Moreover, the electrons can not orbit at any distance from the nucleus, but exist only in specific “allowed” orbits. To explain the allowed orbits, Bohr postulated that electro ...
DNA intro There is a famous quip by Jacques Monod that “what is
... dielectric stabilizes it. As a result formation of the hydrogen bonds the different base pairs is approximately GC=6 kcal/mol AT=4.5 kcal/mol One little-appreciated fact about hydrogen bonding by nucleic acid bases is the large number of different combinations of hydrogen bonds through which nucleic ...
... dielectric stabilizes it. As a result formation of the hydrogen bonds the different base pairs is approximately GC=6 kcal/mol AT=4.5 kcal/mol One little-appreciated fact about hydrogen bonding by nucleic acid bases is the large number of different combinations of hydrogen bonds through which nucleic ...
Hydrogen, Alkalis, and Alkaline Earths
... Same overall result as burning methane: same energy out, same CO2 out. To be clean, H2 must come from something other than fossil fuels. ...
... Same overall result as burning methane: same energy out, same CO2 out. To be clean, H2 must come from something other than fossil fuels. ...
Name……………………………………............................. Index number
... 17. A student prepared ammonia gas and allowed it to pass into a solution of zinc sulphate as shown in the set- up below. ...
... 17. A student prepared ammonia gas and allowed it to pass into a solution of zinc sulphate as shown in the set- up below. ...
Welcome to AP Chemistry!
... 1. The oxidation number of any uncombined element is O. 2. The oxidation number of a monatomic ion equal the charge on the ion. 3. The more electronegative element in a binary compound is assigned the number equal to the charge it would have if it were an ion. 4. The oxidation number of fluorine in ...
... 1. The oxidation number of any uncombined element is O. 2. The oxidation number of a monatomic ion equal the charge on the ion. 3. The more electronegative element in a binary compound is assigned the number equal to the charge it would have if it were an ion. 4. The oxidation number of fluorine in ...
Lectures 1-3: Review of forces and elementary statistical mechanics
... Covalent bonds between hydrogens and electronegative atoms can be quite polarized, with the hydrogen atom effectively having a significant positive partial charge. Because of their relatively small size, these positively polarized hydrogen atoms can interact strongly with electronegative atoms such ...
... Covalent bonds between hydrogens and electronegative atoms can be quite polarized, with the hydrogen atom effectively having a significant positive partial charge. Because of their relatively small size, these positively polarized hydrogen atoms can interact strongly with electronegative atoms such ...
... of the simplest substances, the chemical elements. Familiar elements include hydrogen, helium, sodium, chlorine, iron, lead, carbon, nitrogen and oxygen. The smallest unit into which an element may be divided while keeping all of the characteristics of that element is an atom. Each chemical element ...
development and validation of rp
... Chromatographic system and conditions: Analysis was carried out on Shimadzu HPLC system, 10 AT detector with ...
... Chromatographic system and conditions: Analysis was carried out on Shimadzu HPLC system, 10 AT detector with ...
biochemistry
... ion (H+) and a hydroxide ion (OH-). • As a result, pure water always has a minute, but equal concentrations of H + and OH- ions. Any substance that, when added to water, changes the concentration of H + or OH – is known as an acid or a base, depending on the change to the ...
... ion (H+) and a hydroxide ion (OH-). • As a result, pure water always has a minute, but equal concentrations of H + and OH- ions. Any substance that, when added to water, changes the concentration of H + or OH – is known as an acid or a base, depending on the change to the ...
Hydrogen

Hydrogen is a chemical element with chemical symbol H and atomic number 1. With an atomic weight of 7000100794000000000♠1.00794 u, hydrogen is the lightest element on the periodic table. Its monatomic form (H) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Non-remnant stars are mainly composed of hydrogen in its plasma state. The most common isotope of hydrogen, termed protium (name rarely used, symbol 1H), has one proton and no neutrons.The universal emergence of atomic hydrogen first occurred during the recombination epoch. At standard temperature and pressure, hydrogen is a colorless, odorless, tasteless, non-toxic, nonmetallic, highly combustible diatomic gas with the molecular formula H2. Since hydrogen readily forms covalent compounds with most non-metallic elements, most of the hydrogen on Earth exists in molecular forms such as in the form of water or organic compounds. Hydrogen plays a particularly important role in acid–base reactions as many acid-base reactions involve the exchange of protons between soluble molecules. In ionic compounds, hydrogen can take the form of a negative charge (i.e., anion) when it is known as a hydride, or as a positively charged (i.e., cation) species denoted by the symbol H+. The hydrogen cation is written as though composed of a bare proton, but in reality, hydrogen cations in ionic compounds are always more complex species than that would suggest. As the only neutral atom for which the Schrödinger equation can be solved analytically, study of the energetics and bonding of the hydrogen atom has played a key role in the development of quantum mechanics.Hydrogen gas was first artificially produced in the early 16th century, via the mixing of metals with acids. In 1766–81, Henry Cavendish was the first to recognize that hydrogen gas was a discrete substance, and that it produces water when burned, a property which later gave it its name: in Greek, hydrogen means ""water-former"".Industrial production is mainly from the steam reforming of natural gas, and less often from more energy-intensive hydrogen production methods like the electrolysis of water. Most hydrogen is employed near its production site, with the two largest uses being fossil fuel processing (e.g., hydrocracking) and ammonia production, mostly for the fertilizer market. Hydrogen is a concern in metallurgy as it can embrittle many metals, complicating the design of pipelines and storage tanks.