1.1 to 1.4
... elements - in groups 1,2, and 13-18 g. transition elements in groups 3-12 - exhibit a wide range of properties ...
... elements - in groups 1,2, and 13-18 g. transition elements in groups 3-12 - exhibit a wide range of properties ...
Structure of the Atom Cornell Notes (pg
... carbon? Iron? (see periodic table) What is an isotope? (p.175) Some isotopes are radioactive. What is a radioactive atom? (p. 176) What is the mass number? Why aren’t electrons included in the mass number?(p. 176) How do you write the name of a specific isotope? (p. 177) ...
... carbon? Iron? (see periodic table) What is an isotope? (p.175) Some isotopes are radioactive. What is a radioactive atom? (p. 176) What is the mass number? Why aren’t electrons included in the mass number?(p. 176) How do you write the name of a specific isotope? (p. 177) ...
Study Guide 1-3
... isotopic notation given a drawing of an atom or isotopic notation. You must also be able to determine number of protons, neutrons, and electrons present. A) ...
... isotopic notation given a drawing of an atom or isotopic notation. You must also be able to determine number of protons, neutrons, and electrons present. A) ...
Chapter 3 Practice Test
... _________ 16. Of the following particles, those not found in the nucleus of an atom are a. protons. b. neutrons. c. electrons. d. protons and neutrons. _________ 17. Different atoms of the same element may have different a. numbers of protons. b. atomic numbers. c. atomic masses. d. numbers of elect ...
... _________ 16. Of the following particles, those not found in the nucleus of an atom are a. protons. b. neutrons. c. electrons. d. protons and neutrons. _________ 17. Different atoms of the same element may have different a. numbers of protons. b. atomic numbers. c. atomic masses. d. numbers of elect ...
Average Atomic Mass notes
... – We have different isotopes of the same atom • They are the same type of atom but they have a different mass because they have a different number of neutrons ...
... – We have different isotopes of the same atom • They are the same type of atom but they have a different mass because they have a different number of neutrons ...
Chapter 4 - Germainium.net
... • What happens to the atomic mass number and the atomic number of a radioisotope when it undergoes alpha emission? • High speed electrons emitted by an unstable nucleus are ________ particles. • What isotope of what element is produced if krypton-81 undergoes beta decay? Write out the nuclear reacti ...
... • What happens to the atomic mass number and the atomic number of a radioisotope when it undergoes alpha emission? • High speed electrons emitted by an unstable nucleus are ________ particles. • What isotope of what element is produced if krypton-81 undergoes beta decay? Write out the nuclear reacti ...
Learning About The Atom and Atomic Structure
... The Nucleus of an atom contains both protons and neutrons. Protons have a positive charge of 1+, while a neutron has no charge, but both have relatively the same mass. An electron, on the other hand, has a charge of 1- , and have a mass much lower than that of a proton or neutron (will give mass ...
... The Nucleus of an atom contains both protons and neutrons. Protons have a positive charge of 1+, while a neutron has no charge, but both have relatively the same mass. An electron, on the other hand, has a charge of 1- , and have a mass much lower than that of a proton or neutron (will give mass ...
Unit 3 Rev Pckt - Old Saybrook Public Schools
... 25. Radiation consisting of particles with a charge of +2 and a mass of 4. 26. Alpha radiation has particles identical to a nucleus. 27 . High energy radiation, not consisting of particles, with 0 charge and 0 28. The particles in beta radiation are actually ...
... 25. Radiation consisting of particles with a charge of +2 and a mass of 4. 26. Alpha radiation has particles identical to a nucleus. 27 . High energy radiation, not consisting of particles, with 0 charge and 0 28. The particles in beta radiation are actually ...
History of the Atom
... We identify isotopes by their mass number. The mass number is the total number of protons and neutrons it ...
... We identify isotopes by their mass number. The mass number is the total number of protons and neutrons it ...
Atomic Structure Notes Packet
... John Dalton (1766-1844): used Democritus’s ideas and proposed the first atomic theory in 1803 based on experimentation, which included the following ideas: All ____________ consist of tiny indivisible particles called atoms. Atoms of the same element are ____________, different from those of any oth ...
... John Dalton (1766-1844): used Democritus’s ideas and proposed the first atomic theory in 1803 based on experimentation, which included the following ideas: All ____________ consist of tiny indivisible particles called atoms. Atoms of the same element are ____________, different from those of any oth ...
Atoms and Molecules
... Atoms are NOT indivisible – they can be broken apart into P+, neutrons, and e-. 2. Atoms can be changed from one element to another, but not by chemical means (chemical reactions). Can do it by nuclear reactions. 3. Atoms of the same element are NOT all exactly alike isotopes ...
... Atoms are NOT indivisible – they can be broken apart into P+, neutrons, and e-. 2. Atoms can be changed from one element to another, but not by chemical means (chemical reactions). Can do it by nuclear reactions. 3. Atoms of the same element are NOT all exactly alike isotopes ...
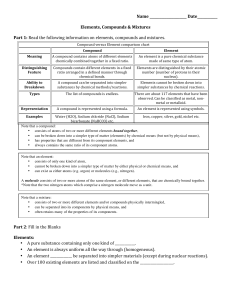
Compound vs Element chart
... • has properties that are different from its component elements, and • always contains the same ratio of its component atoms. Note that an element: • consists of only one kind of atom, • cannot be broken down into a simpler type of matter by either physical or chemical means, and • can exist as eith ...
... • has properties that are different from its component elements, and • always contains the same ratio of its component atoms. Note that an element: • consists of only one kind of atom, • cannot be broken down into a simpler type of matter by either physical or chemical means, and • can exist as eith ...
NUCLEAR CHEMISTRY AND RADIOACTIVE DECAY
... As beta decay was studied over the years following 1899, it was found that the same exact beta decay produced an electron with variable energies. For example, let us study Li-8 becoming Be-8. Each atom of Li-8 produces an electron and the theory says all the electrons should have the same energy. Th ...
... As beta decay was studied over the years following 1899, it was found that the same exact beta decay produced an electron with variable energies. For example, let us study Li-8 becoming Be-8. Each atom of Li-8 produces an electron and the theory says all the electrons should have the same energy. Th ...
Chapter 1000A - U of L Class Index
... 3. All atoms of a given element are identical in mass and other properties. – *Different elements have different kinds of atoms and differ in mass from others. 4. Compounds consist of elements combined in small whole number ratios. ...
... 3. All atoms of a given element are identical in mass and other properties. – *Different elements have different kinds of atoms and differ in mass from others. 4. Compounds consist of elements combined in small whole number ratios. ...
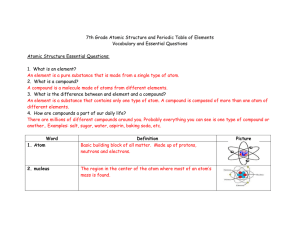
7th Grade Atomic Structure and Periodic Table of Elements
... Periodic Table of Elements Essential Questions: 1. What is the PTE? A chart of elements arranged into rows and columns according to their physical and chemical properties. 2. How is the PTE organized? Today, the periodic table organizes the elements in horizontal rows, or periods, by order of incre ...
... Periodic Table of Elements Essential Questions: 1. What is the PTE? A chart of elements arranged into rows and columns according to their physical and chemical properties. 2. How is the PTE organized? Today, the periodic table organizes the elements in horizontal rows, or periods, by order of incre ...
Atoms and Isotopes
... They are atoms of the same element that have different Number of Neutrons but must have the same number of Protons. ...
... They are atoms of the same element that have different Number of Neutrons but must have the same number of Protons. ...
Atoms, Molecules and Ions Part 2
... • The mass of an atom is decided by the number of protons and the number of neutrons present in the atomic nucleus. • Since atoms are so tiny, using an ordinary unit such as gram or kilogram is inconvenient and the atomic masses are usually reported in a special unit called atomic mass unit (amu). ...
... • The mass of an atom is decided by the number of protons and the number of neutrons present in the atomic nucleus. • Since atoms are so tiny, using an ordinary unit such as gram or kilogram is inconvenient and the atomic masses are usually reported in a special unit called atomic mass unit (amu). ...
Average Atomic Mass
... 26. a multivitamin tablet 27. distilled water 28. tap water Classify the following as A/F. homogeneous mixture or B/G. heterogeneous mixture. 29. chunky peanut butter 30. a solution of copper (II) sulfate 31. a bag of trail mix ...
... 26. a multivitamin tablet 27. distilled water 28. tap water Classify the following as A/F. homogeneous mixture or B/G. heterogeneous mixture. 29. chunky peanut butter 30. a solution of copper (II) sulfate 31. a bag of trail mix ...
notes - van Maarseveen
... Atoms are made up of three different types of subatomic particles o The nucleus contains positive ___________________ and neutral ________________________ o Negatively charged ___________________________ are found outside the nucleus. The electrons are much smaller than the protons and neutrons. Ato ...
... Atoms are made up of three different types of subatomic particles o The nucleus contains positive ___________________ and neutral ________________________ o Negatively charged ___________________________ are found outside the nucleus. The electrons are much smaller than the protons and neutrons. Ato ...
Masses of Atoms and the Periodic Table
... neutrons in an atom is the mass number – A fluoride atom with 9 protons and 10 neutrons has a mass number of _________ – A sodium atom with 11 protons and 12 neutrons has a mass number of _________ – A hydrogen atom with 1 proton and 0 neutrons has a mass number of ________ ...
... neutrons in an atom is the mass number – A fluoride atom with 9 protons and 10 neutrons has a mass number of _________ – A sodium atom with 11 protons and 12 neutrons has a mass number of _________ – A hydrogen atom with 1 proton and 0 neutrons has a mass number of ________ ...
Discussion Notes (cont.)
... How are the smallest bits of matter described? • All matter is made up of extremely small particles called atoms. These particles are too small to be seen directly, even under a microscope. • The atom is composed of even smaller particles called protons, neutrons, and electrons. The protons and neut ...
... How are the smallest bits of matter described? • All matter is made up of extremely small particles called atoms. These particles are too small to be seen directly, even under a microscope. • The atom is composed of even smaller particles called protons, neutrons, and electrons. The protons and neut ...
Unit 5 Notes
... If you started with 24.0 g of phosphorus-32, how many grams of the isotope remain at the end four half-lives? E. ...
... If you started with 24.0 g of phosphorus-32, how many grams of the isotope remain at the end four half-lives? E. ...
LBC1_Sec3_Unit01_Alchemy
... How are the smallest bits of matter described? • All matter is made up of extremely small particles called atoms. These particles are too small to be seen directly, even under a microscope. • The atom is composed of even smaller particles called protons, neutrons, and electrons. The protons and neut ...
... How are the smallest bits of matter described? • All matter is made up of extremely small particles called atoms. These particles are too small to be seen directly, even under a microscope. • The atom is composed of even smaller particles called protons, neutrons, and electrons. The protons and neut ...
Physical Science Chapter 3 Test
... their properties will emerge in a regular pattern. 12. Because atoms of elements in the same group of the periodic table have the same number of ____________________, they have similar properties. 13. Some elements are highly ____________________ because their outermost energy levels are only partia ...
... their properties will emerge in a regular pattern. 12. Because atoms of elements in the same group of the periodic table have the same number of ____________________, they have similar properties. 13. Some elements are highly ____________________ because their outermost energy levels are only partia ...
Neptunium
.png?width=300)
Neptunium is a chemical element with symbol Np and atomic number 93. A radioactive actinide metal, neptunium is the first transuranic element. Its position in the periodic table just after uranium, named after the planet Uranus, led to it being named after Neptune, the next planet beyond Uranus. A neptunium atom has 93 protons and 93 electrons, of which seven are valence electrons. Neptunium metal is silvery and tarnishes when exposed to air. The element occurs in three allotropic forms and it normally exhibits five oxidation states, ranging from +3 to +7. It is radioactive, pyrophoric, and can accumulate in bones, which makes the handling of neptunium dangerous.Although many false claims of its discovery were made over the years, the element was first synthesized by Edwin McMillan and Philip H. Abelson at the Berkeley Radiation Laboratory in 1940. Since then, most neptunium has been and still is produced by neutron irradiation of uranium in nuclear reactors. The vast majority is generated as a by-product in conventional nuclear power reactors. While neptunium itself has no commercial uses at present, it is widely used as a precursor for the formation of plutonium-238, used in radioisotope thermal generators. Neptunium has also been used in detectors of high-energy neutrons.The most stable isotope of neptunium, neptunium-237, is a by-product of nuclear reactors and plutonium production. It, and the isotope neptunium-239, are also found in trace amounts in uranium ores due to neutron capture reactions and beta decay.