The average atomic mass of an element is the sum of the
... atoms exist that contain either one or two neutrons, but both atoms have two protons. These different types of helium atoms have different masses (3 or 4 atomic mass units), and they are called isotopes. For any given isotope, the sum of the numbers of protons and neutrons in the nucleus is called t ...
... atoms exist that contain either one or two neutrons, but both atoms have two protons. These different types of helium atoms have different masses (3 or 4 atomic mass units), and they are called isotopes. For any given isotope, the sum of the numbers of protons and neutrons in the nucleus is called t ...
CHEMISTRY FALL FINAL PRACTICE 2016
... information concerning the element with 15 protons: a. Get the atomic number _________ b. Identify the element __________________ c. Find the mass number of the most common isotope of an element _________ d. Get how many neutrons the most common isotope has _________ e. How many valence electrons an ...
... information concerning the element with 15 protons: a. Get the atomic number _________ b. Identify the element __________________ c. Find the mass number of the most common isotope of an element _________ d. Get how many neutrons the most common isotope has _________ e. How many valence electrons an ...
Reading Assignment Worksheet on Atoms - District 196 e
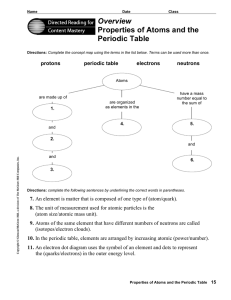
... Directions: Complete the concept map using the terms in the list below. Terms can be used more than once. ...
... Directions: Complete the concept map using the terms in the list below. Terms can be used more than once. ...
Atoms, Elements, and the Periodic Table Part 1: The Atomic Model
... same family, they share similar characteristics. Each element family has a unique name as well! Let’s look at them now… ...
... same family, they share similar characteristics. Each element family has a unique name as well! Let’s look at them now… ...
Atoms, Elements, and the Periodic Table Part 1: The Atomic Model
... same family, they share similar characteristics. Each element family has a unique name as well! Let’s look at them now… ...
... same family, they share similar characteristics. Each element family has a unique name as well! Let’s look at them now… ...
1 - WordPress.com
... Assume the following three isotopes of element Z exist: Z-248, Z-252, and Z-259. If the atomic mass of Z is 258.63 amu, which of these isotopes is most abundant? Since the atomic mass is the weighted average, the most abundant will be the isotope with the mass number closest to the atomic mass: Z-25 ...
... Assume the following three isotopes of element Z exist: Z-248, Z-252, and Z-259. If the atomic mass of Z is 258.63 amu, which of these isotopes is most abundant? Since the atomic mass is the weighted average, the most abundant will be the isotope with the mass number closest to the atomic mass: Z-25 ...
Test #1 Study Guide
... charge of the metal in roman numeral form inside brackets between the metal and the nonmetal. Molecular compounds are made up of two nonmetals and are easily distinguished from ionic compounds because of this. Naming of molecular ions requires you to know the prefixes that indicate the number of a c ...
... charge of the metal in roman numeral form inside brackets between the metal and the nonmetal. Molecular compounds are made up of two nonmetals and are easily distinguished from ionic compounds because of this. Naming of molecular ions requires you to know the prefixes that indicate the number of a c ...
Chemical Element
... Bang nucleosynthesis during the first 20 minutes of the universe[9] in a ratio of around 3:1 by mass (approximately 12:1 by number of atoms). Almost all other elements found in nature, including some further hydrogen and helium created since then, were made by various natural or (at times) artificia ...
... Bang nucleosynthesis during the first 20 minutes of the universe[9] in a ratio of around 3:1 by mass (approximately 12:1 by number of atoms). Almost all other elements found in nature, including some further hydrogen and helium created since then, were made by various natural or (at times) artificia ...
The average atomic mass of an element is the sum of the
... An element can have differing numbers of neutrons in itsnucleus, but it always has the same number of protons. The versions of an element with different neutrons have different masses and are called isotopes. The average atomic mass for an element is calculated by summing the masses of the element's ...
... An element can have differing numbers of neutrons in itsnucleus, but it always has the same number of protons. The versions of an element with different neutrons have different masses and are called isotopes. The average atomic mass for an element is calculated by summing the masses of the element's ...
Chemistry Review- Answer all questions on loose
... Na has only one valence electron (group 1-Alkali metals) making it violently reactive. Na is very close to a full electron shell, it only has to lose one electron and that will happen more readily than Mg losing two (group 2 –Alkaline earth metals). It is harder for Mg to lose two electrons so the r ...
... Na has only one valence electron (group 1-Alkali metals) making it violently reactive. Na is very close to a full electron shell, it only has to lose one electron and that will happen more readily than Mg losing two (group 2 –Alkaline earth metals). It is harder for Mg to lose two electrons so the r ...
Protons
... Compound: a substance that is composed of atoms of two or more different elements that are chemically combined ( due to chemical reactions) Example: Table salt, or NaCl, is a compound. ...
... Compound: a substance that is composed of atoms of two or more different elements that are chemically combined ( due to chemical reactions) Example: Table salt, or NaCl, is a compound. ...
Ch 30 Nuclear Physics
... If daughter product was present when the rock formed, later analysis of the rock will result in an inaccurate parent to daughter ratio. Sometimes it may be possible to determine the amount of daughter nuclide initially present. In order to use radiometric dating techniques at all, the rocks must act ...
... If daughter product was present when the rock formed, later analysis of the rock will result in an inaccurate parent to daughter ratio. Sometimes it may be possible to determine the amount of daughter nuclide initially present. In order to use radiometric dating techniques at all, the rocks must act ...
PowerPoint_Atomic Structure
... suggested that all matter was made up of tiny spheres that were able to bounce around with perfect elasticity and called them ...
... suggested that all matter was made up of tiny spheres that were able to bounce around with perfect elasticity and called them ...
An atom - Ms. Buicke maths and science
... The total number of protons and neutrons in an atom of that element. ...
... The total number of protons and neutrons in an atom of that element. ...
Final Exam Review Day 1 - teacherstroh
... If you keep breaking down matter further and further, you will get to something called the… ...
... If you keep breaking down matter further and further, you will get to something called the… ...
Physical Science
... Elements and compounds ARE related. It takes two or more elements chemically combined to make a compound. B. An element can be broken down into compounds. Elements cannot be broken down into compounds, but compounds can be broken down into elements through a chemical reaction. C. A compound is made ...
... Elements and compounds ARE related. It takes two or more elements chemically combined to make a compound. B. An element can be broken down into compounds. Elements cannot be broken down into compounds, but compounds can be broken down into elements through a chemical reaction. C. A compound is made ...
atomic number - s3.amazonaws.com
... might expect them to repel each other just as the north ends of two magnets tend to push each other apart. • It is true that they normally would do just that. • However, when they are packed together in the nucleus with the neutrons, an even stronger binding force takes over. • That force is called ...
... might expect them to repel each other just as the north ends of two magnets tend to push each other apart. • It is true that they normally would do just that. • However, when they are packed together in the nucleus with the neutrons, an even stronger binding force takes over. • That force is called ...
Chap 03A-Atoms and Elements.pptx
... Ø explains the difference between an element and a compound. Ø explains two scientific laws, and Ø predicts a new scientific law. ...
... Ø explains the difference between an element and a compound. Ø explains two scientific laws, and Ø predicts a new scientific law. ...
Atomic Structure
... balance and measured everything which lead to the Law of conservation of Mass • He named oxygen which means “acid former” and helped determine that air is a mixture not an element ...
... balance and measured everything which lead to the Law of conservation of Mass • He named oxygen which means “acid former” and helped determine that air is a mixture not an element ...
atoms - My CCSD
... identical. Atoms of any one element are different from those of any other element. ...
... identical. Atoms of any one element are different from those of any other element. ...
Properties of Atoms and the Periodic Table
... 7. An element is matter that is composed of one type of (atom/quark). 8. The unit of measurement used for atomic particles is the (atom size/atomic mass unit), 9. Atoms of the same element that have different numbers of neutrons are called ...
... 7. An element is matter that is composed of one type of (atom/quark). 8. The unit of measurement used for atomic particles is the (atom size/atomic mass unit), 9. Atoms of the same element that have different numbers of neutrons are called ...
Atoms, Elements, and Ions
... • The atom is extremely small. One teaspoon of water has 3 times as many atoms as the Atlantic Ocean has teaspoons of water. • If a large sports stadium were an atom, a marble would represent the nucleus. ...
... • The atom is extremely small. One teaspoon of water has 3 times as many atoms as the Atlantic Ocean has teaspoons of water. • If a large sports stadium were an atom, a marble would represent the nucleus. ...
Neptunium
.png?width=300)
Neptunium is a chemical element with symbol Np and atomic number 93. A radioactive actinide metal, neptunium is the first transuranic element. Its position in the periodic table just after uranium, named after the planet Uranus, led to it being named after Neptune, the next planet beyond Uranus. A neptunium atom has 93 protons and 93 electrons, of which seven are valence electrons. Neptunium metal is silvery and tarnishes when exposed to air. The element occurs in three allotropic forms and it normally exhibits five oxidation states, ranging from +3 to +7. It is radioactive, pyrophoric, and can accumulate in bones, which makes the handling of neptunium dangerous.Although many false claims of its discovery were made over the years, the element was first synthesized by Edwin McMillan and Philip H. Abelson at the Berkeley Radiation Laboratory in 1940. Since then, most neptunium has been and still is produced by neutron irradiation of uranium in nuclear reactors. The vast majority is generated as a by-product in conventional nuclear power reactors. While neptunium itself has no commercial uses at present, it is widely used as a precursor for the formation of plutonium-238, used in radioisotope thermal generators. Neptunium has also been used in detectors of high-energy neutrons.The most stable isotope of neptunium, neptunium-237, is a by-product of nuclear reactors and plutonium production. It, and the isotope neptunium-239, are also found in trace amounts in uranium ores due to neutron capture reactions and beta decay.