Atomic Structure PowerPoint Presentation
... Atomic Number oAtomic Number = the number of protons; unique to each element and the way the periodic table is ...
... Atomic Number oAtomic Number = the number of protons; unique to each element and the way the periodic table is ...
electrons and the structure of atoms
... The ancient Greek Democritus first proposed that matter is made up of small, indivisible particles that he called atoms. John Dalton made the first accepted theory on atoms almost 2000 years after the work of Democritus. Dalton’s atomic theory included that all atoms of an element are alike, the ato ...
... The ancient Greek Democritus first proposed that matter is made up of small, indivisible particles that he called atoms. John Dalton made the first accepted theory on atoms almost 2000 years after the work of Democritus. Dalton’s atomic theory included that all atoms of an element are alike, the ato ...
Midterm Review Teacher Answer Key December 21, 2011 `see
... Use the Periodic Table of the Elements. Radon (Rn, atomic number 86) is a noble gas. It is found in Group 18. Elements in Group 18 have completed outer (valence) shells of electrons and do not readily form compounds. [1 point] 'see explanation below' 31. Base your answer on the information below. Th ...
... Use the Periodic Table of the Elements. Radon (Rn, atomic number 86) is a noble gas. It is found in Group 18. Elements in Group 18 have completed outer (valence) shells of electrons and do not readily form compounds. [1 point] 'see explanation below' 31. Base your answer on the information below. Th ...
Chapter_04_Structure_of_the_atom
... still subject to new experimental data, can be modified, and is considered successful if it can be used to make predictions that are true ...
... still subject to new experimental data, can be modified, and is considered successful if it can be used to make predictions that are true ...
Chemistry: Matter and Change
... How many neutrons, protons, and electrons does 12454Xe have? A. 124 neutrons, 54 protons, 54 electrons B. 70 neutrons, 54 protons, 54 electrons C. 124 neutrons, 70 protons, 54 electrons D. 70 neutrons, 70 protons, 54 electrons ...
... How many neutrons, protons, and electrons does 12454Xe have? A. 124 neutrons, 54 protons, 54 electrons B. 70 neutrons, 54 protons, 54 electrons C. 124 neutrons, 70 protons, 54 electrons D. 70 neutrons, 70 protons, 54 electrons ...
Elements and Compounds Chapter 3
... 7A (17) = halogens (four) NOTE: H really belongs to its own group, which is why it’s shown by itself in my periodic table! It has 1 electron in outer shell, like the 1A elements, but it’s not a metal, and reacts more like group 7A. ...
... 7A (17) = halogens (four) NOTE: H really belongs to its own group, which is why it’s shown by itself in my periodic table! It has 1 electron in outer shell, like the 1A elements, but it’s not a metal, and reacts more like group 7A. ...
Atomic Mass
... 7A (17) = halogens (four) NOTE: H really belongs to its own group, which is why it’s shown by itself in my periodic table! It has 1 electron in outer shell, like the 1A elements, but it’s not a metal, and reacts more like group 7A. ...
... 7A (17) = halogens (four) NOTE: H really belongs to its own group, which is why it’s shown by itself in my periodic table! It has 1 electron in outer shell, like the 1A elements, but it’s not a metal, and reacts more like group 7A. ...
Adaptif Atomic Theory Rutherford
... number its(the proton and electron, and happened at different element. Isobar happened equality in atomic mass that is its(the proton amounts and neutron, but differs in every proton amounts, its(the electron and neutron, isobar happened at different element. Difference of one element to other eleme ...
... number its(the proton and electron, and happened at different element. Isobar happened equality in atomic mass that is its(the proton amounts and neutron, but differs in every proton amounts, its(the electron and neutron, isobar happened at different element. Difference of one element to other eleme ...
Unit #3: ATOMIC STRUCTURE - Miss Virga`s Chemistry Class
... (1) They are positive subatomic particles and are found in the nucleus. (2) They are positive subatomic particles and are found surrounding the nucleus. (3) They are negative subatomic particles and are found in the nucleus. (4) They are negative subatomic particles and are found surrounding the nuc ...
... (1) They are positive subatomic particles and are found in the nucleus. (2) They are positive subatomic particles and are found surrounding the nucleus. (3) They are negative subatomic particles and are found in the nucleus. (4) They are negative subatomic particles and are found surrounding the nuc ...
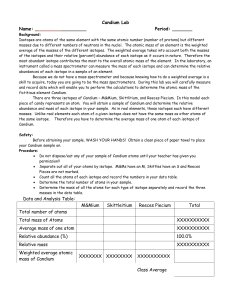
Period:______ Table Number
... 45. A(n) ELEMENT is a pure substance that can not be broken down into any other substance by some physical or chemical method and from which all more complex forms of matter or substances are made when they are combined together in different ways and in different amounts. P. 9, 70, VCR: Atoms and Mo ...
... 45. A(n) ELEMENT is a pure substance that can not be broken down into any other substance by some physical or chemical method and from which all more complex forms of matter or substances are made when they are combined together in different ways and in different amounts. P. 9, 70, VCR: Atoms and Mo ...
Document
... Any given element can have more than one isotope. To distinguish between the different isotopes of an atom, the element is named with its mass number, for example, lithium-7. Remember that the mass number is the number of protons and neutrons added together. When symbols are used to represent an iso ...
... Any given element can have more than one isotope. To distinguish between the different isotopes of an atom, the element is named with its mass number, for example, lithium-7. Remember that the mass number is the number of protons and neutrons added together. When symbols are used to represent an iso ...
Chapter 11 section 2 questions - the atom
... Electrons are the negatively charged particles found in the energy levels that surround the nucleus - this like the rings on a target! Electrons have a VERY small mass - almost inconsequential to the atomic mass. Electrons carry a negative charge and are held in place by the positively charged proto ...
... Electrons are the negatively charged particles found in the energy levels that surround the nucleus - this like the rings on a target! Electrons have a VERY small mass - almost inconsequential to the atomic mass. Electrons carry a negative charge and are held in place by the positively charged proto ...
Chapter 4 Practice Test
... d. Cathode rays were found to be made of protons. All atoms are ____. a. positively charged, with the number of protons exceeding the number of electrons b. negatively charged, with the number of electrons exceeding the number of protons c. neutral, with the number of protons equaling the number of ...
... d. Cathode rays were found to be made of protons. All atoms are ____. a. positively charged, with the number of protons exceeding the number of electrons b. negatively charged, with the number of electrons exceeding the number of protons c. neutral, with the number of protons equaling the number of ...
Atomic Structure_Bohr Flashcards
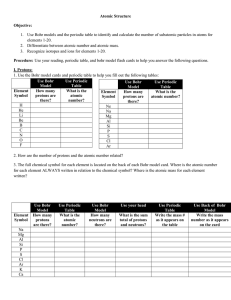
... 1. Use Bohr models and the periodic table to identify and calculate the number of subatomic particles in atoms for elements 1-20. 2. Differentiate between atomic number and atomic mass. 3. Recognize isotopes and ions for elements 1-20. Procedure: Use your reading, periodic table, and bohr model flas ...
... 1. Use Bohr models and the periodic table to identify and calculate the number of subatomic particles in atoms for elements 1-20. 2. Differentiate between atomic number and atomic mass. 3. Recognize isotopes and ions for elements 1-20. Procedure: Use your reading, periodic table, and bohr model flas ...
Students will review concepts from their quiz and then correct it at
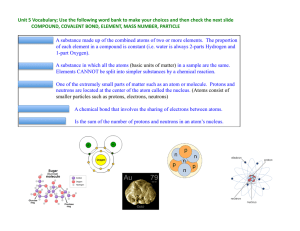
... A pure substance containing two or more kinds of __atoms__. The atoms are ___chemically___ combined in some way. Often times (but not always) they come together to form groups of atoms called molecules. A compound is always homogeneous (uniform). Compounds ___cannot___ be separated by physical means ...
... A pure substance containing two or more kinds of __atoms__. The atoms are ___chemically___ combined in some way. Often times (but not always) they come together to form groups of atoms called molecules. A compound is always homogeneous (uniform). Compounds ___cannot___ be separated by physical means ...
Flavors of the Atom
... ‣ Parts of the atom — electrons, protons, neutrons ‣ Ions differ in electron count ‣ Proton count distinguishes between elements ‣ Isotopes differ in total mass (because they differ in neutrons) ‣ Isotopic Notation ‣ Atomic Number ‣ Mass Number ‣ Electron Counts ...
... ‣ Parts of the atom — electrons, protons, neutrons ‣ Ions differ in electron count ‣ Proton count distinguishes between elements ‣ Isotopes differ in total mass (because they differ in neutrons) ‣ Isotopic Notation ‣ Atomic Number ‣ Mass Number ‣ Electron Counts ...
mack atoms - McClymonds Chemistry
... 1. Most of the atom’s mass and all of its positive charge are contained in a small core called the nucleus. 2. Most of the volume of the atom is empty space through which the tiny, negatively charged electrons are dispersed. 3. The number of negatively charged electrons outside the nucleus is equal ...
... 1. Most of the atom’s mass and all of its positive charge are contained in a small core called the nucleus. 2. Most of the volume of the atom is empty space through which the tiny, negatively charged electrons are dispersed. 3. The number of negatively charged electrons outside the nucleus is equal ...
U1 Atoms, Periodic Table, Variables, Conversions Unit 1
... 17. A certain atom has 20 electrons, 21 neutrons, and 20 protons. What is the atomic mass of the ...
... 17. A certain atom has 20 electrons, 21 neutrons, and 20 protons. What is the atomic mass of the ...
The atom CP and H ONLINE
... its atomic number and also another number- usually a decimal. This decimal number is the average atomic mass. It is not the mass number of any one atom. It represents the average mass of atoms of the element. It is an average of all the different ...
... its atomic number and also another number- usually a decimal. This decimal number is the average atomic mass. It is not the mass number of any one atom. It represents the average mass of atoms of the element. It is an average of all the different ...
Interactive Notebook 2 for 2011-2012
... which are radioactive, are separated for reasons of practical display from the main body of the periodic table. As a general rule elements in both series appear to have three electrons available for bonding. They combine with halogens to form compounds with the general formula MX3, such as LaFz3. Th ...
... which are radioactive, are separated for reasons of practical display from the main body of the periodic table. As a general rule elements in both series appear to have three electrons available for bonding. They combine with halogens to form compounds with the general formula MX3, such as LaFz3. Th ...
Document
... assumptions about atoms and matter, which together make up modern atomic theory: 1. All matter is composed of atoms. 2. The atoms of a given element differ from the atoms of all other elements. 3. Chemical compounds consist of atoms combined in specific ratios. 4. Chemical reactions change only the ...
... assumptions about atoms and matter, which together make up modern atomic theory: 1. All matter is composed of atoms. 2. The atoms of a given element differ from the atoms of all other elements. 3. Chemical compounds consist of atoms combined in specific ratios. 4. Chemical reactions change only the ...
Neptunium
.png?width=300)
Neptunium is a chemical element with symbol Np and atomic number 93. A radioactive actinide metal, neptunium is the first transuranic element. Its position in the periodic table just after uranium, named after the planet Uranus, led to it being named after Neptune, the next planet beyond Uranus. A neptunium atom has 93 protons and 93 electrons, of which seven are valence electrons. Neptunium metal is silvery and tarnishes when exposed to air. The element occurs in three allotropic forms and it normally exhibits five oxidation states, ranging from +3 to +7. It is radioactive, pyrophoric, and can accumulate in bones, which makes the handling of neptunium dangerous.Although many false claims of its discovery were made over the years, the element was first synthesized by Edwin McMillan and Philip H. Abelson at the Berkeley Radiation Laboratory in 1940. Since then, most neptunium has been and still is produced by neutron irradiation of uranium in nuclear reactors. The vast majority is generated as a by-product in conventional nuclear power reactors. While neptunium itself has no commercial uses at present, it is widely used as a precursor for the formation of plutonium-238, used in radioisotope thermal generators. Neptunium has also been used in detectors of high-energy neutrons.The most stable isotope of neptunium, neptunium-237, is a by-product of nuclear reactors and plutonium production. It, and the isotope neptunium-239, are also found in trace amounts in uranium ores due to neutron capture reactions and beta decay.