Unit 8 Chemical Equilibrium Focusing on Acid
... dioxide is allowed to leave the system (Figure 1). Carbonated drinks that have gone “flat” because of the decomposition of carbonic acid can be carbonated again by the addition of pressurized carbon dioxide to the solution to reverse the reaction, and then capping the container to restore the origin ...
... dioxide is allowed to leave the system (Figure 1). Carbonated drinks that have gone “flat” because of the decomposition of carbonic acid can be carbonated again by the addition of pressurized carbon dioxide to the solution to reverse the reaction, and then capping the container to restore the origin ...
György Dombi Gerda Szakonyi Authors
... Visual observation of the end-point by using an acidbase indicator is simple and convenient, but it may cause several problems. Instrumental methods are being used in most of the quantitative analyses in the Pharmacopeia; for acidbase titrations, the measurement of pH can be a possible solution. P ...
... Visual observation of the end-point by using an acidbase indicator is simple and convenient, but it may cause several problems. Instrumental methods are being used in most of the quantitative analyses in the Pharmacopeia; for acidbase titrations, the measurement of pH can be a possible solution. P ...
Document
... vacancy defect. This results the decrease in density of the substance. This defect develops when a substance is heated. # Interstitial defect- When some constituent particles occupy an interstitial site, the crystal is said to have interstitial defect. This defect increases the density of the substa ...
... vacancy defect. This results the decrease in density of the substance. This defect develops when a substance is heated. # Interstitial defect- When some constituent particles occupy an interstitial site, the crystal is said to have interstitial defect. This defect increases the density of the substa ...
Cyanuric Acid and Cyanurates
... amino groups. The dissociation constants of melamine in aqueous solutions were found to be K1 = 1.26 × 10–9; K2 = 1.58 × 10–14, and K3 = 1 × 10–17 [63]. Although melamine is a weak base, it nevertheless can form salts [53, 63–70]. However, it almost always acts as a monoacidic base. The yellow needl ...
... amino groups. The dissociation constants of melamine in aqueous solutions were found to be K1 = 1.26 × 10–9; K2 = 1.58 × 10–14, and K3 = 1 × 10–17 [63]. Although melamine is a weak base, it nevertheless can form salts [53, 63–70]. However, it almost always acts as a monoacidic base. The yellow needl ...
IIT-JEE (Advanced) - Brilliant Public School Sitamarhi
... Silver salt method : (for organic acids) Basicity of an acid : No. of replacable H+ atoms in an acid (H contained to more electronegative atom is acidic) Procedure : Some known amount of silver salt (w1 gm) is heated to obtain w2 gm of while shining residue of silver. Then if the basicity of acid is ...
... Silver salt method : (for organic acids) Basicity of an acid : No. of replacable H+ atoms in an acid (H contained to more electronegative atom is acidic) Procedure : Some known amount of silver salt (w1 gm) is heated to obtain w2 gm of while shining residue of silver. Then if the basicity of acid is ...
National German Competition
... q) Write down the equation of the reaction of compound 1 with lithium dimethylcuprate and water. Give the complete names of the alcohols. Zinc organic compounds are longer known and more often used. These compounds are applied to synthesize alcohols, more exactly in the synthesis of hydroxy esters. ...
... q) Write down the equation of the reaction of compound 1 with lithium dimethylcuprate and water. Give the complete names of the alcohols. Zinc organic compounds are longer known and more often used. These compounds are applied to synthesize alcohols, more exactly in the synthesis of hydroxy esters. ...
1 General Chemistry II Jasperse Entropy, Spontaneity, and Free
... 25. Hydrogen reacts with nitrogen to form ammonia (NH3) according to the reaction 3H2(g) + N2(g) D 2NH3(g) ...
... 25. Hydrogen reacts with nitrogen to form ammonia (NH3) according to the reaction 3H2(g) + N2(g) D 2NH3(g) ...
Chapter 3 2013
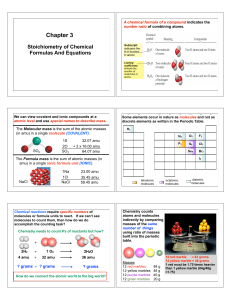
... 3. The molecular formula will be a whole number multiple of the empirical formula determined BY THE MOLAR MASS ratio ...
... 3. The molecular formula will be a whole number multiple of the empirical formula determined BY THE MOLAR MASS ratio ...
Chapter 3 2014
... Borax is the common name of a mineral sodium tetraborate, an industrial cleaning adjunct, Na2B4O7. You are given 20.0 g of borax...... (a) what is the formula mass of Na2B4O7 (b) how many moles of borax is 20.0 g? (c) how many moles of boron are present in 20.0 g Na2B4O7? (d) how many grams of boron ...
... Borax is the common name of a mineral sodium tetraborate, an industrial cleaning adjunct, Na2B4O7. You are given 20.0 g of borax...... (a) what is the formula mass of Na2B4O7 (b) how many moles of borax is 20.0 g? (c) how many moles of boron are present in 20.0 g Na2B4O7? (d) how many grams of boron ...
Experiment 7: Determination of the concentration of a solution of an
... 6. All solids must be discarded in the bins at the outer ends of each bench. Do not throw matches, paper, or any insoluble chemicals into the sink. Liquids must be discarded into the ceramic sinks or designated disposal bottles. ...
... 6. All solids must be discarded in the bins at the outer ends of each bench. Do not throw matches, paper, or any insoluble chemicals into the sink. Liquids must be discarded into the ceramic sinks or designated disposal bottles. ...
The science of chemistry is concerned
... The concept of a limiting reagent was used by the nineteenth century German chemist Justus von Liebig (1807 to 1873) to derive an important biological and ecological law. Liebig’s law of the minimum states that the essential substance available in the smallest amount relative to some critical minimu ...
... The concept of a limiting reagent was used by the nineteenth century German chemist Justus von Liebig (1807 to 1873) to derive an important biological and ecological law. Liebig’s law of the minimum states that the essential substance available in the smallest amount relative to some critical minimu ...
App. Chemistry
... Project Work/ Industrial Training/Review Articles: The evaluation of project work /Industrial Training/ Review Articles will be based on the work carried out in industry/ laboratory/ library and viva-voce/oral examination will be conducted jointly by internal & external examiner at the end of examin ...
... Project Work/ Industrial Training/Review Articles: The evaluation of project work /Industrial Training/ Review Articles will be based on the work carried out in industry/ laboratory/ library and viva-voce/oral examination will be conducted jointly by internal & external examiner at the end of examin ...
Section – B - About iTutoring
... (67). Which instrument is used to determine accurate pH of solution? pH meter is used to measure pH. (68). What is meant by hydrolysis constant ? Hydrolysis reaction is an equilibrium reaction and so its corresponding equilibrium constant can be calculated which is known as hydrolysis constant. (69) ...
... (67). Which instrument is used to determine accurate pH of solution? pH meter is used to measure pH. (68). What is meant by hydrolysis constant ? Hydrolysis reaction is an equilibrium reaction and so its corresponding equilibrium constant can be calculated which is known as hydrolysis constant. (69) ...
Liquid–liquid extraction

Liquid–liquid extraction (LLE) consists in transferring one (or more) solute(s) contained in a feed solution to another immiscible liquid (solvent). The solvent that is enriched in solute(s) is called extract. The feed solution that is depleted in solute(s) is called raffinate.Liquid–liquid extraction also known as solvent extraction and partitioning, is a method to separate compounds based on their relative solubilities in two different immiscible liquids, usually water and an organic solvent. It is an extraction of a substance from one liquid into another liquid phase. Liquid–liquid extraction is a basic technique in chemical laboratories, where it is performed using a variety of apparatus, from separatory funnels to countercurrent distribution equipment. This type of process is commonly performed after a chemical reaction as part of the work-up.The term partitioning is commonly used to refer to the underlying chemical and physical processes involved in liquid–liquid extraction, but on another reading may be fully synonymous with it. The term solvent extraction can also refer to the separation of a substance from a mixture by preferentially dissolving that substance in a suitable solvent. In that case, a soluble compound is separated from an insoluble compound or a complex matrix.Solvent extraction is used in nuclear reprocessing, ore processing, the production of fine organic compounds, the processing of perfumes, the production of vegetable oils and biodiesel, and other industries.Liquid–liquid extraction is possible in non-aqueous systems: In a system consisting of a molten metal in contact with molten salts, metals can be extracted from one phase to the other. This is related to a mercury electrode where a metal can be reduced, the metal will often then dissolve in the mercury to form an amalgam that modifies its electrochemistry greatly. For example, it is possible for sodium cations to be reduced at a mercury cathode to form sodium amalgam, while at an inert electrode (such as platinum) the sodium cations are not reduced. Instead, water is reduced to hydrogen. A detergent or fine solid can be used to stabilize an emulsion, or third phase.