Energy and Changes of State - SCIENCE
... Endothermic changes • Endothermic Reactions are reactions in which energy is taken in. • The Law of Conservation of Energy states that energy cannot be created or destroyed. The energy released in exothermic reactions was first stored in the chemical bonds of the reactants. And the energy taken in ...
... Endothermic changes • Endothermic Reactions are reactions in which energy is taken in. • The Law of Conservation of Energy states that energy cannot be created or destroyed. The energy released in exothermic reactions was first stored in the chemical bonds of the reactants. And the energy taken in ...
(1) Dissolves, accompanied by evolution of flammable gas (2
... SELECT TWO OF THE FOUR ESSAY QUESTIONS, NUMBERED 6 THROUGH 9. (Additional essays will not be scored.) ...
... SELECT TWO OF THE FOUR ESSAY QUESTIONS, NUMBERED 6 THROUGH 9. (Additional essays will not be scored.) ...
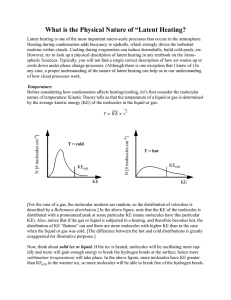
What is the Physical Nature of “Latent Heating?
... If we examine Table 3.1 in Pruppacher and Klett, we see that the percentage of broken bonds in liquid water increases as T increases. This is to be expected for reasons that we discussed in class: Higher temperatures lead to more energetic molecules & so the chance that bonds will be broken in the l ...
... If we examine Table 3.1 in Pruppacher and Klett, we see that the percentage of broken bonds in liquid water increases as T increases. This is to be expected for reasons that we discussed in class: Higher temperatures lead to more energetic molecules & so the chance that bonds will be broken in the l ...
Name ………………………………………………… Unit 7: States of
... 4. The average kinetic energy of water molecules is greatest in which of these samples? Which one has the greatest temperature? (1) 10 g of water at 35°C (2) 10 g of water at 55°C (3) 100 g of water at 25°C (4) 100 g of water at 45°C 5. Using your knowledge of chemistry and the information in Refere ...
... 4. The average kinetic energy of water molecules is greatest in which of these samples? Which one has the greatest temperature? (1) 10 g of water at 35°C (2) 10 g of water at 55°C (3) 100 g of water at 25°C (4) 100 g of water at 45°C 5. Using your knowledge of chemistry and the information in Refere ...
Chemistry
... The total 3-dimensional array of points that describe the arrangement of particles in a crystal is called a crystal lattice. ...
... The total 3-dimensional array of points that describe the arrangement of particles in a crystal is called a crystal lattice. ...
PROPERTIES OF SOLUTIONS
... Osmotic pressure obeys a law similar to the ideal gas law. For n moles, V=volume, M=molarity, R= ideal gas constant and absolute temperature, T, : V= nRT then =(n/v)RT and then = MRT If 2 solutions have the same , they are isotonic Hypotonic solutions have a lower relative to a more c ...
... Osmotic pressure obeys a law similar to the ideal gas law. For n moles, V=volume, M=molarity, R= ideal gas constant and absolute temperature, T, : V= nRT then =(n/v)RT and then = MRT If 2 solutions have the same , they are isotonic Hypotonic solutions have a lower relative to a more c ...
CARNOT CYCLE i) substance starts at with temperature T2
... • Hence at constant T and V, Helmholtz energy sets an upper limit to the non- pdV work that can be done in such a transition. ...
... • Hence at constant T and V, Helmholtz energy sets an upper limit to the non- pdV work that can be done in such a transition. ...
Chapter 13…States of Matter
... If more H2 is added to this reaction, equilibrium will shift (left/right/no shift). If Br2 is removed from this reaction, equilibrium will shift (left/right/no shift). If pressure is increased, equilibrium will shift (left/right/no shift). If temperature is decreased, equilibrium will shift (left/ri ...
... If more H2 is added to this reaction, equilibrium will shift (left/right/no shift). If Br2 is removed from this reaction, equilibrium will shift (left/right/no shift). If pressure is increased, equilibrium will shift (left/right/no shift). If temperature is decreased, equilibrium will shift (left/ri ...
Sample pages 2 PDF
... well-defined point in which the three phases can coexist; this point is named the triple point. This triple point is unique for each system/compound. Direct application of Gibbs rule of phases (Eq. 2.2) to any unary system represented in a pressure—temperature (PT) diagram, results in (Table 2.1). In ...
... well-defined point in which the three phases can coexist; this point is named the triple point. This triple point is unique for each system/compound. Direct application of Gibbs rule of phases (Eq. 2.2) to any unary system represented in a pressure—temperature (PT) diagram, results in (Table 2.1). In ...
Chemical theory for mixtures containing any number of alcohols
... Finally, the model xs compared with statistical assocmtmg fluid theory (SAFT) and the two approaches are found to be strikingly similar ...
... Finally, the model xs compared with statistical assocmtmg fluid theory (SAFT) and the two approaches are found to be strikingly similar ...
Solution FRQs Practice
... The formula and the molecular weight of an unknown hydrocarbon compound are to be determined by elemental analysis and the freezing-point depression method. (a) The hydrocarbon is found to contain 93.46 percent carbon and 6.54 percent hydrogen. Calculate the empirical formula of the unknown hydrocar ...
... The formula and the molecular weight of an unknown hydrocarbon compound are to be determined by elemental analysis and the freezing-point depression method. (a) The hydrocarbon is found to contain 93.46 percent carbon and 6.54 percent hydrogen. Calculate the empirical formula of the unknown hydrocar ...
2nd Semester Final Exam Review
... 8. Which 3 of the above solutions will conduct electricity? Why? 9. Will a precipitate form if sodium chloride is mixed with barium hydroxide? If so, what’s the ppt? Use the ppt chart in back of lab manual or on p. 860 fo your textbook. 10. Will a precipitate form if silver nitrate is mixed with cal ...
... 8. Which 3 of the above solutions will conduct electricity? Why? 9. Will a precipitate form if sodium chloride is mixed with barium hydroxide? If so, what’s the ppt? Use the ppt chart in back of lab manual or on p. 860 fo your textbook. 10. Will a precipitate form if silver nitrate is mixed with cal ...
Test 2
... Gas Constants (R): 0.08206 L@atm/K@mol -or- 8.31451 J/K@ mol Please show all work for partial credit 1. (5 points) Identify the functional groups on the following compounds ...
... Gas Constants (R): 0.08206 L@atm/K@mol -or- 8.31451 J/K@ mol Please show all work for partial credit 1. (5 points) Identify the functional groups on the following compounds ...
Chapter 14…Kinetic Theory
... (Ionic/molecular) compounds cause greater change of boiling/freezing points in solutions. Aluminum chloride (AlCl3) will dissociate into _____(how many) ions and will cause a (greater/smaller) ...
... (Ionic/molecular) compounds cause greater change of boiling/freezing points in solutions. Aluminum chloride (AlCl3) will dissociate into _____(how many) ions and will cause a (greater/smaller) ...
Unit Name: Mixtures and Solutions
... Unit Name: Mixtures and Solutions Topic/Sub-Unit: Separating Mixtures Which Standards are students learning in this unit? Standard 2.2.A Most materials are physical mixtures. Physical mixtures can be composed of different kinds of materials, each having distinct physical properties. These physical p ...
... Unit Name: Mixtures and Solutions Topic/Sub-Unit: Separating Mixtures Which Standards are students learning in this unit? Standard 2.2.A Most materials are physical mixtures. Physical mixtures can be composed of different kinds of materials, each having distinct physical properties. These physical p ...
Spring Benchmark Exam
... until no more KCl would dissolve. She then capped the clear solution and set it aside on the lab bench. After several hours she noticed the solution had become cloudy and some solid had settled to the bottom of the flask. Which statement best describes what happened? A As the solution cooled, evapor ...
... until no more KCl would dissolve. She then capped the clear solution and set it aside on the lab bench. After several hours she noticed the solution had become cloudy and some solid had settled to the bottom of the flask. Which statement best describes what happened? A As the solution cooled, evapor ...