Chapter 9 - ComSizo.com.br
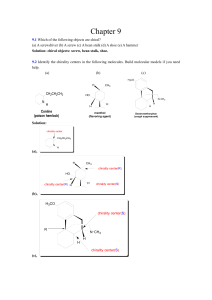
... Draw compounds that fit the following descriptions: (a) A chiral alcohol with four carbons. (b) A chiral carboxylic acid with the formula C5H10O2 (c) A compound with two chirality centers (d) A chiral aldehyde with the formula C3H5BrO ...
... Draw compounds that fit the following descriptions: (a) A chiral alcohol with four carbons. (b) A chiral carboxylic acid with the formula C5H10O2 (c) A compound with two chirality centers (d) A chiral aldehyde with the formula C3H5BrO ...
N-oxidation - WordPress.com
... Many oxidative processes (benzylic, allylic, alicyclic, or aliphatic hydroxylation) generate alcohol or carbinol intermediate. If not conjugated, are further oxidized to aldehydes (if primarily alcohols) or to ketones (if secondary alcohols). Aldehyde metabolites resulting from oxidation of primary ...
... Many oxidative processes (benzylic, allylic, alicyclic, or aliphatic hydroxylation) generate alcohol or carbinol intermediate. If not conjugated, are further oxidized to aldehydes (if primarily alcohols) or to ketones (if secondary alcohols). Aldehyde metabolites resulting from oxidation of primary ...
Chapter 1.4 Alcohols, Ethers and Thiols
... • inhibit pain signals to the brain. • like diethyl ether CH3─CH2─O─CH2─CH3 were used for over a century, but caused nausea and were flammable. • developed by the 1960’s were nonflammable. Cl F ...
... • inhibit pain signals to the brain. • like diethyl ether CH3─CH2─O─CH2─CH3 were used for over a century, but caused nausea and were flammable. • developed by the 1960’s were nonflammable. Cl F ...
chapter 22 organic and biological molecules
... These are the same compounds; they only differ by a simple rotation of the molecule. Therefore, they are not isomers of each other but instead are the same compound. ...
... These are the same compounds; they only differ by a simple rotation of the molecule. Therefore, they are not isomers of each other but instead are the same compound. ...
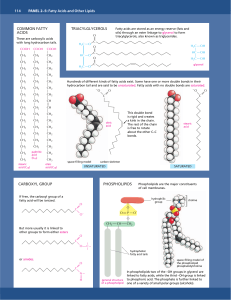
Panel 2–4 Fatty acids and other lipids
... bilayers that are the basis for all cellular membranes. ...
... bilayers that are the basis for all cellular membranes. ...
fats
... In phospholipids two of the –OH groups in glycerol are linked to fatty acids, while the third –OH group is linked to phosphoric acid. The phosphate is further linked to one of a variety of small polar groups (alcohols). ...
... In phospholipids two of the –OH groups in glycerol are linked to fatty acids, while the third –OH group is linked to phosphoric acid. The phosphate is further linked to one of a variety of small polar groups (alcohols). ...
Organic Chemistry - hrsbstaff.ednet.ns.ca
... climates, gave rise to abundant plant and animal life on land and under water. Over time, as these organisms died, the organic substances that made up their bodies were chemically transformed into the materials known today as fossil fuels. ...
... climates, gave rise to abundant plant and animal life on land and under water. Over time, as these organisms died, the organic substances that made up their bodies were chemically transformed into the materials known today as fossil fuels. ...
Chapter 12 Organic Compounds with Oxygen and Sulfur
... • do not have a polar OH group, but have a COC group • do not form hydrogen bonds • with less than four carbons are slightly soluble in water • with more than four carbons are not soluble in water ...
... • do not have a polar OH group, but have a COC group • do not form hydrogen bonds • with less than four carbons are slightly soluble in water • with more than four carbons are not soluble in water ...
Chemistry.of Organic Compounds
... the students majoring in chemistry are preparing themselves for industrial positions. Hence the practical applications and economic aspects of organic chemistry frequently are discussed in some detail. For some years textbooks have been published which present aliphatic and aromatic compounds simult ...
... the students majoring in chemistry are preparing themselves for industrial positions. Hence the practical applications and economic aspects of organic chemistry frequently are discussed in some detail. For some years textbooks have been published which present aliphatic and aromatic compounds simult ...
Document
... Functional Groups • Besides our basic hydrocarbon classes, we can add other elements/ions, groups of elements to an organic structure = Functional Groups • R = radical or, in this case, represents C • Alcohols (R-OH), ethers (R-O-R), carboxylic acids (R-COOH), aldehydes (R-COH) – what are the other ...
... Functional Groups • Besides our basic hydrocarbon classes, we can add other elements/ions, groups of elements to an organic structure = Functional Groups • R = radical or, in this case, represents C • Alcohols (R-OH), ethers (R-O-R), carboxylic acids (R-COOH), aldehydes (R-COH) – what are the other ...
Document
... Functional Groups • Besides our basic hydrocarbon classes, we can add other elements/ions, groups of elements to an organic structure = Functional Groups • R = radical or, in this case, represents C • Alcohols (R-OH), ethers (R-O-R), carboxylic acids (R-COOH), aldehydes (R-COH) – what are the other ...
... Functional Groups • Besides our basic hydrocarbon classes, we can add other elements/ions, groups of elements to an organic structure = Functional Groups • R = radical or, in this case, represents C • Alcohols (R-OH), ethers (R-O-R), carboxylic acids (R-COOH), aldehydes (R-COH) – what are the other ...
Compounds with Oxygen Atoms
... • inhibit pain signals to the brain. • like diethyl ether CH3─CH2─O─CH2─CH3 were used for over a century, but caused nausea and were flammable. • developed by the 1960’s were nonflammable. Cl F ...
... • inhibit pain signals to the brain. • like diethyl ether CH3─CH2─O─CH2─CH3 were used for over a century, but caused nausea and were flammable. • developed by the 1960’s were nonflammable. Cl F ...
Alcohol, Ethers, and Thiols
... • inhibit pain signals to the brain. • like diethyl ether CH3─CH2─O─CH2─CH3 were used for over a century, but caused nausea and were flammable. • developed by the 1960’s were nonflammable. Cl F ...
... • inhibit pain signals to the brain. • like diethyl ether CH3─CH2─O─CH2─CH3 were used for over a century, but caused nausea and were flammable. • developed by the 1960’s were nonflammable. Cl F ...
Charles-Adolphe Wurtz
... and August von Wassermann (1866-1925). It was in his laboratory that Paul Emile Boisbaudran (18381912) discovered gallium in 1875 (André, 1956). Besides being a brilliant lecturer and laboratory teacher Wurtz excelled as a writer. From 1852 to1872 he was responsible for the section devoted to foreig ...
... and August von Wassermann (1866-1925). It was in his laboratory that Paul Emile Boisbaudran (18381912) discovered gallium in 1875 (André, 1956). Besides being a brilliant lecturer and laboratory teacher Wurtz excelled as a writer. From 1852 to1872 he was responsible for the section devoted to foreig ...
Naming Organic Compounds I
... ten alkanes, given in Table 1, should be memorized. Larger alkanes, such as icosane (C20H42), have more complicated names and are outside the scope of this course. If one hydrogen atom is removed from an alkane, the remaining part of the molecule is called an alkyl group. Alkyl groups are named by r ...
... ten alkanes, given in Table 1, should be memorized. Larger alkanes, such as icosane (C20H42), have more complicated names and are outside the scope of this course. If one hydrogen atom is removed from an alkane, the remaining part of the molecule is called an alkyl group. Alkyl groups are named by r ...
Document
... •Epoxides can be named in three different ways—As epoxyalkanes, oxiranes, or alkene oxides. • Epoxides bonded to a chain of carbon atoms can also be named as derivatives of oxirane, the simplest epoxide having two carbons and one oxygen atom in a ring. • The oxirane ring is numbered to put the O at ...
... •Epoxides can be named in three different ways—As epoxyalkanes, oxiranes, or alkene oxides. • Epoxides bonded to a chain of carbon atoms can also be named as derivatives of oxirane, the simplest epoxide having two carbons and one oxygen atom in a ring. • The oxirane ring is numbered to put the O at ...
Topic 22 Notes
... 2. Single and multiple covalent bonds a. Single covalent bonds (1) Where one pair of electrons is shared between two atoms. (2) Symbolized by one pair of dots or by a dash. b. Double covalent bonds (1) Where two pairs of electrons are shared between two atoms. (2) Symbolized by two pairs of dots or ...
... 2. Single and multiple covalent bonds a. Single covalent bonds (1) Where one pair of electrons is shared between two atoms. (2) Symbolized by one pair of dots or by a dash. b. Double covalent bonds (1) Where two pairs of electrons are shared between two atoms. (2) Symbolized by two pairs of dots or ...
CHAPTER 22 ORGANIC AND BIOLOGICAL MOLECULES 1
... Cyclic alkanes: general formula CnH2n (if only one ring is present in the compound); all carbons are sp3 hybridized; prefers 109.5 bond angles, but rings with three carbons or four carbons or five carbons are forced into bond angles less than 109.5. In cyclopropane, a ring compound made up of thre ...
... Cyclic alkanes: general formula CnH2n (if only one ring is present in the compound); all carbons are sp3 hybridized; prefers 109.5 bond angles, but rings with three carbons or four carbons or five carbons are forced into bond angles less than 109.5. In cyclopropane, a ring compound made up of thre ...
Complete Solution Manual
... Cyclic alkanes: general formula CnH2n (if only one ring is present in the compound); all carbons are sp3 hybridized; prefers 109.5 bond angles, but rings with three carbons or four carbons or five carbons are forced into bond angles less than 109.5. In cyclopropane, a ring compound made up of thre ...
... Cyclic alkanes: general formula CnH2n (if only one ring is present in the compound); all carbons are sp3 hybridized; prefers 109.5 bond angles, but rings with three carbons or four carbons or five carbons are forced into bond angles less than 109.5. In cyclopropane, a ring compound made up of thre ...
Polyphenolic and essential oil composition of Mentha and
... high-‐performance liquid chromatography Herpes simplex virus type-‐1 concentration giving 90% inhibition C. pneumoniae strain Kajaani-‐7 lipopolysaccharide Mentha minimal bactericidal concentration negative ion mode ...
... high-‐performance liquid chromatography Herpes simplex virus type-‐1 concentration giving 90% inhibition C. pneumoniae strain Kajaani-‐7 lipopolysaccharide Mentha minimal bactericidal concentration negative ion mode ...
CH 3 - bYTEBoss
... covalently bonded to an aliphatic carbon atom. • The first four halogens— fluorine, chlorine, bromine, and iodine—are found in many organic compounds. ...
... covalently bonded to an aliphatic carbon atom. • The first four halogens— fluorine, chlorine, bromine, and iodine—are found in many organic compounds. ...
Document
... • Contains an –SH (sulfhydryl) group. • Is named by selecting the longest carbon chain that contain the -SH. We add -thiol to the name of the parent alkane. • Parent chain is numbered from the end nearest to the -SH group. CH3─S─H ...
... • Contains an –SH (sulfhydryl) group. • Is named by selecting the longest carbon chain that contain the -SH. We add -thiol to the name of the parent alkane. • Parent chain is numbered from the end nearest to the -SH group. CH3─S─H ...
Silylation Overview - Sigma
... always a necessity for up-to-date reviews. The silylation of organic compounds for synthetic and analytical purposes, an important part of organosilicon chemistry, is the subject matter of this totally revised and enlarged monograph. The term “silylation” is defined as the substitution of a hydrogen ...
... always a necessity for up-to-date reviews. The silylation of organic compounds for synthetic and analytical purposes, an important part of organosilicon chemistry, is the subject matter of this totally revised and enlarged monograph. The term “silylation” is defined as the substitution of a hydrogen ...
Phenols

In organic chemistry, phenols, sometimes called phenolics, are a class of chemical compounds consisting of a hydroxyl group (—OH) bonded directly to an aromatic hydrocarbon group. The simplest of the class is phenol, which is also called carbolic acid C6H5OH. Phenolic compounds are classified as simple phenols or polyphenols based on the number of phenol units in the molecule.Synonyms are arenols or aryl alcohols.Phenolic compounds are synthesized industrially; they also are produced by plants and microorganisms, with variation between and within species.Although similar to alcohols, phenols have unique properties and are not classified as alcohols (since the hydroxyl group is not bonded to a saturated carbon atom). They have higher acidities due to the aromatic ring's tight coupling with the oxygen and a relatively loose bond between the oxygen and hydrogen. The acidity of the hydroxyl group in phenols is commonly intermediate between that of aliphatic alcohols and carboxylic acids (their pKa is usually between 10 and 12).Loss of a positive hydrogen ion (H+) from the hydroxyl group of a phenol forms a corresponding negative phenolate ion or phenoxide ion, and the corresponding salts are called phenolates or phenoxides, although the term aryloxides is preferred according to the IUPAC Gold Book. Phenols can have two or more hydroxy groups bonded to the aromatic ring(s) in the same molecule. The simplest examples are the three benzenediols, each having two hydroxy groups on a benzene ring.Organisms that synthesize phenolic compounds do so in response to ecological pressures such as pathogen and insect attack, UV radiation and wounding. As they are present in food consumed in human diets and in plants used in traditional medicine of several cultures, their role in human health and disease is a subject of research.ref name=Klepacka Some phenols are germicidal and are used in formulating disinfectants. Others possess estrogenic or endocrine disrupting activity.