Alcohols
... • Ethers resemble hydrocarbons in their resistance to chemical reaction – they do not react with oxidizing agents such as potassium dichromate – they do not react with reducing agents such as H2 in the presence of a transition metal catalyst – they are not affected by most acids or bases at moderate ...
... • Ethers resemble hydrocarbons in their resistance to chemical reaction – they do not react with oxidizing agents such as potassium dichromate – they do not react with reducing agents such as H2 in the presence of a transition metal catalyst – they are not affected by most acids or bases at moderate ...
Ch12-Alcohols-Grignard
... What remains is the combination of Grignard reagent and carbonyl compound that can be used to prepare the alcohol. ...
... What remains is the combination of Grignard reagent and carbonyl compound that can be used to prepare the alcohol. ...
Chemistry 30 – Organic Chemistry
... • polar but lack of OH bond means no hydrogen bonding, so lower boiling points than alcohols and carboxylic acids • esters with few carbons are polar enough to be soluble in water ...
... • polar but lack of OH bond means no hydrogen bonding, so lower boiling points than alcohols and carboxylic acids • esters with few carbons are polar enough to be soluble in water ...
Alkaloids - ISpatula
... Extract with ether, concentrate, Pseudo-ephedrine will crystallize at first then ephedrine latter. • N.B. CHCl3 is avoided in Ephedrine extraction otherwise undesirable reaction ...
... Extract with ether, concentrate, Pseudo-ephedrine will crystallize at first then ephedrine latter. • N.B. CHCl3 is avoided in Ephedrine extraction otherwise undesirable reaction ...
Alcohols, Phenols and Ethers
... 71) Ether molecules are polar, but do not form hydrogen bonds with other ether molecules because A) ether molecules are so reactive that they never have an opportunity to form hydrogen bonds. B) the molecules are generally too large. C) there are too many hydrogen atoms on the molecules to bond with ...
... 71) Ether molecules are polar, but do not form hydrogen bonds with other ether molecules because A) ether molecules are so reactive that they never have an opportunity to form hydrogen bonds. B) the molecules are generally too large. C) there are too many hydrogen atoms on the molecules to bond with ...
Learning Guide for Chapter 5 - NMR Spectroscopy
... Learning Guide for Chapter 5 - NMR Spectroscopy I. Introduction to NMR spectroscopy - p 1 II. Distinguishing equivalent H's - p 3 III. Chemical shift - p 4 IV. Integration - p 7 V. Spin-spin Splitting - p 9 VI. Practice with NMR spectra VI. Deuterium in NMR - p 12 VII. Carbon-13 NMR- p 13 I. Introdu ...
... Learning Guide for Chapter 5 - NMR Spectroscopy I. Introduction to NMR spectroscopy - p 1 II. Distinguishing equivalent H's - p 3 III. Chemical shift - p 4 IV. Integration - p 7 V. Spin-spin Splitting - p 9 VI. Practice with NMR spectra VI. Deuterium in NMR - p 12 VII. Carbon-13 NMR- p 13 I. Introdu ...
The Impact of Amino Acids on Growth Performance
... The first goal of this study was to evaluate the effect of single amino acids on growth kinetics and major volatile production of two industrial wine yeast strains under conditions resembling wine fermentations. To facilitate these fermentation conditions while also allowing for easy reproducibility ...
... The first goal of this study was to evaluate the effect of single amino acids on growth kinetics and major volatile production of two industrial wine yeast strains under conditions resembling wine fermentations. To facilitate these fermentation conditions while also allowing for easy reproducibility ...
PREPARATION OF ALDEHYDES
... EXAMPLES OF NUCLEOPHILIC ADDITION TO ALDEHYDES & KETONES Addition of HCN (neutral-basic conditions). CN Ө is a very good nucleophile (ionic nucleophile). The use of the actual compound HCN is not experimentally feasible, as it is a lethal gas, bp 26 oC. Addition of the elements of HCN to a C=O grou ...
... EXAMPLES OF NUCLEOPHILIC ADDITION TO ALDEHYDES & KETONES Addition of HCN (neutral-basic conditions). CN Ө is a very good nucleophile (ionic nucleophile). The use of the actual compound HCN is not experimentally feasible, as it is a lethal gas, bp 26 oC. Addition of the elements of HCN to a C=O grou ...
acetyl coenzyme A - Fakultas Farmasi Unand
... Lipids Lipids are naturally occurring substances grouped together on the basis of a common property—they are more soluble in nonpolar solvents than in water. ...
... Lipids Lipids are naturally occurring substances grouped together on the basis of a common property—they are more soluble in nonpolar solvents than in water. ...
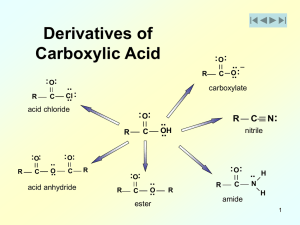
Esterification and Esters
... zinc salts, aluminum halides, and organo−titanates have been used. Cation-exchange resins and zeolites are often employed also. In laboratory preparations, sulfuric acid and hydrochloric acid have classically been used as esterification catalysts. However, formation of alkyl chlorides or dehydration ...
... zinc salts, aluminum halides, and organo−titanates have been used. Cation-exchange resins and zeolites are often employed also. In laboratory preparations, sulfuric acid and hydrochloric acid have classically been used as esterification catalysts. However, formation of alkyl chlorides or dehydration ...
iNTRODUCTiON TO ORGANiC COMPOUNDS
... Organic acids are molecules that contain a carboxyl group (sometimes called a carboxylic acid group). This functional group consists of a carbon which is doubled bonded to an oxygen atom, as was the case with aldehydes and ketones. However, in an acid a hydroxy group (—OH) is also bonded to that sam ...
... Organic acids are molecules that contain a carboxyl group (sometimes called a carboxylic acid group). This functional group consists of a carbon which is doubled bonded to an oxygen atom, as was the case with aldehydes and ketones. However, in an acid a hydroxy group (—OH) is also bonded to that sam ...
Document
... present investigation and data from the literature relative to the composition of comb waxes reveals a general similarity between the latter and propolis waxes, although notable differences are apparent as to the relative proportions of some constituents. This observation suggests that propolis waxe ...
... present investigation and data from the literature relative to the composition of comb waxes reveals a general similarity between the latter and propolis waxes, although notable differences are apparent as to the relative proportions of some constituents. This observation suggests that propolis waxe ...
Science Curriculum Resources Overview* – GRADE 11A
... describe the formation of aldehydes and ketones by oxidation of the corresponding alcohol by acidified ...
... describe the formation of aldehydes and ketones by oxidation of the corresponding alcohol by acidified ...
Chapter 4
... (usually through reactions but then the chemist must know exactly how the reactions proceed – things we will learn as course continues) ...
... (usually through reactions but then the chemist must know exactly how the reactions proceed – things we will learn as course continues) ...
Camp 1 - TypePad
... Ethers are polar compounds in which oxygen bears a partial negative charge and each carbon bonded to it bears a partial positive charge. • However, only weak forces of attraction exist between ether molecules in the pure liquid. • Consequently, boiling points of ethers are close to those of hydrocar ...
... Ethers are polar compounds in which oxygen bears a partial negative charge and each carbon bonded to it bears a partial positive charge. • However, only weak forces of attraction exist between ether molecules in the pure liquid. • Consequently, boiling points of ethers are close to those of hydrocar ...
Slide 1
... Carboxylic acid derivatives commonly undergo nucleophilic substitution at the carbonyl carbon rather than addition. The first step of the mechanism is the same. The C=O p bond breaks and the pair of electrons are stabilized on the electronegative O atom. A tetrahedral alkoxide is temporarily formed. ...
... Carboxylic acid derivatives commonly undergo nucleophilic substitution at the carbonyl carbon rather than addition. The first step of the mechanism is the same. The C=O p bond breaks and the pair of electrons are stabilized on the electronegative O atom. A tetrahedral alkoxide is temporarily formed. ...
Chapter 19
... – Carbon: normally forms four covalent bonds and has no unshared pairs of electrons. C – Hydrogen: forms one covalent bond and no unshared pairs of electrons. H – Nitrogen: normally forms three covalent bonds and has one unshared pair of electrons. ...
... – Carbon: normally forms four covalent bonds and has no unshared pairs of electrons. C – Hydrogen: forms one covalent bond and no unshared pairs of electrons. H – Nitrogen: normally forms three covalent bonds and has one unshared pair of electrons. ...
HOMOLOGATION OF HETEROCYCLES BY A SEQUENTIAL REDUCTIVE OPENING LITHIATION – S
... In sharp contrast to the behaviour of epoxides and oxetanes, tetrahydrofuran (26a) do not undergo reductive opening by means of lithium metal itself and in the presence of arenes as electron carriers at low temperatures. However, it is also possible to carry out this process at low temperature but n ...
... In sharp contrast to the behaviour of epoxides and oxetanes, tetrahydrofuran (26a) do not undergo reductive opening by means of lithium metal itself and in the presence of arenes as electron carriers at low temperatures. However, it is also possible to carry out this process at low temperature but n ...
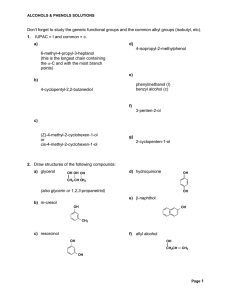
Don`t forget to study the generic functional groups and the common
... allows hydrogenation of H2/Rh an aromatic ring at standard pressure and temperature. Carboxylic acids are not reduced by hydrogen. ...
... allows hydrogenation of H2/Rh an aromatic ring at standard pressure and temperature. Carboxylic acids are not reduced by hydrogen. ...
02B naming alcohols and ethersFeb2013
... Number from the end nearest -OH group. The suffix of an alcohol always ends with –Ol. Indicate the position of each hydroxyl group. If more than one -OH group place a prefix (di, tri, tetra) at the beginning of the suffix ...
... Number from the end nearest -OH group. The suffix of an alcohol always ends with –Ol. Indicate the position of each hydroxyl group. If more than one -OH group place a prefix (di, tri, tetra) at the beginning of the suffix ...
Chapter 23 SG5e
... The Planarity of –NH2 Groups on Heterocyclic Rings A. The angle found in p-nitroaniline means that the amine group is planar and in the same plane as the benzene ring. Why is this the case? 1. The nitro group withdraws the lone pair electron from the amine, primarily via induction, making the N atom ...
... The Planarity of –NH2 Groups on Heterocyclic Rings A. The angle found in p-nitroaniline means that the amine group is planar and in the same plane as the benzene ring. Why is this the case? 1. The nitro group withdraws the lone pair electron from the amine, primarily via induction, making the N atom ...
Organic Chemistry – Who needs it?
... Special reactivity in organic reactions such as electrophilic aromatic substitution and nucleophilic aromatic substitution. within the aromatic family one encounters: A. Benzenoid—Aromatic ring connected to compound—C6H5 B. Phenolic—Phenolic compounds are benzenoid compounds that in addition to th ...
... Special reactivity in organic reactions such as electrophilic aromatic substitution and nucleophilic aromatic substitution. within the aromatic family one encounters: A. Benzenoid—Aromatic ring connected to compound—C6H5 B. Phenolic—Phenolic compounds are benzenoid compounds that in addition to th ...
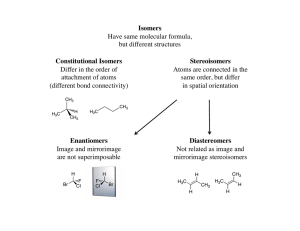
isomeria geometrica
... • Different molecules (enantiomers) must have different names. • Usually only one enantiomer will be biologically active. • Configuration around the chiral carbon is specified with (R) and (S). ...
... • Different molecules (enantiomers) must have different names. • Usually only one enantiomer will be biologically active. • Configuration around the chiral carbon is specified with (R) and (S). ...
History of Organic Chemistry
... Confused, the student tells the naive chemistry-challenged person that the task is impossible. a) Why can=t the student show the structure of C4H10? (2) b) Rephrase the question by changing one word so that the question can be answered, and then answer it. (Keep the flavor of the question - don't ch ...
... Confused, the student tells the naive chemistry-challenged person that the task is impossible. a) Why can=t the student show the structure of C4H10? (2) b) Rephrase the question by changing one word so that the question can be answered, and then answer it. (Keep the flavor of the question - don't ch ...
Phenols

In organic chemistry, phenols, sometimes called phenolics, are a class of chemical compounds consisting of a hydroxyl group (—OH) bonded directly to an aromatic hydrocarbon group. The simplest of the class is phenol, which is also called carbolic acid C6H5OH. Phenolic compounds are classified as simple phenols or polyphenols based on the number of phenol units in the molecule.Synonyms are arenols or aryl alcohols.Phenolic compounds are synthesized industrially; they also are produced by plants and microorganisms, with variation between and within species.Although similar to alcohols, phenols have unique properties and are not classified as alcohols (since the hydroxyl group is not bonded to a saturated carbon atom). They have higher acidities due to the aromatic ring's tight coupling with the oxygen and a relatively loose bond between the oxygen and hydrogen. The acidity of the hydroxyl group in phenols is commonly intermediate between that of aliphatic alcohols and carboxylic acids (their pKa is usually between 10 and 12).Loss of a positive hydrogen ion (H+) from the hydroxyl group of a phenol forms a corresponding negative phenolate ion or phenoxide ion, and the corresponding salts are called phenolates or phenoxides, although the term aryloxides is preferred according to the IUPAC Gold Book. Phenols can have two or more hydroxy groups bonded to the aromatic ring(s) in the same molecule. The simplest examples are the three benzenediols, each having two hydroxy groups on a benzene ring.Organisms that synthesize phenolic compounds do so in response to ecological pressures such as pathogen and insect attack, UV radiation and wounding. As they are present in food consumed in human diets and in plants used in traditional medicine of several cultures, their role in human health and disease is a subject of research.ref name=Klepacka Some phenols are germicidal and are used in formulating disinfectants. Others possess estrogenic or endocrine disrupting activity.