CHAPTER TWENTY-TWO ORGANIC AND BIOLOGICAL MOLECULES
... Cyclic alkanes: general formula CnH2n (if only one ring is present in the compound); all carbons are sp3 hybridized; prefers 109.5Ε bond angles, but rings with three carbons or four carbons or five carbons are forced into bond angles less than 109.5Ε. In cyclopropane, a ring compound made up of thre ...
... Cyclic alkanes: general formula CnH2n (if only one ring is present in the compound); all carbons are sp3 hybridized; prefers 109.5Ε bond angles, but rings with three carbons or four carbons or five carbons are forced into bond angles less than 109.5Ε. In cyclopropane, a ring compound made up of thre ...
Synthesis And Antifungal Activity Of Terbinafine Analogues
... colony-forming units (cfu)/ml was applied to these plates and incubated at 37ºC for 48 h. The MIC was considered to be the lowest concentration of the test substance exhibiting no visible growth of fungi on the plate10. The antifungal activity data of the synthesized compounds is given in Table 2 an ...
... colony-forming units (cfu)/ml was applied to these plates and incubated at 37ºC for 48 h. The MIC was considered to be the lowest concentration of the test substance exhibiting no visible growth of fungi on the plate10. The antifungal activity data of the synthesized compounds is given in Table 2 an ...
Efficient and Convenient Procedure for Protection of Hydroxyl
... when the reaction mixtures were stirred at room temperature for one day, and the starting materials were quantitatively recovered. The reaction conditions are mild enough not to induce any isomerization for conjugated alcohols or damage to moieties such as methoxy, which often undergoes cleavage in ...
... when the reaction mixtures were stirred at room temperature for one day, and the starting materials were quantitatively recovered. The reaction conditions are mild enough not to induce any isomerization for conjugated alcohols or damage to moieties such as methoxy, which often undergoes cleavage in ...
Urea-formaldehyde (UF) resins have been one of the mainstays of
... modification is selected from the group consisting of (1) esterification of said maleated unsaturated fatty acids with ricinoleic acid, (2) amidation of said maleated unsaturated fatty acids using a polyamine supplied in an amount sufficient to cause cross linking between maleated fatty acid molecul ...
... modification is selected from the group consisting of (1) esterification of said maleated unsaturated fatty acids with ricinoleic acid, (2) amidation of said maleated unsaturated fatty acids using a polyamine supplied in an amount sufficient to cause cross linking between maleated fatty acid molecul ...
Unit 2
... The _ _ _ _ _ _ _ fraction is tapped _ _ _ at the bottom of the fractionating column. This material is used in road building and as a protective roof covering. The _ _ _ fraction, used as a _ _ _ _ _ _ _ _ _ or as fuel for _ _ _ _ _ and central heating, condenses at the following level and next come ...
... The _ _ _ _ _ _ _ fraction is tapped _ _ _ at the bottom of the fractionating column. This material is used in road building and as a protective roof covering. The _ _ _ fraction, used as a _ _ _ _ _ _ _ _ _ or as fuel for _ _ _ _ _ and central heating, condenses at the following level and next come ...
REVIEWS Environmental remediation by photocatalysis R. Vinu AND Giridhar Madras
... advanced oxidation processes (AOPs), i.e., processes which employ oxidizing agents like hydrogen peroxide (H2 O2 ), ozone (O3 ) and Fenton’s reagent (H2 O2 + Fe2+ ) for the effective detoxification of the pollutants. Invariably, the above oxidants are used in conjunction with UV radiation and/or photo ...
... advanced oxidation processes (AOPs), i.e., processes which employ oxidizing agents like hydrogen peroxide (H2 O2 ), ozone (O3 ) and Fenton’s reagent (H2 O2 + Fe2+ ) for the effective detoxification of the pollutants. Invariably, the above oxidants are used in conjunction with UV radiation and/or photo ...
14A
... • Ethers are polar compounds in which oxygen bears a partial negative charge and each carbon bonded to it bears a partial positive charge – however, only weak forces of attraction exist between ether molecules in the pure liquid – consequently, boiling points of ethers are close to those of hydrocar ...
... • Ethers are polar compounds in which oxygen bears a partial negative charge and each carbon bonded to it bears a partial positive charge – however, only weak forces of attraction exist between ether molecules in the pure liquid – consequently, boiling points of ethers are close to those of hydrocar ...
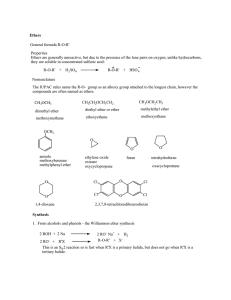
Ethers General formula R-O-R` Properties Ethers are generally
... CH3 CH3 Hg(OCOCH 3)2/CH3 CH2OH H2C C CH 3 H2C C CH 3 Hg OCH2 CH3 O C CH 3 ...
... CH3 CH3 Hg(OCOCH 3)2/CH3 CH2OH H2C C CH 3 H2C C CH 3 Hg OCH2 CH3 O C CH 3 ...
Ethers - Home - KSU Faculty Member websites
... The common names of ethers are derived by naming the alkyl groups bonded to the oxygen then listing them in alphabetical order followed by the word "ether". IUPAC names The ether functional group does not have a characteristic IUPAC nomenclature suffix, so it is necessary to designate the smaller al ...
... The common names of ethers are derived by naming the alkyl groups bonded to the oxygen then listing them in alphabetical order followed by the word "ether". IUPAC names The ether functional group does not have a characteristic IUPAC nomenclature suffix, so it is necessary to designate the smaller al ...
INŻYNIERIA POWIERZCHNI - AGH University of Science and
... • silica SiO2, • mica K2O• 3Al2O3• 6SiO2• H2O ...
... • silica SiO2, • mica K2O• 3Al2O3• 6SiO2• H2O ...
Ether - Clayton State University
... Rules for naming alcohols For single hydroxy (-OH) group • Step 1: Identify longest chain that includes (-OH) group. Drop –e from hydrocarbon name, and replace with ending –ol. • Step 2: Number this parent chain to give lowest number to carbon with attached (-OH) group. • Step 3: Locate position of ...
... Rules for naming alcohols For single hydroxy (-OH) group • Step 1: Identify longest chain that includes (-OH) group. Drop –e from hydrocarbon name, and replace with ending –ol. • Step 2: Number this parent chain to give lowest number to carbon with attached (-OH) group. • Step 3: Locate position of ...
8.1 Alcohols, Phenols, and Ethers 8.2 Naming Alcohols
... Methyl Alcohol (CH3OH, methanol) Methyl alcohol is the simplest (smallest) alcohol and is commonly known as wood alcohol because it was once prepared by heating wood in the absence of air. Ethyl Alcohol (CH3CH2OH, ethanol) Ethyl alcohol is one of the oldest known pure organic compounds. Ethyl alcoho ...
... Methyl Alcohol (CH3OH, methanol) Methyl alcohol is the simplest (smallest) alcohol and is commonly known as wood alcohol because it was once prepared by heating wood in the absence of air. Ethyl Alcohol (CH3CH2OH, ethanol) Ethyl alcohol is one of the oldest known pure organic compounds. Ethyl alcoho ...
Chapter 17 Amines
... to prepare rosaniline and its derivatives, thereby laying the basis for the aniline dye industry. He also discovered a reaction for deriving amines from amides and developed the Hofmann method August Wilhelm von of finding the vapor densities, and from these the molecular weights, ...
... to prepare rosaniline and its derivatives, thereby laying the basis for the aniline dye industry. He also discovered a reaction for deriving amines from amides and developed the Hofmann method August Wilhelm von of finding the vapor densities, and from these the molecular weights, ...
Organic Chemistry II CHM 2211 Sample Exam 2 Answer Key
... When an aldehyde is considered as a group, it is called carbaldehyde group. When a ketone cannot be named as an -one, it is named as an oxo- group. ...
... When an aldehyde is considered as a group, it is called carbaldehyde group. When a ketone cannot be named as an -one, it is named as an oxo- group. ...
Chapter 13 Alcohols, Phenols, and Thiols
... 1-propanol would have the higher boiling point because an alcohol can form hydrogen bonds, but the ether cannot. ...
... 1-propanol would have the higher boiling point because an alcohol can form hydrogen bonds, but the ether cannot. ...
Chapter 13
... electronegative O in the OH groups. Form hydrogen bonds between alcohol molecules. Have higher boiling points than alkanes and ethers of similar mass. Copyright © 2007 by Pearson Education, Inc. Publishing as Benjamin Cummings ...
... electronegative O in the OH groups. Form hydrogen bonds between alcohol molecules. Have higher boiling points than alkanes and ethers of similar mass. Copyright © 2007 by Pearson Education, Inc. Publishing as Benjamin Cummings ...
Alcohols, Phenols, Thiols and Ethers
... electronegative O in the OH groups. Form hydrogen bonds between alcohol molecules. Have higher boiling points than alkanes and ethers of similar mass. ...
... electronegative O in the OH groups. Form hydrogen bonds between alcohol molecules. Have higher boiling points than alkanes and ethers of similar mass. ...
Oxidation
... same). Such as: HBr, HOH, HNO2, HCl, etc 10. Addition of a species Y-Y’ will definitely change the oxidation state of the reaction. Therefore, addition of Y-Y’ (eg. Br-Br) to a double bond is an Oxidation, however, elimination of Y-Y’ from a single bond is reduction. ...
... same). Such as: HBr, HOH, HNO2, HCl, etc 10. Addition of a species Y-Y’ will definitely change the oxidation state of the reaction. Therefore, addition of Y-Y’ (eg. Br-Br) to a double bond is an Oxidation, however, elimination of Y-Y’ from a single bond is reduction. ...
CHM 103 Lecture 23 S07
... • With a common name, the alkyl groups attached to the carbonyl group are named alphabetically followed ...
... • With a common name, the alkyl groups attached to the carbonyl group are named alphabetically followed ...
Ch14_PT MULTIPLE CHOICE. Choose the one alternative that best
... A) isobutyl ether B) 1,2-etherbutane C) butyl ether D) methyl propyl ether E) propyl methyl ether 71) Ether molecules are polar, but do not form hydrogen bonds with other ether molecules because A) ether molecules are so reactive that they never have an opportunity to form hydrogen bonds. B) the mol ...
... A) isobutyl ether B) 1,2-etherbutane C) butyl ether D) methyl propyl ether E) propyl methyl ether 71) Ether molecules are polar, but do not form hydrogen bonds with other ether molecules because A) ether molecules are so reactive that they never have an opportunity to form hydrogen bonds. B) the mol ...
Name_____________________________________ 22 • Organic
... 21. In order to determine the identity of an alkene, a chemist reacts it with water. She then separates the product of this reaction into two equal amounts in container X and container Y. To container X, a strong oxidizing agent is added. The resulting product will react with a base to form a salt a ...
... 21. In order to determine the identity of an alkene, a chemist reacts it with water. She then separates the product of this reaction into two equal amounts in container X and container Y. To container X, a strong oxidizing agent is added. The resulting product will react with a base to form a salt a ...
Phenols

In organic chemistry, phenols, sometimes called phenolics, are a class of chemical compounds consisting of a hydroxyl group (—OH) bonded directly to an aromatic hydrocarbon group. The simplest of the class is phenol, which is also called carbolic acid C6H5OH. Phenolic compounds are classified as simple phenols or polyphenols based on the number of phenol units in the molecule.Synonyms are arenols or aryl alcohols.Phenolic compounds are synthesized industrially; they also are produced by plants and microorganisms, with variation between and within species.Although similar to alcohols, phenols have unique properties and are not classified as alcohols (since the hydroxyl group is not bonded to a saturated carbon atom). They have higher acidities due to the aromatic ring's tight coupling with the oxygen and a relatively loose bond between the oxygen and hydrogen. The acidity of the hydroxyl group in phenols is commonly intermediate between that of aliphatic alcohols and carboxylic acids (their pKa is usually between 10 and 12).Loss of a positive hydrogen ion (H+) from the hydroxyl group of a phenol forms a corresponding negative phenolate ion or phenoxide ion, and the corresponding salts are called phenolates or phenoxides, although the term aryloxides is preferred according to the IUPAC Gold Book. Phenols can have two or more hydroxy groups bonded to the aromatic ring(s) in the same molecule. The simplest examples are the three benzenediols, each having two hydroxy groups on a benzene ring.Organisms that synthesize phenolic compounds do so in response to ecological pressures such as pathogen and insect attack, UV radiation and wounding. As they are present in food consumed in human diets and in plants used in traditional medicine of several cultures, their role in human health and disease is a subject of research.ref name=Klepacka Some phenols are germicidal and are used in formulating disinfectants. Others possess estrogenic or endocrine disrupting activity.