Chapter 3
... experiments measured the charge-to-mass ratio of an electron. • Robert A. Millikan’s oil drop experiment measured the charge of an electron. • With this information, scientists were able to determine the mass of an electron. ...
... experiments measured the charge-to-mass ratio of an electron. • Robert A. Millikan’s oil drop experiment measured the charge of an electron. • With this information, scientists were able to determine the mass of an electron. ...
chem pre ap atom and nuclear practice test
... c. of each element are identical in size, mass, and other properties. d. of different elements cannot combine. 2. Which of the following statements is true? a. Atoms of the same element may have different masses. b. Atoms may be divided in ordinary chemical reactions. c. Atoms can never combine with ...
... c. of each element are identical in size, mass, and other properties. d. of different elements cannot combine. 2. Which of the following statements is true? a. Atoms of the same element may have different masses. b. Atoms may be divided in ordinary chemical reactions. c. Atoms can never combine with ...
What are atoms?
... • An atom is the smallest particle into which an element can be divided and still be the same substance. • In 1808, John Dalton published an atomic theory that said all atoms of a particular element are identical. • Dalton also said that atoms of an element differ from atoms of other elements. ...
... • An atom is the smallest particle into which an element can be divided and still be the same substance. • In 1808, John Dalton published an atomic theory that said all atoms of a particular element are identical. • Dalton also said that atoms of an element differ from atoms of other elements. ...
Unit 1 Section 4 - Atomic Structure PPT
... Isotopes - atoms with the same number of protons but different numbers of neutrons. Show almost identical chemical properties; chemistry of atom is due to its electrons. In nature most elements contain mixtures of isotopes. ...
... Isotopes - atoms with the same number of protons but different numbers of neutrons. Show almost identical chemical properties; chemistry of atom is due to its electrons. In nature most elements contain mixtures of isotopes. ...
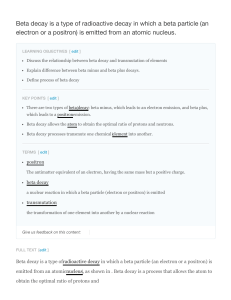
Beta decay is a type of radioactive decay in which a beta
... Beta decay does not change the number of nucleons, A, in the nucleus; it changes only its charge, Z. Therefore the set of allnuclides with the same A can be introduced; these isobaric nuclides may turn into each other via beta decay. A beta-stable nucleus may undergo other kinds of radioactive decay ...
... Beta decay does not change the number of nucleons, A, in the nucleus; it changes only its charge, Z. Therefore the set of allnuclides with the same A can be introduced; these isobaric nuclides may turn into each other via beta decay. A beta-stable nucleus may undergo other kinds of radioactive decay ...
catch some rays: alpha, beta, gamma (modified for adeed)
... from. Explain that in each example, energy was released, but that no matter was changed. These are chemical reactions, not nuclear. The difference is, in a nuclear reaction matter is actually changed to energy!! If you measure the mass before and after a chemical reaction, the masses would be the sa ...
... from. Explain that in each example, energy was released, but that no matter was changed. These are chemical reactions, not nuclear. The difference is, in a nuclear reaction matter is actually changed to energy!! If you measure the mass before and after a chemical reaction, the masses would be the sa ...
KNIGHT Physics for Scientists and Engineers
... Chemical behavior is determined by the orbiting electrons. All isotopes of one element have the same number of orbiting electrons (if the atoms are electrically neutral) and thus have the same chemical properties, but different isotopes of the same element can have quite different nuclear properties ...
... Chemical behavior is determined by the orbiting electrons. All isotopes of one element have the same number of orbiting electrons (if the atoms are electrically neutral) and thus have the same chemical properties, but different isotopes of the same element can have quite different nuclear properties ...
Sample pages 2 PDF
... orbiting electrons which occupy the vast bulk of the atoms volume. The simplest atom—hydrogen—consisted of a proton and a single orbital electron. Later, in 1920, the radii of a few heavy nuclei were measured by Chadwick and were found to be in the order of 10−14 m, much smaller than the order of 10 ...
... orbiting electrons which occupy the vast bulk of the atoms volume. The simplest atom—hydrogen—consisted of a proton and a single orbital electron. Later, in 1920, the radii of a few heavy nuclei were measured by Chadwick and were found to be in the order of 10−14 m, much smaller than the order of 10 ...
Atomic Structure and the Periodic Table
... All matter is made of very tiny particles. These particles keep the same characteristics or properties that the matter has. These particles are called atoms. All atoms are about the same size and they are VERY tiny. An atom is more than one million times smaller than the thickness of a single hair ...
... All matter is made of very tiny particles. These particles keep the same characteristics or properties that the matter has. These particles are called atoms. All atoms are about the same size and they are VERY tiny. An atom is more than one million times smaller than the thickness of a single hair ...
Radioactive Decays – transmutations of nuclides
... are expressed in decay constant and half life. Variations of radioactivity in mixtures of radioactive nuclides and consecutive decays are often considered, and decay kinetics serves science and technology in many applications. In radioactive decay processes, some of the things are conserved, meaning ...
... are expressed in decay constant and half life. Variations of radioactivity in mixtures of radioactive nuclides and consecutive decays are often considered, and decay kinetics serves science and technology in many applications. In radioactive decay processes, some of the things are conserved, meaning ...
The Atoms Family
... explore the world of electrons and quarks that are at least 9,000 times smaller than a nucleus. • We can not see anything smaller than an atom with our eyes, even with the electron microscope. Physicists study much smaller things without seeing them directly. ...
... explore the world of electrons and quarks that are at least 9,000 times smaller than a nucleus. • We can not see anything smaller than an atom with our eyes, even with the electron microscope. Physicists study much smaller things without seeing them directly. ...
Atoms and Molecules
... Note: Typically, positively charged particles would repel each other, but they are held together in the nucleus with a force called the strong atomic force. This is the strongest force in the universe. The other part of the nucleus is the neutron. Neutrons are about the same size as protons. The wor ...
... Note: Typically, positively charged particles would repel each other, but they are held together in the nucleus with a force called the strong atomic force. This is the strongest force in the universe. The other part of the nucleus is the neutron. Neutrons are about the same size as protons. The wor ...
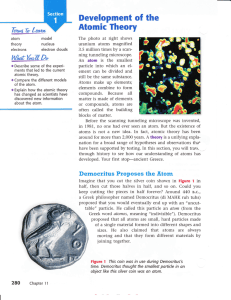
Development of the Atomic Theory
... learned how the atomic theory developed through centuries of observation and experimentation. Now it's time to learn about the atom itself. In this section, you'll learn about the particles inside the atom, and you'll learn about the forces that act on those particles. But first you'll find out just ...
... learned how the atomic theory developed through centuries of observation and experimentation. Now it's time to learn about the atom itself. In this section, you'll learn about the particles inside the atom, and you'll learn about the forces that act on those particles. But first you'll find out just ...
Chapter 14 Inside an Atom
... of rocks, they cannot use carbon dating. • Instead, geologists examine the decay of uranium. • Uranium-238 decays to lead-206 with a halflife of 4.5 billion years. • By comparing the amount of uranium to lead, the scientist can determine the age of a rock. ...
... of rocks, they cannot use carbon dating. • Instead, geologists examine the decay of uranium. • Uranium-238 decays to lead-206 with a halflife of 4.5 billion years. • By comparing the amount of uranium to lead, the scientist can determine the age of a rock. ...
Chromatographic Enrichment of Lithium Isotopes by Hydrous
... role in nuclear science and industry. The heavy isotope of lithium, 7Li could be used as a pH control agent (7LiOH) of the coolant in nuclear fission reactors. And the lithium compounds rich in 6Li will be required for the tritium breeder blanket in deuterium-tritium fusion power reactors in the fut ...
... role in nuclear science and industry. The heavy isotope of lithium, 7Li could be used as a pH control agent (7LiOH) of the coolant in nuclear fission reactors. And the lithium compounds rich in 6Li will be required for the tritium breeder blanket in deuterium-tritium fusion power reactors in the fut ...
The Atom - cloudfront.net
... A similar force acts when two neutrons are very close together, or when a neutron and a proton are very close together. Together, these short-range proton-proton, neutron-neutron, and proton-neutron forces are called nuderr forces. These forces allow atoms with up to 83 positivelycharged protons in ...
... A similar force acts when two neutrons are very close together, or when a neutron and a proton are very close together. Together, these short-range proton-proton, neutron-neutron, and proton-neutron forces are called nuderr forces. These forces allow atoms with up to 83 positivelycharged protons in ...
Introduction to Atomic Structure - New Jersey Center for Teaching
... than those in hydrogen, it was assumed that oxygen had 8 protons compared to hydrogen's 1. The number of protons an atom has is it's atomic number (Z) and it is different for each element on the periodic table. ...
... than those in hydrogen, it was assumed that oxygen had 8 protons compared to hydrogen's 1. The number of protons an atom has is it's atomic number (Z) and it is different for each element on the periodic table. ...
Isotope

Isotopes are variants of a particular chemical element which differ in neutron number, although all isotopes of a given element have the same number of protons in each atom. The term isotope is formed from the Greek roots isos (ἴσος ""equal"") and topos (τόπος ""place""), meaning ""the same place""; thus, the meaning behind the name it is that different isotopes of a single element occupy the same position on the periodic table. The number of protons within the atom's nucleus is called atomic number and is equal to the number of electrons in the neutral (non-ionized) atom. Each atomic number identifies a specific element, but not the isotope; an atom of a given element may have a wide range in its number of neutrons. The number of nucleons (both protons and neutrons) in the nucleus is the atom's mass number, and each isotope of a given element has a different mass number.For example, carbon-12, carbon-13 and carbon-14 are three isotopes of the element carbon with mass numbers 12, 13 and 14 respectively. The atomic number of carbon is 6, which means that every carbon atom has 6 protons, so that the neutron numbers of these isotopes are 6, 7 and 8 respectively.