Scheme of work
... that electrons orbit the nucleus at specific distances. Later experiments led to the idea that the positive charge of any nucleus could be subdivided into a whole number of smaller particles, each particle having the same amount of positive charge. The name ‘proton’ was given to these particles. ...
... that electrons orbit the nucleus at specific distances. Later experiments led to the idea that the positive charge of any nucleus could be subdivided into a whole number of smaller particles, each particle having the same amount of positive charge. The name ‘proton’ was given to these particles. ...
Physics HW Chapters 383940 (Due May 23, Test May 28)
... 10. The number of electrons ejected in the photoelectric effect depends on the light beam's a. amplitude. b. speed. c. frequency. d. intensity. e. wavelength. 11. A photosensitive surface is illuminated with both blue and violet light. The light that will cause more electrons to be ejected is the a. ...
... 10. The number of electrons ejected in the photoelectric effect depends on the light beam's a. amplitude. b. speed. c. frequency. d. intensity. e. wavelength. 11. A photosensitive surface is illuminated with both blue and violet light. The light that will cause more electrons to be ejected is the a. ...
Counting Atoms
... • Atoms of different elements have different numbers of protons. • Atoms of the same element all have the same number of protons. • The atomic number (Z) of an element is the number of protons of each atom of that element. ...
... • Atoms of different elements have different numbers of protons. • Atoms of the same element all have the same number of protons. • The atomic number (Z) of an element is the number of protons of each atom of that element. ...
Atomic Structure
... supported the hypothesis that the cathode ray consisted of a stream of particles. 1. When an object was placed between the cathode and the opposite end of the tube, it cast a shadow on the glass. 2. A cathode ray tube was constructed with a small metal rail between the two electrodes. Attached to th ...
... supported the hypothesis that the cathode ray consisted of a stream of particles. 1. When an object was placed between the cathode and the opposite end of the tube, it cast a shadow on the glass. 2. A cathode ray tube was constructed with a small metal rail between the two electrodes. Attached to th ...
Atomic Theory PPT
... 1. Atoms are not indivisible. They are made up of protons, electrons, and neutrons. 2. Atoms of the same element can, and do have different masses. These atoms are called isotopes. Isotopes have the same number of protons but a different number of neutrons. 3. Atoms of different elements are differe ...
... 1. Atoms are not indivisible. They are made up of protons, electrons, and neutrons. 2. Atoms of the same element can, and do have different masses. These atoms are called isotopes. Isotopes have the same number of protons but a different number of neutrons. 3. Atoms of different elements are differe ...
Once scientists concluded that all matter contains negatively
... Figure: The Scientific Method: As depicted in this flowchart, the scientific method consists of making observations, formulating hypotheses, and designing experiments. A scientist may enter the cycle at any point. Observations can be qualitative or quantitative. Qualitative observations describe pro ...
... Figure: The Scientific Method: As depicted in this flowchart, the scientific method consists of making observations, formulating hypotheses, and designing experiments. A scientist may enter the cycle at any point. Observations can be qualitative or quantitative. Qualitative observations describe pro ...
Chapter42015.1 STUDENT
... Target 2 - Identify the atomic number and the atomic mass of all elements and explain what they mean. A. All of the elements are listed on the ___________________ _______ of Elements. B. Elements are different kinds of atoms with a name, symbol, and unique properties. C. The Periodic Table lists the ...
... Target 2 - Identify the atomic number and the atomic mass of all elements and explain what they mean. A. All of the elements are listed on the ___________________ _______ of Elements. B. Elements are different kinds of atoms with a name, symbol, and unique properties. C. The Periodic Table lists the ...
Dalton`s Atomic Theory
... 2. All atoms of the same element are exactly alike; in particular, they all have the same mass. 3. Atoms of different elements are different; in particular, they have different masses. 4. Compounds are formed by the joining of atoms of two or more elements. They are joined in a definite whole-number ...
... 2. All atoms of the same element are exactly alike; in particular, they all have the same mass. 3. Atoms of different elements are different; in particular, they have different masses. 4. Compounds are formed by the joining of atoms of two or more elements. They are joined in a definite whole-number ...
irm_ch11
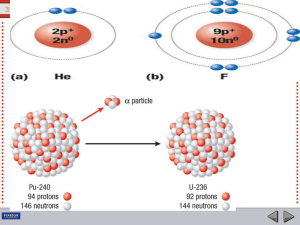
... 11.61 a. Balance the mass numbers: products [2(3)] = reactants [2(1) + x] = 6; x = 4 Balance the atomic numbers: products (2 + x) = reactants [2(2)] = 4; x = 2 The particle having a mass number of 4 and an atomic number of 2 is: ...
... 11.61 a. Balance the mass numbers: products [2(3)] = reactants [2(1) + x] = 6; x = 4 Balance the atomic numbers: products (2 + x) = reactants [2(2)] = 4; x = 2 The particle having a mass number of 4 and an atomic number of 2 is: ...
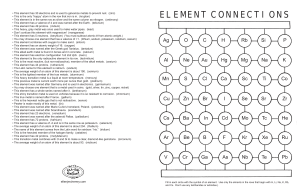
element connections
... • The name of this element comes from the Latin word for rainbow: “iris.” (iridium) • This is the heaviest member of the halogen family. (astatine) • This element has 42 protons. (molybdenum) • This transition metal combines with O and Si to make a clear, diamond-like gemstone. (zirconium) • The ave ...
... • The name of this element comes from the Latin word for rainbow: “iris.” (iridium) • This is the heaviest member of the halogen family. (astatine) • This element has 42 protons. (molybdenum) • This transition metal combines with O and Si to make a clear, diamond-like gemstone. (zirconium) • The ave ...
Atoms - RCSD
... uniformly distrubuted throughout the atoms of the gold foil, like the plum pudding ...
... uniformly distrubuted throughout the atoms of the gold foil, like the plum pudding ...
View - Rutgers Physics
... with C4 = 23.6 MeV. It is called semiempirical because each term has a theoretical explanation but the constants are not calculated theoretically but are instead determined by a fit to the actual nuclear masses. Two other things the independent particle model gives us, if we consider it a bit more c ...
... with C4 = 23.6 MeV. It is called semiempirical because each term has a theoretical explanation but the constants are not calculated theoretically but are instead determined by a fit to the actual nuclear masses. Two other things the independent particle model gives us, if we consider it a bit more c ...
set2
... • Atoms are held together by chemical bonds, which are forces between them – Ionic bonds arise from attraction between positive ions (cations) and negative ones (anions): NaCl, NH4NO3, ...
... • Atoms are held together by chemical bonds, which are forces between them – Ionic bonds arise from attraction between positive ions (cations) and negative ones (anions): NaCl, NH4NO3, ...
4 Structure of The Atom
... to overcome the objections raised against Rutherford's atomic model. (iii) Chlorine occurs in nature in two isotopic forms, with atomic masses 35 u and 37 u in the ratio of 3 : 1. Calculate the average atomic mass of chlorine atom. [2011 (T-II)] 5. (a) Write the name of the sub-atomic partic ...
... to overcome the objections raised against Rutherford's atomic model. (iii) Chlorine occurs in nature in two isotopic forms, with atomic masses 35 u and 37 u in the ratio of 3 : 1. Calculate the average atomic mass of chlorine atom. [2011 (T-II)] 5. (a) Write the name of the sub-atomic partic ...
Isotope

Isotopes are variants of a particular chemical element which differ in neutron number, although all isotopes of a given element have the same number of protons in each atom. The term isotope is formed from the Greek roots isos (ἴσος ""equal"") and topos (τόπος ""place""), meaning ""the same place""; thus, the meaning behind the name it is that different isotopes of a single element occupy the same position on the periodic table. The number of protons within the atom's nucleus is called atomic number and is equal to the number of electrons in the neutral (non-ionized) atom. Each atomic number identifies a specific element, but not the isotope; an atom of a given element may have a wide range in its number of neutrons. The number of nucleons (both protons and neutrons) in the nucleus is the atom's mass number, and each isotope of a given element has a different mass number.For example, carbon-12, carbon-13 and carbon-14 are three isotopes of the element carbon with mass numbers 12, 13 and 14 respectively. The atomic number of carbon is 6, which means that every carbon atom has 6 protons, so that the neutron numbers of these isotopes are 6, 7 and 8 respectively.