19a - The BOD
... What is the dependence? Is the dependence linear or super-linear or sub-linear? ...
... What is the dependence? Is the dependence linear or super-linear or sub-linear? ...
Types of Chemical Reactions Name_________________________
... reaction into a word equation and then a balanced chemical equation. Note: Access to some of the websites may not be possible dependent upon your computer system and or the network connection. You are still required to work through each main type of reaction providing balanced chemical equations bas ...
... reaction into a word equation and then a balanced chemical equation. Note: Access to some of the websites may not be possible dependent upon your computer system and or the network connection. You are still required to work through each main type of reaction providing balanced chemical equations bas ...
CHEMISTRY
... Consider three of the alkali metals; lithium, sodium and potassium. What are some physical properties that these elements have in common? ____________________________________________________________________ ____________________________________________________________________ ________________________ ...
... Consider three of the alkali metals; lithium, sodium and potassium. What are some physical properties that these elements have in common? ____________________________________________________________________ ____________________________________________________________________ ________________________ ...
Predicting Reactions • AP Chemistry CLASSIFYING REACTIONS
... Oxides of nonmetals (acid anhydrides) and oxides of metals (base anhydrides) are also examples: CO2 + H2O H2CO3 CaO + H2O Ca(OH)2 When you get one of each, the result is a salt: CaO + CO2 CaCO3 Aluminum ions (and Fe3+ ions) make a solution acidic because they are Lewis acids and draw electrons a ...
... Oxides of nonmetals (acid anhydrides) and oxides of metals (base anhydrides) are also examples: CO2 + H2O H2CO3 CaO + H2O Ca(OH)2 When you get one of each, the result is a salt: CaO + CO2 CaCO3 Aluminum ions (and Fe3+ ions) make a solution acidic because they are Lewis acids and draw electrons a ...
2015 AP Chemistry Summer Assignment
... match on a matchbox. If you were to react 52.9 g of potassium chlorate (KClO3) with excess red phosphorus, what mass of tetraphosphorus decaoxide (P4O10) could be produced? KClO3(s) + P4(s) → P4O10(s) + KCl(s) (unbalanced) 44. Elixirs such as Alka-Seltzer use the reaction of sodium bicarbonate with ...
... match on a matchbox. If you were to react 52.9 g of potassium chlorate (KClO3) with excess red phosphorus, what mass of tetraphosphorus decaoxide (P4O10) could be produced? KClO3(s) + P4(s) → P4O10(s) + KCl(s) (unbalanced) 44. Elixirs such as Alka-Seltzer use the reaction of sodium bicarbonate with ...
Grade 9 Academic Science
... 21. If you have an alkaline-earth metal “A” combine with a halogen “B”, what will be the resulting chemical formula? Use your knowledge of the # of valence electrons of alkaline-earth metals and halogens to answer this question. 22. Give 3 examples of how elements & compounds can be extremely useful ...
... 21. If you have an alkaline-earth metal “A” combine with a halogen “B”, what will be the resulting chemical formula? Use your knowledge of the # of valence electrons of alkaline-earth metals and halogens to answer this question. 22. Give 3 examples of how elements & compounds can be extremely useful ...
13. Condensed azines. Quinoline. Isoquinoline. Acridine. Diazines
... composed of a benzene ring fused to a pyridine ring. In a broader sense, the term isoquinoline is used to make reference to isoquinoline derivatives. 1-Benzylisoquinoline is the structural backbone in naturally occurring alkaloids including papaverine and morphine. The isoquinoline ring in these nat ...
... composed of a benzene ring fused to a pyridine ring. In a broader sense, the term isoquinoline is used to make reference to isoquinoline derivatives. 1-Benzylisoquinoline is the structural backbone in naturally occurring alkaloids including papaverine and morphine. The isoquinoline ring in these nat ...
Chemical Reactions
... reaction (doesn’t appear as a reactant or a product) Catalysts lower the activation energy required for a reaction to occur. Thus a catalyst creates a different pathway from reactants to products – one that requires less ...
... reaction (doesn’t appear as a reactant or a product) Catalysts lower the activation energy required for a reaction to occur. Thus a catalyst creates a different pathway from reactants to products – one that requires less ...
PCSD General Chemistry Pacing Guide
... Relate electron configuration to the arrangement of the Periodic Table Follow Hund's Rule to describe the orbitals in an atom using orbital diagrams Relate quantum numbers to the position of the electron in the atom Draw electron dot diagrams to describe the valence electrons in an atom ...
... Relate electron configuration to the arrangement of the Periodic Table Follow Hund's Rule to describe the orbitals in an atom using orbital diagrams Relate quantum numbers to the position of the electron in the atom Draw electron dot diagrams to describe the valence electrons in an atom ...
Balancing Chemical Equations
... 3. You can only add coefficients a. coefficients are numbers that go in front of the chemical formula b. 3 FeCl2 + 2 Al the 3 and 2 are coefficients.. ...
... 3. You can only add coefficients a. coefficients are numbers that go in front of the chemical formula b. 3 FeCl2 + 2 Al the 3 and 2 are coefficients.. ...
Multivalent Ionic Compounds
... BCLN SCIENCE 10 - Rev. Mar /2016 12. Draw Lewis Diagrams for the atoms and ions of the first 18 elements in the Periodic Table. If the element does not form an ion simply state “no ion”. The first two are done for you as an example. Hint: Ions are usually formed by losing all electrons in the Lewis ...
... BCLN SCIENCE 10 - Rev. Mar /2016 12. Draw Lewis Diagrams for the atoms and ions of the first 18 elements in the Periodic Table. If the element does not form an ion simply state “no ion”. The first two are done for you as an example. Hint: Ions are usually formed by losing all electrons in the Lewis ...
Chapters 9 and 10
... structure. Indicate whether the molecule is polar or nonpolar, and justify your answer. 13. 2002 #6c. Use the principles of atomic structure and/or chemical bonding to explain each of the following. In each part, your answer must include references to both substances. a. The carbon – to – carbon bon ...
... structure. Indicate whether the molecule is polar or nonpolar, and justify your answer. 13. 2002 #6c. Use the principles of atomic structure and/or chemical bonding to explain each of the following. In each part, your answer must include references to both substances. a. The carbon – to – carbon bon ...
CBSE/12th Class/2010/CHEMISTRY
... (ii)The E0 value for the Mn3/Mn2+ couple is much more positive than that for Cr3/Cr2+ couple or Fe3+/Fe2+ couple.Because Mn3+ has the outer electronic configuration of 3d4 and Mn2+ has the outer electronic configuration of 3d5. Thus, the conversion of Mn3+ to Mn2+ will be a favourable reaction since ...
... (ii)The E0 value for the Mn3/Mn2+ couple is much more positive than that for Cr3/Cr2+ couple or Fe3+/Fe2+ couple.Because Mn3+ has the outer electronic configuration of 3d4 and Mn2+ has the outer electronic configuration of 3d5. Thus, the conversion of Mn3+ to Mn2+ will be a favourable reaction since ...
AP Chemistry Summer Assignment
... 15. Why do we call Ba(NO3)2 barium nitrate, but we call Fe(NO3)2 iron(II) nitrate? 16. Calculate the mass of O2 produced if 3.450 g potassium chlorate is completely decomposed by heating in presence of a catalyst ( Manganese dioxide). 17. Write the formula of the following compounds? a. Calcium sulf ...
... 15. Why do we call Ba(NO3)2 barium nitrate, but we call Fe(NO3)2 iron(II) nitrate? 16. Calculate the mass of O2 produced if 3.450 g potassium chlorate is completely decomposed by heating in presence of a catalyst ( Manganese dioxide). 17. Write the formula of the following compounds? a. Calcium sulf ...
Chemical Reactions and Stoichiometry
... Indicators of a Chemical Reaction – evidence of a chemical reaction a. Evolution of heat and light (simultaneously) b. Production of a gas (bubbles, odor change) c. Formation of a precipitate (solid, cloudy) d. Color change (not introduced by an outside source such as dye or ink) Characteristics of ...
... Indicators of a Chemical Reaction – evidence of a chemical reaction a. Evolution of heat and light (simultaneously) b. Production of a gas (bubbles, odor change) c. Formation of a precipitate (solid, cloudy) d. Color change (not introduced by an outside source such as dye or ink) Characteristics of ...
Chapter 3 Secondary Organic Aerosol Formation by Heterogeneous
... amount of SOA formed in a number of systems exceeds that based purely on G/P partitioning of low vapor pressure oxidation product [6-10]. Evidence also indicates that relatively volatile oxidation products, especially aldehydes and ketones, are being absorbed into the aerosol phase where they underg ...
... amount of SOA formed in a number of systems exceeds that based purely on G/P partitioning of low vapor pressure oxidation product [6-10]. Evidence also indicates that relatively volatile oxidation products, especially aldehydes and ketones, are being absorbed into the aerosol phase where they underg ...
ch14 lecture 7e
... Compounds of 3A elements have more covalent character than similar 2A compounds. Aluminum has the physical properties of a metal, but its halides exist as covalent dimers. ...
... Compounds of 3A elements have more covalent character than similar 2A compounds. Aluminum has the physical properties of a metal, but its halides exist as covalent dimers. ...
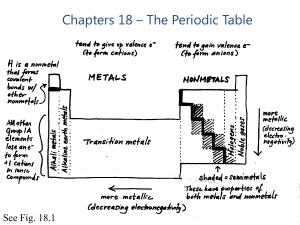
Chapters 18 – The Periodic Table
... element behaves like a true metal Common behavior: reaction with metal to become 2- ion in ionic compound; for most metals, most common minerals are oxides or sulfides Covalent bonds with other NMs; series of covalent hydrides (H2X) All but O have d orbitals available, so more than an octet is ...
... element behaves like a true metal Common behavior: reaction with metal to become 2- ion in ionic compound; for most metals, most common minerals are oxides or sulfides Covalent bonds with other NMs; series of covalent hydrides (H2X) All but O have d orbitals available, so more than an octet is ...
hit and lead generation: beyond high-throughput screening
... and X-ray crystallography, as well as the recognition information that can be derived from endogenous ligands or non-natural small-molecule surrogates retrieved from literature and patents. At the other extreme are the technologies that do not require any prior information on target or ligand, and w ...
... and X-ray crystallography, as well as the recognition information that can be derived from endogenous ligands or non-natural small-molecule surrogates retrieved from literature and patents. At the other extreme are the technologies that do not require any prior information on target or ligand, and w ...
Chapter 6A Chemical Reactions CHAPTER OUTLINE
... the transfer of hydrogen atoms produces energy in the cells. q For example, cellular respiration is an oxidationreduction process that transfers energy from the bonds in glucose to form ATP. C6H12O6 + 6 O2 ...
... the transfer of hydrogen atoms produces energy in the cells. q For example, cellular respiration is an oxidationreduction process that transfers energy from the bonds in glucose to form ATP. C6H12O6 + 6 O2 ...
Elements (NonMetals)
... Gas at room Temp B.P. –253°C (20K) and M.P.-259°C (14K) Insoluble in water: 2mL gas/ 1L of water Found in H2O, organic and biological molecules Most common element in universe H2 (H-H) isoelectronic with He H has a small radius Unique properties of both group 1 and 17 Bond energy 431kJ/mol – very st ...
... Gas at room Temp B.P. –253°C (20K) and M.P.-259°C (14K) Insoluble in water: 2mL gas/ 1L of water Found in H2O, organic and biological molecules Most common element in universe H2 (H-H) isoelectronic with He H has a small radius Unique properties of both group 1 and 17 Bond energy 431kJ/mol – very st ...
AP Chemistry Summer Assignment 2016
... (aq) + BaCl2 (aq) 2 AgCl(s) + Ba (NO3) 2(aq) The silver chloride is the precipitate in this reaction. Solubility Rules are available in general chemistry textbooks in the library. 10) This is a combustion reaction. The products of complete combustion reactions involving organic compounds as fuels ...
... (aq) + BaCl2 (aq) 2 AgCl(s) + Ba (NO3) 2(aq) The silver chloride is the precipitate in this reaction. Solubility Rules are available in general chemistry textbooks in the library. 10) This is a combustion reaction. The products of complete combustion reactions involving organic compounds as fuels ...
Chapter 5—Chemical Reactions
... 5.6—Replacement Reactions • Single-replacement reactions—one element replaces another in a compound. They are always redox reactions because an element turns into an ion and an ion turns into an element • The general form of the equation for a single replacement reaction (also known as substitution ...
... 5.6—Replacement Reactions • Single-replacement reactions—one element replaces another in a compound. They are always redox reactions because an element turns into an ion and an ion turns into an element • The general form of the equation for a single replacement reaction (also known as substitution ...
Chapter 3 Molecules, Compounds, and Chemical Equations
... • shorthand way of describing a reaction • provides information about the reaction formulas of reactants and products states of reactants and products relative numbers of reactant and product molecules that are required can be used to determine weights of reactants used and products that can ...
... • shorthand way of describing a reaction • provides information about the reaction formulas of reactants and products states of reactants and products relative numbers of reactant and product molecules that are required can be used to determine weights of reactants used and products that can ...
Organic chemistry

Organic chemistry is a chemistry subdiscipline involving the scientific study of the structure, properties, and reactions of organic compounds and organic materials, i.e., matter in its various forms that contain carbon atoms. Study of structure includes many physical and chemical methods to determine the chemical composition and the chemical constitution of organic compounds and materials. Study of properties includes both physical properties and chemical properties, and uses similar methods as well as methods to evaluate chemical reactivity, with the aim to understand the behavior of the organic matter in its pure form (when possible), but also in solutions, mixtures, and fabricated forms. The study of organic reactions includes probing their scope through use in preparation of target compounds (e.g., natural products, drugs, polymers, etc.) by chemical synthesis, as well as the focused study of the reactivities of individual organic molecules, both in the laboratory and via theoretical (in silico) study.The range of chemicals studied in organic chemistry include hydrocarbons (compounds containing only carbon and hydrogen), as well as myriad compositions based always on carbon, but also containing other elements, especially oxygen, nitrogen, sulfur, phosphorus (these, included in many organic chemicals in biology) and the radiostable elements of the halogens.In the modern era, the range extends further into the periodic table, with main group elements, including:Group 1 and 2 organometallic compounds, i.e., involving alkali (e.g., lithium, sodium, and potassium) or alkaline earth metals (e.g., magnesium)Metalloids (e.g., boron and silicon) or other metals (e.g., aluminium and tin)In addition, much modern research focuses on organic chemistry involving further organometallics, including the lanthanides, but especially the transition metals; (e.g., zinc, copper, palladium, nickel, cobalt, titanium and chromium)Finally, organic compounds form the basis of all earthly life and constitute a significant part of human endeavors in chemistry. The bonding patterns open to carbon, with its valence of four—formal single, double, and triple bonds, as well as various structures with delocalized electrons—make the array of organic compounds structurally diverse, and their range of applications enormous. They either form the basis of, or are important constituents of, many commercial products including pharmaceuticals; petrochemicals and products made from them (including lubricants, solvents, etc.); plastics; fuels and explosives; etc. As indicated, the study of organic chemistry overlaps with organometallic chemistry and biochemistry, but also with medicinal chemistry, polymer chemistry, as well as many aspects of materials science.