Week 4
... What drives protons out of matrix into intermembrane space? • Energy released as electron moves down the electron transport chain. • Quantify the energy DGo = - n (0.023) (DE’o); n = number of electrons used; DE’o = difference in redox potential between oxidizing agent and reducing agent (see bel ...
... What drives protons out of matrix into intermembrane space? • Energy released as electron moves down the electron transport chain. • Quantify the energy DGo = - n (0.023) (DE’o); n = number of electrons used; DE’o = difference in redox potential between oxidizing agent and reducing agent (see bel ...
Week 4
... What drives protons out of matrix into intermembrane space? • Energy released as electron moves down the electron transport chain. • Quantify the energy DGo = - n (0.023) (DE’o); n = number of electrons used; DE’o = difference in redox potential between oxidizing agent and reducing agent (see bel ...
... What drives protons out of matrix into intermembrane space? • Energy released as electron moves down the electron transport chain. • Quantify the energy DGo = - n (0.023) (DE’o); n = number of electrons used; DE’o = difference in redox potential between oxidizing agent and reducing agent (see bel ...
Chemolithotrophs
... inorganic electron donor for energy and electrons. • Chemolithotrophs: reduced inorganic electron donor for energy and electrons. • Phototrophs: use light energy and an electron donor molecule (H2O, H2S, organic). • Both may be autotrophs: fix CO2 into organic carbon via the Calvin Cycle. ...
... inorganic electron donor for energy and electrons. • Chemolithotrophs: reduced inorganic electron donor for energy and electrons. • Phototrophs: use light energy and an electron donor molecule (H2O, H2S, organic). • Both may be autotrophs: fix CO2 into organic carbon via the Calvin Cycle. ...
Krebs Cycle - Deranged Physiology
... operated by NADH and FADH. Electrons move down the redox gradient and the resulting energy is used to pump H+ ions out of the inner mitochondrial membrane. Purpose is to build a negative charge inside membrane and thus attract H+ ions back into the mitochondrion. (the membrane is impervious to H+ ex ...
... operated by NADH and FADH. Electrons move down the redox gradient and the resulting energy is used to pump H+ ions out of the inner mitochondrial membrane. Purpose is to build a negative charge inside membrane and thus attract H+ ions back into the mitochondrion. (the membrane is impervious to H+ ex ...
File
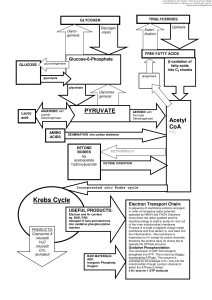
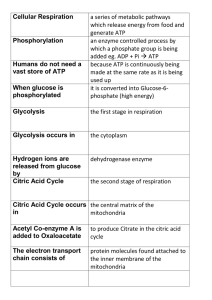
... because ATP is continuously being made at the same rate as it is being used up it is converted into Glucose-6phosphate (high energy) ...
... because ATP is continuously being made at the same rate as it is being used up it is converted into Glucose-6phosphate (high energy) ...
Oxidative phosphorylation (mitochondria)
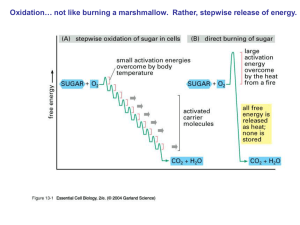
... Oxidation… not like burning a marshmallow. Rather, stepwise release of energy. ...
... Oxidation… not like burning a marshmallow. Rather, stepwise release of energy. ...
Electron Transport Chain _ETC
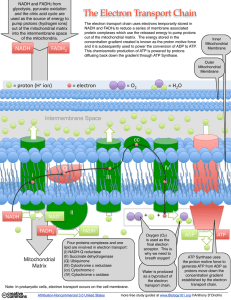
... captured and stored by the production of ATP from ADP and inorganic phosphate (Pi). This process is called oxidative phosphorylation. The remainder of the free energy that not trapped as ATP is released as heat. ETC is a chain of protein (enzyme) complexes embedded in the inner mitochondrial membran ...
... captured and stored by the production of ATP from ADP and inorganic phosphate (Pi). This process is called oxidative phosphorylation. The remainder of the free energy that not trapped as ATP is released as heat. ETC is a chain of protein (enzyme) complexes embedded in the inner mitochondrial membran ...
File
... G1-5: The electron transport chain captures free energy from electrons in a series of coupled reactions that establish an electrochemical gradient across membranes. Evidence of student learning is a demonstrated understanding of each of the following: 1. Electron transport chain reactions occur in c ...
... G1-5: The electron transport chain captures free energy from electrons in a series of coupled reactions that establish an electrochemical gradient across membranes. Evidence of student learning is a demonstrated understanding of each of the following: 1. Electron transport chain reactions occur in c ...
Exam Name___________________________________
... 11) To reduce one molecule of O2 , ________ electron(s) must be passed through the electron transport chain and ________ molecule(s) of NADH is(are) oxidized. B) 4; 4 C) 1; 2 A) 1; 1 12) Which is a component of complex I? A) FMN B) FAD ...
... 11) To reduce one molecule of O2 , ________ electron(s) must be passed through the electron transport chain and ________ molecule(s) of NADH is(are) oxidized. B) 4; 4 C) 1; 2 A) 1; 1 12) Which is a component of complex I? A) FMN B) FAD ...
T/F 1. Pyruvate, the end product of glycolysis, is processed
... 6. How many molecules of CO2 are produced for each molecule of glucose that passes through glycolysis and the Krebs cycle? a. 2 b. 3 c. 6 d. 7 7. The electrons generated from the Krebs cycle are transferred to ____________ and then are shuttled to _______________. a. NAD+ / oxygen b. NAD+ / electron ...
... 6. How many molecules of CO2 are produced for each molecule of glucose that passes through glycolysis and the Krebs cycle? a. 2 b. 3 c. 6 d. 7 7. The electrons generated from the Krebs cycle are transferred to ____________ and then are shuttled to _______________. a. NAD+ / oxygen b. NAD+ / electron ...
Microbial Metabolism - ASAB-NUST
... • Acetyl-CoA is energy rich because a high energy thiol links acetic acid to coenzyme A. ...
... • Acetyl-CoA is energy rich because a high energy thiol links acetic acid to coenzyme A. ...
Slide 1
... In Animals In the presence of oxygen pyruvate is transported into the mitochondria where it enters the next major metabolic pathway for the production of ATP energy. The citric acid cycle. Aerobic. If there is no oxygen present then the pyruvate is converted to a substance called lactate. ...
... In Animals In the presence of oxygen pyruvate is transported into the mitochondria where it enters the next major metabolic pathway for the production of ATP energy. The citric acid cycle. Aerobic. If there is no oxygen present then the pyruvate is converted to a substance called lactate. ...
What is the Electron Transport Chain?
... The electron transport chain uses electrons temporarily stored in NADH and FADH2 to reduce a series of membrane associated protein complexes which use the released energy to pump protons out of the mitochondrial matrix. The energy stored in the concentration gradient created is known as the proton m ...
... The electron transport chain uses electrons temporarily stored in NADH and FADH2 to reduce a series of membrane associated protein complexes which use the released energy to pump protons out of the mitochondrial matrix. The energy stored in the concentration gradient created is known as the proton m ...
Electron transport chain
An electron transport chain (ETC) is a series of compounds that transfer electrons from electron donors to electron acceptors via redox reactions, and couples this electron transfer with the transfer of protons (H+ ions) across a membrane. This creates an electrochemical proton gradient that drives ATP synthesis, or the generation of chemical energy in the form of adenosine triphosphate (ATP). The final acceptor of electrons in the electron transport chain is molecular oxygen.Electron transport chains are used for extracting energy via redox reactions from sunlight in photosynthesis or, such as in the case of the oxidation of sugars, cellular respiration. In eukaryotes, an important electron transport chain is found in the inner mitochondrial membrane where it serves as the site of oxidative phosphorylation through the use of ATP synthase. It is also found in the thylakoid membrane of the chloroplast in photosynthetic eukaryotes. In bacteria, the electron transport chain is located in their cell membrane.In chloroplasts, light drives the conversion of water to oxygen and NADP+ to NADPH with transfer of H+ ions across chloroplast membranes. In mitochondria, it is the conversion of oxygen to water, NADH to NAD+ and succinate to fumarate that are required to generate the proton gradient. Electron transport chains are major sites of premature electron leakage to oxygen, generating superoxide and potentially resulting in increased oxidative stress.