Atomic Structure
... Generally, when the level is full, extra electrons begin to fill up the next available energy level. In the case of the third level, after eight electrons have been added the next two electrons occupy the fourth level before the next ten electrons then complete the third level. Principal quantum ...
... Generally, when the level is full, extra electrons begin to fill up the next available energy level. In the case of the third level, after eight electrons have been added the next two electrons occupy the fourth level before the next ten electrons then complete the third level. Principal quantum ...
Chapter 2
... in a chemical reaction. Elements can only be converted into other elements in nuclear reactions. 3. All atoms of an element have the same number of protons and electrons, which determines the chemical behavior of the element. Isotopes of an element differ in the number of neutrons, and thus in mass ...
... in a chemical reaction. Elements can only be converted into other elements in nuclear reactions. 3. All atoms of an element have the same number of protons and electrons, which determines the chemical behavior of the element. Isotopes of an element differ in the number of neutrons, and thus in mass ...
Unit 1. Materials: Formulating Matter A. How do chemists describe
... 31. You melted and burned paraffin wax in the Lab Investigating Matter. Write the chemical formula of paraffin wax given its model below. (Note: The carbon and hydrogen atoms are smaller than in the key so that this molecule can fit on the page.) ...
... 31. You melted and burned paraffin wax in the Lab Investigating Matter. Write the chemical formula of paraffin wax given its model below. (Note: The carbon and hydrogen atoms are smaller than in the key so that this molecule can fit on the page.) ...
Question, hints, and answers. Look at hints if you need help. Look at
... 9. Which properties are most common in nonmetals? (1 point) *hint nonmetals tend to gain electrons more often and often you need a higher energy to remove an electron from it. Definition of electronegative and ionization energy. a ...
... 9. Which properties are most common in nonmetals? (1 point) *hint nonmetals tend to gain electrons more often and often you need a higher energy to remove an electron from it. Definition of electronegative and ionization energy. a ...
Review # 3
... What is the total number of nucleons (protons and neutrons) in an atom of selenium ( at. # = 34; mass # = 79) a. 34 b. 45 c. 79 d. 113 ...
... What is the total number of nucleons (protons and neutrons) in an atom of selenium ( at. # = 34; mass # = 79) a. 34 b. 45 c. 79 d. 113 ...
Calculating the Atomic Radius of Polonium
... The next step involves calculating the packing efficiency of the simple cubic structure ‐ in other words, the ratio of the atomic and effective volumes. We see that only 52.4% of the space is occupied by polonium atoms. ...
... The next step involves calculating the packing efficiency of the simple cubic structure ‐ in other words, the ratio of the atomic and effective volumes. We see that only 52.4% of the space is occupied by polonium atoms. ...
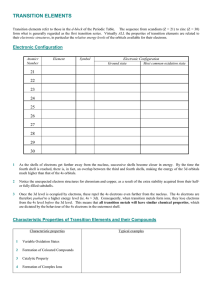
TRANSITION ELEMENTS
... Most of the compounds and complexes of transition elements are coloured. The colour of these compounds can often be related to incompletely filled d-orbitals in the transition metal ion. The outer electronic orbitals of transition metal ions have only small energy difference and many of these ions h ...
... Most of the compounds and complexes of transition elements are coloured. The colour of these compounds can often be related to incompletely filled d-orbitals in the transition metal ion. The outer electronic orbitals of transition metal ions have only small energy difference and many of these ions h ...
PS.3 The Atom Model
... PS.3 Atomic Model: The student will understand the historical & modern model of the atomic structure ...
... PS.3 Atomic Model: The student will understand the historical & modern model of the atomic structure ...
Essential Question: What type of model did Thompson ,Rutherford
... Corresponds to group number in the periodic table. Group 2 elements have 2 valence electrons: ...
... Corresponds to group number in the periodic table. Group 2 elements have 2 valence electrons: ...
Chemistry I Exams and Answer Keys 2015 Season
... A pure metal is made up of atoms that are held together by all valence electrons that are not held exclusively by any particular atoms, but move freely around them. This statement is best described as A. a correct definition of a chemical term or expression, either in terms of experimental behavior ...
... A pure metal is made up of atoms that are held together by all valence electrons that are not held exclusively by any particular atoms, but move freely around them. This statement is best described as A. a correct definition of a chemical term or expression, either in terms of experimental behavior ...
CHAPTER 4 TEST
... matter and are indivisible. Atoms of the same element are alike; Was able to base his theory on experimental evidence. Parts of his theory included the Law of Multiple Proportions (used to form compounds) and The Law of Conservation of Mass. ...
... matter and are indivisible. Atoms of the same element are alike; Was able to base his theory on experimental evidence. Parts of his theory included the Law of Multiple Proportions (used to form compounds) and The Law of Conservation of Mass. ...
Atoms and the Periodic Table
... successful than Democritus's theory because it had a scientific basis. ...
... successful than Democritus's theory because it had a scientific basis. ...
atoms - West Ada
... mass of 12.00000 amu(98.892%), and 13C with a mass of 13.00335 amu (1.108%) There are two isotopes of nitrogen , one with an atomic mass of 14.0031 amu and one with a mass of 15.0001 amu. What is the percent abundance of each? ...
... mass of 12.00000 amu(98.892%), and 13C with a mass of 13.00335 amu (1.108%) There are two isotopes of nitrogen , one with an atomic mass of 14.0031 amu and one with a mass of 15.0001 amu. What is the percent abundance of each? ...
atomic number - Cloudfront.net
... • By the 1700’s nearly all chemists had accepted the modern definition of an element as a particle that is indivisible • It was also understood at that time that elements combine to form compounds that are different in their properties than the elements that composed them – However, these understan ...
... • By the 1700’s nearly all chemists had accepted the modern definition of an element as a particle that is indivisible • It was also understood at that time that elements combine to form compounds that are different in their properties than the elements that composed them – However, these understan ...
Atomic Structures Part
... Each element differs from the preceding element by having one more positive charge in its nucleus ...
... Each element differs from the preceding element by having one more positive charge in its nucleus ...
Name_________________________ Date________
... Based on the results, he formulated his now famous Dalton’s Atomic Theory. ...
... Based on the results, he formulated his now famous Dalton’s Atomic Theory. ...
solid metal
... Names have several origins: Hydrogen is derived from Greek. Carbon is derived from Latin. Scandium is named for Scandinavia. ...
... Names have several origins: Hydrogen is derived from Greek. Carbon is derived from Latin. Scandium is named for Scandinavia. ...
Atomic number
... 1. All matter is composed of atoms. Although atoms are composed of smaller particles (electrons, protons, and neutrons), the atom is the smallest body that retains the unique identity of the element. 2. Atoms of one element cannot be converted into atoms of another element in a chemical reaction. El ...
... 1. All matter is composed of atoms. Although atoms are composed of smaller particles (electrons, protons, and neutrons), the atom is the smallest body that retains the unique identity of the element. 2. Atoms of one element cannot be converted into atoms of another element in a chemical reaction. El ...
History of the Atom Model
... distance from the nucleus. For example, if we were to draw a hydrogen atom to scale and used a 1-cm proton (about the size of this picture ), the atom's electron would spin at a distance of ~0.5 km from the nucleus. In other words, the atom would be larger than a football field! Elements / Atomic Nu ...
... distance from the nucleus. For example, if we were to draw a hydrogen atom to scale and used a 1-cm proton (about the size of this picture ), the atom's electron would spin at a distance of ~0.5 km from the nucleus. In other words, the atom would be larger than a football field! Elements / Atomic Nu ...
electron configuration
... electron occupies. Beginning on the leftmost element in this block, take the negative number of l, and count up until you hit the positive value for l. Start back over at – l if needed. Thus, count -2, -1, 0, 1, 2, -2, -1, 0, 1. Here, you hit gold, so ml =1. 4. Finally, find the magnetic spin number ...
... electron occupies. Beginning on the leftmost element in this block, take the negative number of l, and count up until you hit the positive value for l. Start back over at – l if needed. Thus, count -2, -1, 0, 1, 2, -2, -1, 0, 1. Here, you hit gold, so ml =1. 4. Finally, find the magnetic spin number ...
What do you already know about atoms?
... – Electrons exist in ‘clouds’ called orbitals w/ specific energy levels – Mathematical predictions for probability of finding electrons – Electrons have particle and wave properties ...
... – Electrons exist in ‘clouds’ called orbitals w/ specific energy levels – Mathematical predictions for probability of finding electrons – Electrons have particle and wave properties ...
Periodic table
The periodic table is a tabular arrangement of the chemical elements, ordered by their atomic number (number of protons in the nucleus), electron configurations, and recurring chemical properties. The table also shows four rectangular blocks: s-, p- d- and f-block. In general, within one row (period) the elements are metals on the lefthand side, and non-metals on the righthand side.The rows of the table are called periods; the columns are called groups. Six groups (columns) have names as well as numbers: for example, group 17 elements are the halogens; and group 18, the noble gases. The periodic table can be used to derive relationships between the properties of the elements, and predict the properties of new elements yet to be discovered or synthesized. The periodic table provides a useful framework for analyzing chemical behavior, and is widely used in chemistry and other sciences.Although precursors exist, Dmitri Mendeleev is generally credited with the publication, in 1869, of the first widely recognized periodic table. He developed his table to illustrate periodic trends in the properties of the then-known elements. Mendeleev also predicted some properties of then-unknown elements that would be expected to fill gaps in this table. Most of his predictions were proved correct when the elements in question were subsequently discovered. Mendeleev's periodic table has since been expanded and refined with the discovery or synthesis of further new elements and the development of new theoretical models to explain chemical behavior.All elements from atomic numbers 1 (hydrogen) to 118 (ununoctium) have been discovered or reportedly synthesized, with elements 113, 115, 117, and 118 having yet to be confirmed. The first 94 elements exist naturally, although some are found only in trace amounts and were synthesized in laboratories before being found in nature. Elements with atomic numbers from 95 to 118 have only been synthesized in laboratories. It has been shown that einsteinium and fermium once occurred in nature but currently do not. Synthesis of elements having higher atomic numbers is being pursued. Numerous synthetic radionuclides of naturally occurring elements have also been produced in laboratories.