Always in Motion
... The single electron would be found in the "K" shell and the "s" suborbital. If you go on to learn about chemistry, you may see its description written as 1s1. Helium (He) is still in the K shell (top row), but it has two electrons. The first electron would be 1s1 and the second would be 1s2. What ab ...
... The single electron would be found in the "K" shell and the "s" suborbital. If you go on to learn about chemistry, you may see its description written as 1s1. Helium (He) is still in the K shell (top row), but it has two electrons. The first electron would be 1s1 and the second would be 1s2. What ab ...
Lecture 11 - U of L Class Index
... medieval alchemy. To alchemists, any solid substance that did not melt and was not changed by fire into another substance was called an ‘‘earth.’’ Various compounds of Group 1 and 2 elements that were known in those times, such as NaOH and CaO, were alkaline according to the experimental tests of th ...
... medieval alchemy. To alchemists, any solid substance that did not melt and was not changed by fire into another substance was called an ‘‘earth.’’ Various compounds of Group 1 and 2 elements that were known in those times, such as NaOH and CaO, were alkaline according to the experimental tests of th ...
1.1 - cloudfront.net
... Each carbon atom has the same number of protons (6), which is equal to its atomic number. Each carbon atom also contains six electrons, allowing the atom to remain electrically neutral. However the number of neutrons varies from six to eight. Isotopes are atoms that have the same atomic number but d ...
... Each carbon atom has the same number of protons (6), which is equal to its atomic number. Each carbon atom also contains six electrons, allowing the atom to remain electrically neutral. However the number of neutrons varies from six to eight. Isotopes are atoms that have the same atomic number but d ...
Reduction and Emergence in Chemistry - Philsci
... number of anti-bonding electrons the molecule is predicted to be a stable one.1 Moreover, the electrons occupy these orbitals two by two in pairs. The deeper understanding lies in the fact that the electrons are regarded as spinning in opposite directions within all such pairs. Indeed it is the exch ...
... number of anti-bonding electrons the molecule is predicted to be a stable one.1 Moreover, the electrons occupy these orbitals two by two in pairs. The deeper understanding lies in the fact that the electrons are regarded as spinning in opposite directions within all such pairs. Indeed it is the exch ...
Reduction and Emergence in Chemistry
... number of anti-bonding electrons the molecule is predicted to be a stable one.1 Moreover, the electrons occupy these orbitals two by two in pairs. The deeper understanding lies in the fact that the electrons are regarded as spinning in opposite directions within all such pairs. Indeed it is the exch ...
... number of anti-bonding electrons the molecule is predicted to be a stable one.1 Moreover, the electrons occupy these orbitals two by two in pairs. The deeper understanding lies in the fact that the electrons are regarded as spinning in opposite directions within all such pairs. Indeed it is the exch ...
Which of the following statements correctly describes the relative
... Which of the following statements accurately describes the locations of the three subatomic particles that make up an atom? ...
... Which of the following statements accurately describes the locations of the three subatomic particles that make up an atom? ...
Atoms: The Building Blocks of Matter
... • Electrons act like particles (because they have a mass) and waves (because they have certain frequencies corresponding to their energy levels) • Electrons are located in orbitals around the nucleus that correspond to specific energy levels • Electron clouds = orbitals that do not have sharp bounda ...
... • Electrons act like particles (because they have a mass) and waves (because they have certain frequencies corresponding to their energy levels) • Electrons are located in orbitals around the nucleus that correspond to specific energy levels • Electron clouds = orbitals that do not have sharp bounda ...
Ch 04 AtomicStructure
... The comparison of the number of atoms in a copper coin the size of a penny with the number of people on Earth is used to illustrate which of the following? A. that atoms are indivisible B. that atoms are very small C. that atoms are very large D. that in a copper penny, there is one atom for every p ...
... The comparison of the number of atoms in a copper coin the size of a penny with the number of people on Earth is used to illustrate which of the following? A. that atoms are indivisible B. that atoms are very small C. that atoms are very large D. that in a copper penny, there is one atom for every p ...
Atomic Structure power point
... STRUCTURE OF THE ATOM • THE FIRST DISCOVERY OF A SUBATOMIC PARTICLE RESULTED FROM THE INVESTIGATIONS INTO THE RELATIONSHIP BETWEEN ELECTRICITY AND MATTER. • IN THE LATE 1800’S, MANY EXPERIMENTS WERE PERFORMED IN WHICH ELECTRIC CURRENT WAS PASSED THROUGH VARIOUS ...
... STRUCTURE OF THE ATOM • THE FIRST DISCOVERY OF A SUBATOMIC PARTICLE RESULTED FROM THE INVESTIGATIONS INTO THE RELATIONSHIP BETWEEN ELECTRICITY AND MATTER. • IN THE LATE 1800’S, MANY EXPERIMENTS WERE PERFORMED IN WHICH ELECTRIC CURRENT WAS PASSED THROUGH VARIOUS ...
SEKOLAH MENENGAH KEBANGSAAN RAJA PEREMPUAN, IPOH
... a. the similarities in chemical properties of lithium, sodium and potassium b. the relationship between the chemical properties of Group 1 elements and their electron arrangements Study the reaction of lithium, sodium and potassium with chlorine and bromine through computer simulation. Discuss chang ...
... a. the similarities in chemical properties of lithium, sodium and potassium b. the relationship between the chemical properties of Group 1 elements and their electron arrangements Study the reaction of lithium, sodium and potassium with chlorine and bromine through computer simulation. Discuss chang ...
Atoms - Chemistry R: 4(AE)
... Only a very concentrated (dense) positive charge in a tiny space within the gold atom could possibly repel the fast-moving, positively charged alpha particles enough to reverse the direction of the alpha particles. ...
... Only a very concentrated (dense) positive charge in a tiny space within the gold atom could possibly repel the fast-moving, positively charged alpha particles enough to reverse the direction of the alpha particles. ...
Atoms, Isotopes, and Ions.pptx
... • Atomic Number = 1 (stays the same as normal hydrogen because the protons stay the same) ...
... • Atomic Number = 1 (stays the same as normal hydrogen because the protons stay the same) ...
Chapter 4 - The Structure of the Atom Atomic Models PIONEERS OF
... o Composed of electrons, protons, and neutrons The protons and the neutrons make up the center of the atom. o Center of the atom is called the nucleus. The protons and neutrons occupy the mass of the atom. The electrons surround the nucleus and occupy most of the atom’s volume How, then, are atoms o ...
... o Composed of electrons, protons, and neutrons The protons and the neutrons make up the center of the atom. o Center of the atom is called the nucleus. The protons and neutrons occupy the mass of the atom. The electrons surround the nucleus and occupy most of the atom’s volume How, then, are atoms o ...
Chapter 3
... b. Atoms of a given element are identical in size, mass, and other properties. c. Atoms cannot be subdivided, created, or destroyed. d. Atoms of different chemical elements combine in simple whole-number ratios to form chemical compounds. ...
... b. Atoms of a given element are identical in size, mass, and other properties. c. Atoms cannot be subdivided, created, or destroyed. d. Atoms of different chemical elements combine in simple whole-number ratios to form chemical compounds. ...
Chemistry Ch3 Honors
... • His experiments showed that the nucleus is very small and positively charged. • He also hypothesized that the mass of the nucleus must be larger than the mass of the alpha particles, otherwise the alpha particles would have knocked the nucleus out of the way. • He also argued that most of the alph ...
... • His experiments showed that the nucleus is very small and positively charged. • He also hypothesized that the mass of the nucleus must be larger than the mass of the alpha particles, otherwise the alpha particles would have knocked the nucleus out of the way. • He also argued that most of the alph ...
PSI AP CHEMISTRY Summer Assignment Review Unit Free
... a) Give the number of electrons, protons, and neutrons in an atom of americium 241. b) Write the proper nuclide symbol. 2. What characteristics do atoms of carbon-12, carbon-13, and carbon-14 have in common? IN what ways are they different? 3. Identify the isotope that has atoms with a) 117 neutrons ...
... a) Give the number of electrons, protons, and neutrons in an atom of americium 241. b) Write the proper nuclide symbol. 2. What characteristics do atoms of carbon-12, carbon-13, and carbon-14 have in common? IN what ways are they different? 3. Identify the isotope that has atoms with a) 117 neutrons ...
3.091 - Introduction to Solid State Chemistry Lecture Notes No
... Consider the atom of an element containing one extra-nuclear electron: hydrogen; for electro-neutrality the charge on the nucleus must be +1. This orbiting electron (in the ground state - the lowest possible, most stable state) will have the lowest available quantum numbers in n, l and m. That is, n ...
... Consider the atom of an element containing one extra-nuclear electron: hydrogen; for electro-neutrality the charge on the nucleus must be +1. This orbiting electron (in the ground state - the lowest possible, most stable state) will have the lowest available quantum numbers in n, l and m. That is, n ...
TOPIC 12. THE ELEMENTS
... elements as compounds. Few non-gaseous elements are found in the free state. For thousands of years gold, silver, copper, sulfur and carbon had been known because they do occur in the free form, although they were not necessarily recognised as elements - indeed the concept of an element as we know i ...
... elements as compounds. Few non-gaseous elements are found in the free state. For thousands of years gold, silver, copper, sulfur and carbon had been known because they do occur in the free form, although they were not necessarily recognised as elements - indeed the concept of an element as we know i ...
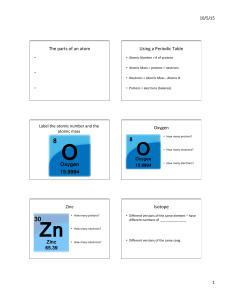
Periodic table
The periodic table is a tabular arrangement of the chemical elements, ordered by their atomic number (number of protons in the nucleus), electron configurations, and recurring chemical properties. The table also shows four rectangular blocks: s-, p- d- and f-block. In general, within one row (period) the elements are metals on the lefthand side, and non-metals on the righthand side.The rows of the table are called periods; the columns are called groups. Six groups (columns) have names as well as numbers: for example, group 17 elements are the halogens; and group 18, the noble gases. The periodic table can be used to derive relationships between the properties of the elements, and predict the properties of new elements yet to be discovered or synthesized. The periodic table provides a useful framework for analyzing chemical behavior, and is widely used in chemistry and other sciences.Although precursors exist, Dmitri Mendeleev is generally credited with the publication, in 1869, of the first widely recognized periodic table. He developed his table to illustrate periodic trends in the properties of the then-known elements. Mendeleev also predicted some properties of then-unknown elements that would be expected to fill gaps in this table. Most of his predictions were proved correct when the elements in question were subsequently discovered. Mendeleev's periodic table has since been expanded and refined with the discovery or synthesis of further new elements and the development of new theoretical models to explain chemical behavior.All elements from atomic numbers 1 (hydrogen) to 118 (ununoctium) have been discovered or reportedly synthesized, with elements 113, 115, 117, and 118 having yet to be confirmed. The first 94 elements exist naturally, although some are found only in trace amounts and were synthesized in laboratories before being found in nature. Elements with atomic numbers from 95 to 118 have only been synthesized in laboratories. It has been shown that einsteinium and fermium once occurred in nature but currently do not. Synthesis of elements having higher atomic numbers is being pursued. Numerous synthetic radionuclides of naturally occurring elements have also been produced in laboratories.