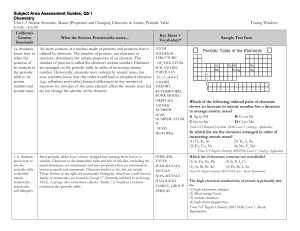
Name: Period:______ Table Number:______
... 51. Each element found on the periodic table of elements has a unique single letter (Hydrogen – H), two letter (Helium – He ) or three letter (Unnilquadiam – Unq) abbreviation which is called the CHEMICAL SYMBOL of that element. P. 83, Bill Nye the Science Guy Video 52. JONS BERZELIUS created the un ...
... 51. Each element found on the periodic table of elements has a unique single letter (Hydrogen – H), two letter (Helium – He ) or three letter (Unnilquadiam – Unq) abbreviation which is called the CHEMICAL SYMBOL of that element. P. 83, Bill Nye the Science Guy Video 52. JONS BERZELIUS created the un ...
Atoms and orbitals
... behavior that is unknown. While the mathematics for this can be formidable, the gist of the outcome is that we define regions where the electron is most likely to be found via experiment. These regions are called orbitals en-lieu or the atomic orbits above. These orbitals come in different flavors c ...
... behavior that is unknown. While the mathematics for this can be formidable, the gist of the outcome is that we define regions where the electron is most likely to be found via experiment. These regions are called orbitals en-lieu or the atomic orbits above. These orbitals come in different flavors c ...
"ALICE" CHAPTER 12 MODERN VIEW OF ATOMIC STRUCTURE
... Isotopes are atoms that have the same number of electrons and protons, but different numbers of {13}______________. As a result, {14}________________ of an element are atoms that have different masses. The atomic masses that appear on the periodic table are merely "weighted averages" of the masses o ...
... Isotopes are atoms that have the same number of electrons and protons, but different numbers of {13}______________. As a result, {14}________________ of an element are atoms that have different masses. The atomic masses that appear on the periodic table are merely "weighted averages" of the masses o ...
Practice Packet Level 3: Atomics - Mr. Palermo`s Flipped Chemistry
... 7.) In 1897, J. J. Thomson demonstrated in an experiment that cathode rays were deflected by an electric field. This suggested that cathode rays were composed of negatively charged particles found in al ...
... 7.) In 1897, J. J. Thomson demonstrated in an experiment that cathode rays were deflected by an electric field. This suggested that cathode rays were composed of negatively charged particles found in al ...
Subject Area Assessment Guides
... A few other groups are given family names. These include the alkali metals (Group 1), such as sodium and potassium, which are soft and white and extremely reactive chemically. Alkaline earth metals (Group 2), such as magnesium and calcium, are found in the second column of the periodic table. The tr ...
... A few other groups are given family names. These include the alkali metals (Group 1), such as sodium and potassium, which are soft and white and extremely reactive chemically. Alkaline earth metals (Group 2), such as magnesium and calcium, are found in the second column of the periodic table. The tr ...
Hydrogen, Alkalis, and Alkaline Earths
... Chemistry of the Main Group Elements: Hydrogen, Alkali and Alkaline Earth Metals Sections 8.1-8.4 Wednesday, November 4, 2015 ...
... Chemistry of the Main Group Elements: Hydrogen, Alkali and Alkaline Earth Metals Sections 8.1-8.4 Wednesday, November 4, 2015 ...
A an electron and an alpha particle B an electron and a proton C a
... empty space ® electrons exist in orbitals outside the nucleus B the atom is a hard sphere ® electrons exist in orbitals outside the nucleus ® most of the atom is empty space C most of the atom is empty space ® electrons exist in orbitals outside the nucleus ® the atom is a hard sphere D most of the ...
... empty space ® electrons exist in orbitals outside the nucleus B the atom is a hard sphere ® electrons exist in orbitals outside the nucleus ® most of the atom is empty space C most of the atom is empty space ® electrons exist in orbitals outside the nucleus ® the atom is a hard sphere D most of the ...
Activity 4 Are Atoms Indivisible?
... Prior to Thomson’s work with cathode rays many scientists were not sure that the cathode rays that were being produced were negatively charged. Scientists used partially evacuated tubes and a high-voltage source to produce radiation in the tube. Since the rays originated at the negative terminal or ...
... Prior to Thomson’s work with cathode rays many scientists were not sure that the cathode rays that were being produced were negatively charged. Scientists used partially evacuated tubes and a high-voltage source to produce radiation in the tube. Since the rays originated at the negative terminal or ...
Electron - CoolHub
... to or slightly greater than the number of protons. For example, the vast majority of carbon atoms have 6 protons and 6 neutrons, but a small percentage have 6 protons and 7 neutrons, and an even smaller percentage have 6 protons and 8 neutrons. Since the majority of carbon atoms have a mass very clo ...
... to or slightly greater than the number of protons. For example, the vast majority of carbon atoms have 6 protons and 6 neutrons, but a small percentage have 6 protons and 7 neutrons, and an even smaller percentage have 6 protons and 8 neutrons. Since the majority of carbon atoms have a mass very clo ...
A2 Module 2814: Chains, Rings and Spectroscopy
... A2 Module F325: Equilibria, Energetics and Elements EEE 2: Transition elements For the elements up to Ca the 3d orbitals are higher in energy than the 4s orbital. Therefore, after argon (element 18), the 4s orbital is filled: Ca has electron configuration [Ar] 4s2. From scandium on, the 3d orbitals ...
... A2 Module F325: Equilibria, Energetics and Elements EEE 2: Transition elements For the elements up to Ca the 3d orbitals are higher in energy than the 4s orbital. Therefore, after argon (element 18), the 4s orbital is filled: Ca has electron configuration [Ar] 4s2. From scandium on, the 3d orbitals ...
DEFINING THE ATOM - BradyMathScience
... 6. _______________________ 7. _______________________ 8. _______________________ ...
... 6. _______________________ 7. _______________________ 8. _______________________ ...
Unit 1 - doctortang.com
... alchemists. Their purpose was to find a chemical recipe to make gold from other less valuable metals. (We now know that it is only possible now if we can change the number of protons in the nucleus). In 1808, a British scientist by the name of John Dalton published his theory of atoms that would hav ...
... alchemists. Their purpose was to find a chemical recipe to make gold from other less valuable metals. (We now know that it is only possible now if we can change the number of protons in the nucleus). In 1808, a British scientist by the name of John Dalton published his theory of atoms that would hav ...
Electron - HCC Learning Web
... • Atoms of an element have the same number of protons but may differ in number of neutrons • Isotopes are two atoms of an element that differ in number of neutrons • Most isotopes are stable, but some are radioactive, giving off particles and energy ...
... • Atoms of an element have the same number of protons but may differ in number of neutrons • Isotopes are two atoms of an element that differ in number of neutrons • Most isotopes are stable, but some are radioactive, giving off particles and energy ...
Foundations of Atomic Theory
... Atoms cannot be subdivided, created, or destroyed. Atoms of different elements combine in simple whole-number ratios to form chemical compounds In chemical reactions, atoms are combined, separated, or rearranged. ...
... Atoms cannot be subdivided, created, or destroyed. Atoms of different elements combine in simple whole-number ratios to form chemical compounds In chemical reactions, atoms are combined, separated, or rearranged. ...
Chapter 3 - Higher Education | Kendall Hunt Publishing
... exactly like every other part. The salt can be separated from the water by physical means. Seawater and air are also examples of homogeneous mixtures. We know that there are two types of homogeneous matter: pure substances and homogeneous mixtures. According to this classification scheme, matter can ...
... exactly like every other part. The salt can be separated from the water by physical means. Seawater and air are also examples of homogeneous mixtures. We know that there are two types of homogeneous matter: pure substances and homogeneous mixtures. According to this classification scheme, matter can ...
Assignment # 6 Atomic Structure Drill
... These are enclosed in an evacuated (air removed) glass container and when a charge is applied, the electrons flow from anode to cathode through the open space of the glass container. Thomson observed these particles and determined that the particles: Move at a very high speed (about 10% the speed ...
... These are enclosed in an evacuated (air removed) glass container and when a charge is applied, the electrons flow from anode to cathode through the open space of the glass container. Thomson observed these particles and determined that the particles: Move at a very high speed (about 10% the speed ...
File - Riske Science
... As you are made from atoms, you are also mainly empty space. The particles which make up your mass would occupy the same volume as a flea if they were all squashed together, but a flea with your mass. This gives you an idea of the density of the nucleus. None of these subatomic particles can be (or ...
... As you are made from atoms, you are also mainly empty space. The particles which make up your mass would occupy the same volume as a flea if they were all squashed together, but a flea with your mass. This gives you an idea of the density of the nucleus. None of these subatomic particles can be (or ...
Thomson`s Model of the Atom
... Isotopes are atoms of the same element that have different numbers of neutrons and different mass numbers. To distinguish one isotope from another, the isotopes are referred by their mass numbers. For example, oxygen has 3 isotopes: oxygen-16, oxygen-17, and oxygen-18. All three oxygen isotopes can ...
... Isotopes are atoms of the same element that have different numbers of neutrons and different mass numbers. To distinguish one isotope from another, the isotopes are referred by their mass numbers. For example, oxygen has 3 isotopes: oxygen-16, oxygen-17, and oxygen-18. All three oxygen isotopes can ...
Atoms, Molecules and Ions Chapter 2
... • An atom or molecule can gain or lose more than one electron. • Many atoms gain or lose enough electrons to have the same number of electrons as the nearest noble gas (group 8A). ...
... • An atom or molecule can gain or lose more than one electron. • Many atoms gain or lose enough electrons to have the same number of electrons as the nearest noble gas (group 8A). ...
Periodic table
The periodic table is a tabular arrangement of the chemical elements, ordered by their atomic number (number of protons in the nucleus), electron configurations, and recurring chemical properties. The table also shows four rectangular blocks: s-, p- d- and f-block. In general, within one row (period) the elements are metals on the lefthand side, and non-metals on the righthand side.The rows of the table are called periods; the columns are called groups. Six groups (columns) have names as well as numbers: for example, group 17 elements are the halogens; and group 18, the noble gases. The periodic table can be used to derive relationships between the properties of the elements, and predict the properties of new elements yet to be discovered or synthesized. The periodic table provides a useful framework for analyzing chemical behavior, and is widely used in chemistry and other sciences.Although precursors exist, Dmitri Mendeleev is generally credited with the publication, in 1869, of the first widely recognized periodic table. He developed his table to illustrate periodic trends in the properties of the then-known elements. Mendeleev also predicted some properties of then-unknown elements that would be expected to fill gaps in this table. Most of his predictions were proved correct when the elements in question were subsequently discovered. Mendeleev's periodic table has since been expanded and refined with the discovery or synthesis of further new elements and the development of new theoretical models to explain chemical behavior.All elements from atomic numbers 1 (hydrogen) to 118 (ununoctium) have been discovered or reportedly synthesized, with elements 113, 115, 117, and 118 having yet to be confirmed. The first 94 elements exist naturally, although some are found only in trace amounts and were synthesized in laboratories before being found in nature. Elements with atomic numbers from 95 to 118 have only been synthesized in laboratories. It has been shown that einsteinium and fermium once occurred in nature but currently do not. Synthesis of elements having higher atomic numbers is being pursued. Numerous synthetic radionuclides of naturally occurring elements have also been produced in laboratories.