PowerPoint
... What is a weighted average? • A weighted average accounts for the percent abundance and mass of each isotope in an element. • Percent abundance is a measure of how common or rare that isotope is. • The average atomic mass of an element can be calculated by multiplying the mass of each isotope by it ...
... What is a weighted average? • A weighted average accounts for the percent abundance and mass of each isotope in an element. • Percent abundance is a measure of how common or rare that isotope is. • The average atomic mass of an element can be calculated by multiplying the mass of each isotope by it ...
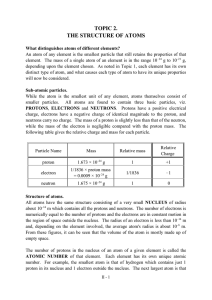
TOPIC 2. THE STRUCTURE OF ATOMS
... The formation of cations from atoms is not limited to those elements which have only 1 electron more than a noble gas. The atoms of the elements in the second group of Table 2 all have two more electrons in their outer orbits than a noble gas. These atoms require only a relatively small amount of en ...
... The formation of cations from atoms is not limited to those elements which have only 1 electron more than a noble gas. The atoms of the elements in the second group of Table 2 all have two more electrons in their outer orbits than a noble gas. These atoms require only a relatively small amount of en ...
Section 3 Electron Configurations
... Atomic Orbitals and Quantum Numbers • Quantum numbers specify the properties of atomic orbitals and the properties of electrons in orbitals. 1. The principal quantum number, symbolized by n, indicates the main energy level occupied by the electron. – Orbitals possible is n^2**** 2. The angular momen ...
... Atomic Orbitals and Quantum Numbers • Quantum numbers specify the properties of atomic orbitals and the properties of electrons in orbitals. 1. The principal quantum number, symbolized by n, indicates the main energy level occupied by the electron. – Orbitals possible is n^2**** 2. The angular momen ...
TOPIC 2. THE STRUCTURE OF ATOMS
... independent of those models. Experiments show that the electrons occupy only certain ORBITS around the nucleus, each orbit being characterised by its own associated energy and average distance from the nucleus. This model of an atom was proposed by Neils Bohr in 1919. In this model the orbits are gr ...
... independent of those models. Experiments show that the electrons occupy only certain ORBITS around the nucleus, each orbit being characterised by its own associated energy and average distance from the nucleus. This model of an atom was proposed by Neils Bohr in 1919. In this model the orbits are gr ...
Bohr, Niels Henrik David
... their atoms and that only the atomic weight and possible radioactive behaviour are determined by the small but massive nucleus itself. Rutherford's nuclear atom was both mechanically and electromagnetically unstable, but Bohr imposed stability on it by introducing the new and not yet clarified idea ...
... their atoms and that only the atomic weight and possible radioactive behaviour are determined by the small but massive nucleus itself. Rutherford's nuclear atom was both mechanically and electromagnetically unstable, but Bohr imposed stability on it by introducing the new and not yet clarified idea ...
KISS Notes
... like sodium, always seem to react readily and vigorously. Others, like copper, always react slowly or not at all. ...
... like sodium, always seem to react readily and vigorously. Others, like copper, always react slowly or not at all. ...
Atoms and Elements - Melvin R. Kantz, Ph.D.
... oxygen for every 1.00 g of carbon carbon dioxide contains 2.67 g of oxygen for every 1.00 g of carbon since there are twice as many oxygen atoms per carbon atom in carbon dioxide than in carbon monoxide, the ...
... oxygen for every 1.00 g of carbon carbon dioxide contains 2.67 g of oxygen for every 1.00 g of carbon since there are twice as many oxygen atoms per carbon atom in carbon dioxide than in carbon monoxide, the ...
Materials Required
... 5. Indicate the number of protons and neutrons in the nucleus of a particular element or isotope of an element 6. Define isotope as having a different number of neutrons Standards Ohio Standard: Physical Sciences 9-10 Benchmark A: Describe that matter is made of minute particles called atoms and ato ...
... 5. Indicate the number of protons and neutrons in the nucleus of a particular element or isotope of an element 6. Define isotope as having a different number of neutrons Standards Ohio Standard: Physical Sciences 9-10 Benchmark A: Describe that matter is made of minute particles called atoms and ato ...
10 Chemistry
... • To saEsfy the octet rule and have stable configuraEon with 8 valence electrons, fluorine is pracEcally begging to gain 1 electron • On the other hand, fluorine (and the other halogens) isn’t too keen ...
... • To saEsfy the octet rule and have stable configuraEon with 8 valence electrons, fluorine is pracEcally begging to gain 1 electron • On the other hand, fluorine (and the other halogens) isn’t too keen ...
Document
... Any given element can have more than one isotope. To distinguish between the different isotopes of an atom, the element is named with its mass number, for example, lithium-7. Remember that the mass number is the number of protons and neutrons added together. When symbols are used to represent an iso ...
... Any given element can have more than one isotope. To distinguish between the different isotopes of an atom, the element is named with its mass number, for example, lithium-7. Remember that the mass number is the number of protons and neutrons added together. When symbols are used to represent an iso ...
XIX. Chemistry, High School
... Reference Materials and Tools Each student taking the high school Chemistry test was provided with a Chemistry Formula and Constants Sheet/Periodic Table of the Elements. Copies of both sides of this formula sheet follow the final question in this chapter. Each student also had sole access to a calc ...
... Reference Materials and Tools Each student taking the high school Chemistry test was provided with a Chemistry Formula and Constants Sheet/Periodic Table of the Elements. Copies of both sides of this formula sheet follow the final question in this chapter. Each student also had sole access to a calc ...
Section 2.6 Subatomic Particles: Protons, Neutrons and Electrons in
... Modern Atomic theory and the Laws that led to it The Discovery of the Electron The Structure of the Atom Subatomic Particles: Protons, Neutrons and Electrons in Atoms Finding Patterns: The Periodic Law and the Periodic ...
... Modern Atomic theory and the Laws that led to it The Discovery of the Electron The Structure of the Atom Subatomic Particles: Protons, Neutrons and Electrons in Atoms Finding Patterns: The Periodic Law and the Periodic ...
Introduction to Chemical Bonding
... the atoms to have a full outer level. When an atom loses or gains electrons, it becomes what is called an ion. An ion is no longer neutrally charged because it has different numbers of protons and electrons. Ions of opposite charges attract and so they stick together. The Oxidation number is the ove ...
... the atoms to have a full outer level. When an atom loses or gains electrons, it becomes what is called an ion. An ion is no longer neutrally charged because it has different numbers of protons and electrons. Ions of opposite charges attract and so they stick together. The Oxidation number is the ove ...
What is Matter? Classwork Name
... 1) If you were to split one atom of Oxygen in half, you would no longer have Oxygen. All atoms of Oxygen must have 8 Protons and, in order to be stable, must have 8 Electrons as well. 2) Model description: electrons with a negative charge surrounding the nucleus (neutral) which contains protons (pos ...
... 1) If you were to split one atom of Oxygen in half, you would no longer have Oxygen. All atoms of Oxygen must have 8 Protons and, in order to be stable, must have 8 Electrons as well. 2) Model description: electrons with a negative charge surrounding the nucleus (neutral) which contains protons (pos ...
Atomic Mass
... The atomic number tells you the number of protons in one an atom of an element. It also tells you the number of electrons in a neutral atom of that element. The atomic number gives the identity of an element as well as its location on the periodic table. No two different elements will have the same ...
... The atomic number tells you the number of protons in one an atom of an element. It also tells you the number of electrons in a neutral atom of that element. The atomic number gives the identity of an element as well as its location on the periodic table. No two different elements will have the same ...
Flexbook - Ions and Ion Formation
... The magnesium atom has two electrons in the outermost energy level. When those two are removed, the resulting Mg2+ ion has the same electron configuration as neon does, but it is smaller than neon because the magnesium ion has two more protons in the nucleus. The first two ionization energies for ma ...
... The magnesium atom has two electrons in the outermost energy level. When those two are removed, the resulting Mg2+ ion has the same electron configuration as neon does, but it is smaller than neon because the magnesium ion has two more protons in the nucleus. The first two ionization energies for ma ...
The Nuclear Atom
... The elements in a column (up and down) are known as a group. Several ways to label groups. North American convention: A and B designations. Elements belonging to the same group exhibit similar chemical and physical properties. “Halogens” are in group VII (VIIA or 17), “alkali metals”, “noble gases”… ...
... The elements in a column (up and down) are known as a group. Several ways to label groups. North American convention: A and B designations. Elements belonging to the same group exhibit similar chemical and physical properties. “Halogens” are in group VII (VIIA or 17), “alkali metals”, “noble gases”… ...
Is That a Fact!
... Shrinking Man. In this story, which was later made into the film called The Incredible Shrinking Man, a man is contaminated by a radioactive cloud and begins to shrink down to atomic size. Obtain a copy of Matheson’s story, and read excerpts to the students. The story suggests an eerie connection be ...
... Shrinking Man. In this story, which was later made into the film called The Incredible Shrinking Man, a man is contaminated by a radioactive cloud and begins to shrink down to atomic size. Obtain a copy of Matheson’s story, and read excerpts to the students. The story suggests an eerie connection be ...
Atomic structure and periodic table
... A periodic table is a horizontal and vertical arrangement of elements according to their atomic numbers. This table was successfully arranged in 1913 by the British scientist Henry Moseley from the previous work of the Russian Scientist Dmitri Mendeleev. The horizontal arrangement forms period. Atom ...
... A periodic table is a horizontal and vertical arrangement of elements according to their atomic numbers. This table was successfully arranged in 1913 by the British scientist Henry Moseley from the previous work of the Russian Scientist Dmitri Mendeleev. The horizontal arrangement forms period. Atom ...
MISE - Physical Basis of Chemistry
... working in ratio - using the “triangle”. Since individual weights appear in the periodic table, there has to be a mass standard, i.e., a reference mass - so that the ratio of atomic weights can become individual values. Since hydrogen was believed to be the lightest element , H was assigned the weig ...
... working in ratio - using the “triangle”. Since individual weights appear in the periodic table, there has to be a mass standard, i.e., a reference mass - so that the ratio of atomic weights can become individual values. Since hydrogen was believed to be the lightest element , H was assigned the weig ...
Student
... except that the odds of hitting the nucleus with one of their missiles (the alpha particles) were extremely low. The vast majority of particles went straight through the gold foil; they expected all of the particles to go through the foil. But a few were deflected or bounced back—the particles were ...
... except that the odds of hitting the nucleus with one of their missiles (the alpha particles) were extremely low. The vast majority of particles went straight through the gold foil; they expected all of the particles to go through the foil. But a few were deflected or bounced back—the particles were ...
Name Date Class 4.1 Follow Along Notes – Review Questions. How
... • By using __________________ methods, Dalton transformed Democritus’s ideas on atoms into a scientific theory. Dalton’s Atomic Theory Dalton studied the ________________ in which elements combine in chemical reactions. The result of his work is known as Dalton’s atomic theory. 1. All elements are c ...
... • By using __________________ methods, Dalton transformed Democritus’s ideas on atoms into a scientific theory. Dalton’s Atomic Theory Dalton studied the ________________ in which elements combine in chemical reactions. The result of his work is known as Dalton’s atomic theory. 1. All elements are c ...
Assignment 20 ELECTRONIC STRUCTURE OF ATOMS AND IONS I
... The elements in the Periodic Table are arranged according to increasing atomic number. Thus each new element in the sequence has a neutral atom containing not only one more proton but also one more electron than the element immediately preceding it. (Unfortunately, there is no similar convenient inf ...
... The elements in the Periodic Table are arranged according to increasing atomic number. Thus each new element in the sequence has a neutral atom containing not only one more proton but also one more electron than the element immediately preceding it. (Unfortunately, there is no similar convenient inf ...
p,d,f
... I) Review: Energy can travel through space by electromagnetic radiation … which are those forms of energy represented on the electromagnetic spectrum. All forms travel at the speed of light (in a vacuum) and exhibit the same type of wavelike behavior. All forms of electromagnetic radiation exhibit: ...
... I) Review: Energy can travel through space by electromagnetic radiation … which are those forms of energy represented on the electromagnetic spectrum. All forms travel at the speed of light (in a vacuum) and exhibit the same type of wavelike behavior. All forms of electromagnetic radiation exhibit: ...
Periodic table
The periodic table is a tabular arrangement of the chemical elements, ordered by their atomic number (number of protons in the nucleus), electron configurations, and recurring chemical properties. The table also shows four rectangular blocks: s-, p- d- and f-block. In general, within one row (period) the elements are metals on the lefthand side, and non-metals on the righthand side.The rows of the table are called periods; the columns are called groups. Six groups (columns) have names as well as numbers: for example, group 17 elements are the halogens; and group 18, the noble gases. The periodic table can be used to derive relationships between the properties of the elements, and predict the properties of new elements yet to be discovered or synthesized. The periodic table provides a useful framework for analyzing chemical behavior, and is widely used in chemistry and other sciences.Although precursors exist, Dmitri Mendeleev is generally credited with the publication, in 1869, of the first widely recognized periodic table. He developed his table to illustrate periodic trends in the properties of the then-known elements. Mendeleev also predicted some properties of then-unknown elements that would be expected to fill gaps in this table. Most of his predictions were proved correct when the elements in question were subsequently discovered. Mendeleev's periodic table has since been expanded and refined with the discovery or synthesis of further new elements and the development of new theoretical models to explain chemical behavior.All elements from atomic numbers 1 (hydrogen) to 118 (ununoctium) have been discovered or reportedly synthesized, with elements 113, 115, 117, and 118 having yet to be confirmed. The first 94 elements exist naturally, although some are found only in trace amounts and were synthesized in laboratories before being found in nature. Elements with atomic numbers from 95 to 118 have only been synthesized in laboratories. It has been shown that einsteinium and fermium once occurred in nature but currently do not. Synthesis of elements having higher atomic numbers is being pursued. Numerous synthetic radionuclides of naturally occurring elements have also been produced in laboratories.