Chapter 2 Notes
... Solution (a) The number of protons (22) is the atomic number of the element, which means this element is titanium (Ti). The mass number of this isotope is 22 + 26 = 48 (the sum of the protons and neutrons). Because the ion has three more protons than electrons, it has a net charge of 3+. Thus, the s ...
... Solution (a) The number of protons (22) is the atomic number of the element, which means this element is titanium (Ti). The mass number of this isotope is 22 + 26 = 48 (the sum of the protons and neutrons). Because the ion has three more protons than electrons, it has a net charge of 3+. Thus, the s ...
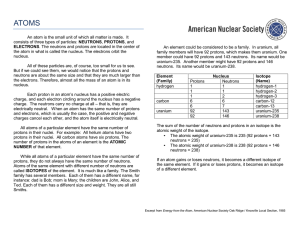
An atom is the small unit of which all matter is made. It consists of
... An atom is the small unit of which all matter is made. It consists of three types of particles: NEUTRONS, PROTONS, and ELECTRONS. The neutrons and protons are located in the center of the atom in what is called the nucleus. The electrons orbit the nucleus. All of these particles are, of course, too ...
... An atom is the small unit of which all matter is made. It consists of three types of particles: NEUTRONS, PROTONS, and ELECTRONS. The neutrons and protons are located in the center of the atom in what is called the nucleus. The electrons orbit the nucleus. All of these particles are, of course, too ...
The Periodic Law and Ionic Charge
... 3. Chlorine has 8 more protons in it’s nucleus than does fluorine. Provide a reason for it’s observed electronegativity (3.2) being lower than that of fluorine (4.0). 4. Draw a graph that shows the trend in metallic character for group 16 when plotted vs. atomic number. Do not use quantitative value ...
... 3. Chlorine has 8 more protons in it’s nucleus than does fluorine. Provide a reason for it’s observed electronegativity (3.2) being lower than that of fluorine (4.0). 4. Draw a graph that shows the trend in metallic character for group 16 when plotted vs. atomic number. Do not use quantitative value ...
Isi dengan Judul Halaman Terkait Adaptif
... energy first ionization of element increasing in one periods based on improvement of nuclear charge Solvent student of description and explains various energy first ionization of decline element in one factions based on improvement of atom radius ...
... energy first ionization of element increasing in one periods based on improvement of nuclear charge Solvent student of description and explains various energy first ionization of decline element in one factions based on improvement of atom radius ...
Chemistry Entrance Material for Grade 11 to 12
... Filtration: to separate heterogeneous mixture of solid in liquid 45. To obtain dry sand and salt from a mixture of sand and salt we need to follow which of the following steps and in what order? 1. Add excess water to the mixture and stir. 2. Heat the solution to crystallize. 3. Filter, and allow th ...
... Filtration: to separate heterogeneous mixture of solid in liquid 45. To obtain dry sand and salt from a mixture of sand and salt we need to follow which of the following steps and in what order? 1. Add excess water to the mixture and stir. 2. Heat the solution to crystallize. 3. Filter, and allow th ...
File
... 1. the positively charged subatomic particle called the electron 2. the positively charged subatomic particle called the proton 3. the positively charged subatomic particle called the electron 4. the negatively charged subatomic particle called the electron 2.) According to the Bohr Model, 1. electr ...
... 1. the positively charged subatomic particle called the electron 2. the positively charged subatomic particle called the proton 3. the positively charged subatomic particle called the electron 4. the negatively charged subatomic particle called the electron 2.) According to the Bohr Model, 1. electr ...
as a PDF
... the very large downward break after the half-filled shell, and the main contribution to it comes from the exchange energy [15]. This arises from the fact that electrons with parallel spins experience a smaller repulsion than do those with opposed spins. Blake [16] showed that whether one chooses the ...
... the very large downward break after the half-filled shell, and the main contribution to it comes from the exchange energy [15]. This arises from the fact that electrons with parallel spins experience a smaller repulsion than do those with opposed spins. Blake [16] showed that whether one chooses the ...
The Cubic Atomic Model
... proton and electron have a definite size and possesses a non-zero radius. Objects we see in the world have a definite size, so it is not unreasonable to assume that protons and electrons take up measureable amounts of space and are not theoretical point particles that have zero radius. A particle of ...
... proton and electron have a definite size and possesses a non-zero radius. Objects we see in the world have a definite size, so it is not unreasonable to assume that protons and electrons take up measureable amounts of space and are not theoretical point particles that have zero radius. A particle of ...
Document
... b) Chlorine contains 17 protons and 18.5 neutrons c) Chlorine normally exists in an excited state, and so it weighs more than 35. d) The chlorine was not pure when its atomic mass was measured. e) Chlorine, as found in nature, contains a mixture of the isotopes 35Cl and 37Cl, in such proportions as ...
... b) Chlorine contains 17 protons and 18.5 neutrons c) Chlorine normally exists in an excited state, and so it weighs more than 35. d) The chlorine was not pure when its atomic mass was measured. e) Chlorine, as found in nature, contains a mixture of the isotopes 35Cl and 37Cl, in such proportions as ...
BỘ GIÁO DỤC VÀ ĐÀO TẠO - THPT Chuyên Võ Nguyên Giáp
... atomic number and does not alter the identity of the element. For this reason, it is possible to have two atoms of the same element with differing mass numbers, because they have different numbers of neutrons. Atoms of the same element with different masses are called isotopes. For example, there ar ...
... atomic number and does not alter the identity of the element. For this reason, it is possible to have two atoms of the same element with differing mass numbers, because they have different numbers of neutrons. Atoms of the same element with different masses are called isotopes. For example, there ar ...
A Review of High School Chemistry
... Long about 1870, Mendeleev was inspired to put together a tabulation of the known elements in such a way as to describe in a periodic way their physical and chemical properties. The modern version of Mendeleev’s Periodic Table is an attractive addition to most science lecture halls—it is worth study ...
... Long about 1870, Mendeleev was inspired to put together a tabulation of the known elements in such a way as to describe in a periodic way their physical and chemical properties. The modern version of Mendeleev’s Periodic Table is an attractive addition to most science lecture halls—it is worth study ...
A Level Chemistry.pub
... You will need to have done well in Chemistry at GCSE (which will normally mean at least a B Grade) or to have performed strongly in chemical elements of Combined Science. ...
... You will need to have done well in Chemistry at GCSE (which will normally mean at least a B Grade) or to have performed strongly in chemical elements of Combined Science. ...
Chapter 4
... Introduction to the Modern Concept of Atomic Structure Isotopes Introduction to the Periodic Table Natural States of the Elements ...
... Introduction to the Modern Concept of Atomic Structure Isotopes Introduction to the Periodic Table Natural States of the Elements ...
Atomic Structure
... atomic structure structure of an atom chemistry - each atom consists of a very small nucleus composed of protons and neutrons which is encircled by moving electrons some of the important properties depend on, chem4kids com atoms structure - chem4kids com this tutorial introduces atomic structure in ...
... atomic structure structure of an atom chemistry - each atom consists of a very small nucleus composed of protons and neutrons which is encircled by moving electrons some of the important properties depend on, chem4kids com atoms structure - chem4kids com this tutorial introduces atomic structure in ...
Regents Chemistry Topic Review Packet
... You can recognize an excited state electron configuration. If the configuration does not match that on the Periodic Table for that number of electrons, then it is an excited state. 9. When an electron returns from a higher energy state to a lower energy state, it emits a specific amount of energy ...
... You can recognize an excited state electron configuration. If the configuration does not match that on the Periodic Table for that number of electrons, then it is an excited state. 9. When an electron returns from a higher energy state to a lower energy state, it emits a specific amount of energy ...
Atom
... kinds of atoms. _____________________ 3. Discovered radioactivity while working with a sample of Uranium. _____________________ 4. Determined that a cathode ray is made of electrons. _____________________ 5. Isolated two new elements, radium and polonium. _____________________ 6. Understood the atom ...
... kinds of atoms. _____________________ 3. Discovered radioactivity while working with a sample of Uranium. _____________________ 4. Determined that a cathode ray is made of electrons. _____________________ 5. Isolated two new elements, radium and polonium. _____________________ 6. Understood the atom ...
MISE - Physical Basis of Chemistry
... working in ratio - using the “triangle”. Since individual weights appear in the periodic table, there has to be a mass standard, i.e., a reference mass - so that the ratio of atomic weights can become individual values. Since hydrogen was believed to be the lightest element , H was assigned the weig ...
... working in ratio - using the “triangle”. Since individual weights appear in the periodic table, there has to be a mass standard, i.e., a reference mass - so that the ratio of atomic weights can become individual values. Since hydrogen was believed to be the lightest element , H was assigned the weig ...
Nuts,Bolts and Isotopes- Average Atomic Mass Activity
... (for example carbon is composed of carbon atoms). However, not all of the atoms found in that element are the same. For example, carbon contains three different types of atoms (carbon-12, 13 and 14). Each atom has the same number of protons and electrons but differing numbers of neutrons. These are ...
... (for example carbon is composed of carbon atoms). However, not all of the atoms found in that element are the same. For example, carbon contains three different types of atoms (carbon-12, 13 and 14). Each atom has the same number of protons and electrons but differing numbers of neutrons. These are ...
Term 1 and 2 Powerpoints
... • Title: The word “title” is written and underlined; followed then by the name of the lab. • Purpose: The word “purpose” is written and underlined; followed by the purpose of the lab. • Procedure: Usually extremely detailed. You can summarize. Just a couple of sentences is fine. Procedure questions ...
... • Title: The word “title” is written and underlined; followed then by the name of the lab. • Purpose: The word “purpose” is written and underlined; followed by the purpose of the lab. • Procedure: Usually extremely detailed. You can summarize. Just a couple of sentences is fine. Procedure questions ...
Chapter 4
... b. An iron atom has 15 orbitals that contain electrons. They consist of one 1s orbital, one 2s orbital, three 2p orbitals, one 3s orbital, three 3p orbitals, five 3d orbitals, and one 4s orbital. Eleven of these orbitals are filled, and there are four unpaired electrons. They are located in the 3d s ...
... b. An iron atom has 15 orbitals that contain electrons. They consist of one 1s orbital, one 2s orbital, three 2p orbitals, one 3s orbital, three 3p orbitals, five 3d orbitals, and one 4s orbital. Eleven of these orbitals are filled, and there are four unpaired electrons. They are located in the 3d s ...
2. CHEMICAL ACTIVITY of the METALS 3. PATTERNS of the
... and is not i)........................................ like stone • metal can made into intricate shapes, such as j)......................................., not possible in stone. ...
... and is not i)........................................ like stone • metal can made into intricate shapes, such as j)......................................., not possible in stone. ...
Introductory Chemistry: A Foundation FOURTH EDITION by Steven
... • All atoms of an element have the same number of protons • The number of protons in an atom of a given element is the same as the atomic number (found on the periodic table) • Atoms of an element with different numbers of neutrons are called isotopes • All isotopes of an element are chemically iden ...
... • All atoms of an element have the same number of protons • The number of protons in an atom of a given element is the same as the atomic number (found on the periodic table) • Atoms of an element with different numbers of neutrons are called isotopes • All isotopes of an element are chemically iden ...
Periodic table
The periodic table is a tabular arrangement of the chemical elements, ordered by their atomic number (number of protons in the nucleus), electron configurations, and recurring chemical properties. The table also shows four rectangular blocks: s-, p- d- and f-block. In general, within one row (period) the elements are metals on the lefthand side, and non-metals on the righthand side.The rows of the table are called periods; the columns are called groups. Six groups (columns) have names as well as numbers: for example, group 17 elements are the halogens; and group 18, the noble gases. The periodic table can be used to derive relationships between the properties of the elements, and predict the properties of new elements yet to be discovered or synthesized. The periodic table provides a useful framework for analyzing chemical behavior, and is widely used in chemistry and other sciences.Although precursors exist, Dmitri Mendeleev is generally credited with the publication, in 1869, of the first widely recognized periodic table. He developed his table to illustrate periodic trends in the properties of the then-known elements. Mendeleev also predicted some properties of then-unknown elements that would be expected to fill gaps in this table. Most of his predictions were proved correct when the elements in question were subsequently discovered. Mendeleev's periodic table has since been expanded and refined with the discovery or synthesis of further new elements and the development of new theoretical models to explain chemical behavior.All elements from atomic numbers 1 (hydrogen) to 118 (ununoctium) have been discovered or reportedly synthesized, with elements 113, 115, 117, and 118 having yet to be confirmed. The first 94 elements exist naturally, although some are found only in trace amounts and were synthesized in laboratories before being found in nature. Elements with atomic numbers from 95 to 118 have only been synthesized in laboratories. It has been shown that einsteinium and fermium once occurred in nature but currently do not. Synthesis of elements having higher atomic numbers is being pursued. Numerous synthetic radionuclides of naturally occurring elements have also been produced in laboratories.