Full text
... and many others. Sometimes it was dangerous to support atomic views. Giordiano Bruno and Galileo were prosecuted and condemned, the first to be burned, the second to life imprisonment, which happened not only for propagating the heliocentric theory of Copernicus but also for supporting the theory of ...
... and many others. Sometimes it was dangerous to support atomic views. Giordiano Bruno and Galileo were prosecuted and condemned, the first to be burned, the second to life imprisonment, which happened not only for propagating the heliocentric theory of Copernicus but also for supporting the theory of ...
1 Which of the following has the least mass
... C isotope D quark 2 Which of the following has the least mass in an atom? A neutron B proton C electron D nucleus 3 Which of the following showed electrons in specific, fixed orbits? A Rutherford Model B Planck Model C Bohr Model D Quantum Model 4 An atom that is electrically neutral must contain A ...
... C isotope D quark 2 Which of the following has the least mass in an atom? A neutron B proton C electron D nucleus 3 Which of the following showed electrons in specific, fixed orbits? A Rutherford Model B Planck Model C Bohr Model D Quantum Model 4 An atom that is electrically neutral must contain A ...
Joseph Proust: Law of Definite Proportions
... mass in the center of the atom. The realization that things were not just loosely "hanging out" around an atom but instead are arranged in a specific way around a center led him to discover the Atomic Nucleus. See diagram below ...
... mass in the center of the atom. The realization that things were not just loosely "hanging out" around an atom but instead are arranged in a specific way around a center led him to discover the Atomic Nucleus. See diagram below ...
Ch 11 ppt: Introduction to Atoms
... theory in 1803. His theory stated that all substances are made of atoms. Atoms are small particles that cannot be created, divided, or destroyed. Atoms of the same element are exactly alike, and atoms of different elements are different. Atoms join with other atoms to make new substances. ...
... theory in 1803. His theory stated that all substances are made of atoms. Atoms are small particles that cannot be created, divided, or destroyed. Atoms of the same element are exactly alike, and atoms of different elements are different. Atoms join with other atoms to make new substances. ...
Electronic Structure and the Periodic Table
... Quantum model of the atom Schrödinger developed an equation to describe the behavior and energies of electrons in atoms. • His equation is used to plot the position of the electron relative to the nucleus as a function of time. • While the equation is too complicated to write here, we can still use ...
... Quantum model of the atom Schrödinger developed an equation to describe the behavior and energies of electrons in atoms. • His equation is used to plot the position of the electron relative to the nucleus as a function of time. • While the equation is too complicated to write here, we can still use ...
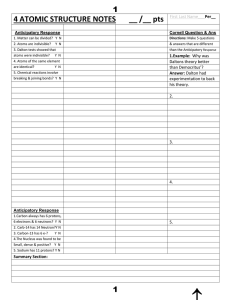
4 ATOMIC STRUCTURE NOTES __ /__ pts
... mass)?_____________________________________________________________________ 7. What happens when you cluster the electrons or move them into another level? They r______. Why do they behave the way they do?____________________________________________ 8. What do you get if the # of protons and electro ...
... mass)?_____________________________________________________________________ 7. What happens when you cluster the electrons or move them into another level? They r______. Why do they behave the way they do?____________________________________________ 8. What do you get if the # of protons and electro ...
Chapter 0 - Bakersfield College
... Most elements are mixtures of two or more stable isotopes Each isotope has slightly different mass Chemically, isotopes have virtually identical chemical properties Relative proportions of different isotopes are essentially constant Isotopes distinguished by mass number (A): e.g., Three ...
... Most elements are mixtures of two or more stable isotopes Each isotope has slightly different mass Chemically, isotopes have virtually identical chemical properties Relative proportions of different isotopes are essentially constant Isotopes distinguished by mass number (A): e.g., Three ...
Unit 6 1 Quantum Mechanics
... Hund’s Rule: When a sublevel contains degenerate orbitals, electrons are configured into the orbitals, one at a time, and are paired only when energy concerns become dominant. Pauli Exclusion Principle: No two electrons may share the exact same quantum numbers – hence, when two electrons occupy a si ...
... Hund’s Rule: When a sublevel contains degenerate orbitals, electrons are configured into the orbitals, one at a time, and are paired only when energy concerns become dominant. Pauli Exclusion Principle: No two electrons may share the exact same quantum numbers – hence, when two electrons occupy a si ...
Isotopes
... must equal the number of protons. Therefore, a sodium atom has 11 electrons in the space around its nucleus. It is always true that a sodium atom has 11 protons and 11 electrons. However, each sodium atom also has neutrons in its nucleus, and different types of sodium atoms exist that have different ...
... must equal the number of protons. Therefore, a sodium atom has 11 electrons in the space around its nucleus. It is always true that a sodium atom has 11 protons and 11 electrons. However, each sodium atom also has neutrons in its nucleus, and different types of sodium atoms exist that have different ...
Ch 3 Sec 3 Highlighted
... Mass Number: the total number of protons and neutrons in the nucleus of an isotope. From the periodic table the Mass Number is the atomic mass rounded to the nearest whole number. SECTION 3-3 OBJECTIVE 3 Given the identity of a nuclide, determine its number of protons, neutrons, and electrons. ...
... Mass Number: the total number of protons and neutrons in the nucleus of an isotope. From the periodic table the Mass Number is the atomic mass rounded to the nearest whole number. SECTION 3-3 OBJECTIVE 3 Given the identity of a nuclide, determine its number of protons, neutrons, and electrons. ...
Chemistry Entrance Material for Grade 11 to 12 Answer Key
... Definition of molar heat of vaporization 09. Define the molar heat of vaporization? The heat requires to change one mole of liquid to gas at constant temperature. Variation of molar heat of vaporization 10. Which of the following liquids has the lowest molar heat of vaporization? A Which one has the ...
... Definition of molar heat of vaporization 09. Define the molar heat of vaporization? The heat requires to change one mole of liquid to gas at constant temperature. Variation of molar heat of vaporization 10. Which of the following liquids has the lowest molar heat of vaporization? A Which one has the ...
The atom: Introduction and models
... There were, however, some problems with this model: for example it could not explain the very interesting observation that atoms only emit light at certain wavelengths or frequencies. Niels Bohr solved this problem by proposing that the electrons could only orbit the nucleus in certain special orbit ...
... There were, however, some problems with this model: for example it could not explain the very interesting observation that atoms only emit light at certain wavelengths or frequencies. Niels Bohr solved this problem by proposing that the electrons could only orbit the nucleus in certain special orbit ...
NSCC Chem 121 chapter2
... represents an isotope of nickel that contains 28 protons and 32 neutrons in the nucleus. • Isotopes are also represented by the notation: Name-A, where Name is the name of the element and A is the mass number of the isotope. • An example of this isotope notation is magnesium-26. This represents an i ...
... represents an isotope of nickel that contains 28 protons and 32 neutrons in the nucleus. • Isotopes are also represented by the notation: Name-A, where Name is the name of the element and A is the mass number of the isotope. • An example of this isotope notation is magnesium-26. This represents an i ...
4 Structure of The Atom
... the Sun. The stability of the atom could not be explained by this model. 7. Neils Bohr’s model best explains the model of atom. According to him, electrons are distributed in different shells/orbits/energy levels around the nucleus and are associated with discrete amounts of energy. When an atomi ...
... the Sun. The stability of the atom could not be explained by this model. 7. Neils Bohr’s model best explains the model of atom. According to him, electrons are distributed in different shells/orbits/energy levels around the nucleus and are associated with discrete amounts of energy. When an atomi ...
Balancing Chemical Equations – A Primer
... cannot be created or destroyed. If I start with two atoms of Na, there must be two Na atoms in the products. Likewise if there are two atoms of an element in the products, I must have started with two atoms of that element in the reactants. ...
... cannot be created or destroyed. If I start with two atoms of Na, there must be two Na atoms in the products. Likewise if there are two atoms of an element in the products, I must have started with two atoms of that element in the reactants. ...
Chapter 2
... • In this series of elements, the mass number (A) varies but the atomic number (Z) is constant. • This means that we are looking at a series of isotopes. ...
... • In this series of elements, the mass number (A) varies but the atomic number (Z) is constant. • This means that we are looking at a series of isotopes. ...
Chemistry (SPA)
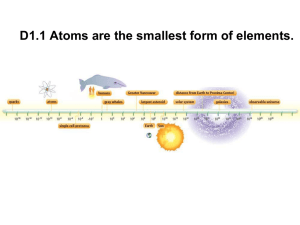
... For over 2000 years, people have wondered about the fundamental building blocks of matter. As far back as 440 BC, the Greek Leucippus and his pupil Democritus coined the term atomos to describe the smallest particle of matter. It translates to mean something that is indivisible. In the eighteenth ce ...
... For over 2000 years, people have wondered about the fundamental building blocks of matter. As far back as 440 BC, the Greek Leucippus and his pupil Democritus coined the term atomos to describe the smallest particle of matter. It translates to mean something that is indivisible. In the eighteenth ce ...
Step 2 - The Grange School Blogs
... Nanoscience is a new branch of science that refers to structures built from a few hundred atoms and are 1100nm big. They show different properties to the same materials in bulk. They also have a large surface area to volume ratio and their properties could lead to new developments in computers, buil ...
... Nanoscience is a new branch of science that refers to structures built from a few hundred atoms and are 1100nm big. They show different properties to the same materials in bulk. They also have a large surface area to volume ratio and their properties could lead to new developments in computers, buil ...
Step 2
... Nanoscience is a new branch of science that refers to structures built from a few hundred atoms and are 1100nm big. They show different properties to the same materials in bulk. They also have a large surface area to volume ratio and their properties could lead to new developments in computers, buil ...
... Nanoscience is a new branch of science that refers to structures built from a few hundred atoms and are 1100nm big. They show different properties to the same materials in bulk. They also have a large surface area to volume ratio and their properties could lead to new developments in computers, buil ...
Chapter 11 Atoms, Energy and Electron Configurations
... A. Electron Arrangements in the First 18 Atoms on the Periodic Table Classifying Electrons • Valence electrons – electrons in the outermost (highest) principal energy level of an atom • Core electrons – inner electrons • Elements with the same valence electron arrangement (same group) show very s ...
... A. Electron Arrangements in the First 18 Atoms on the Periodic Table Classifying Electrons • Valence electrons – electrons in the outermost (highest) principal energy level of an atom • Core electrons – inner electrons • Elements with the same valence electron arrangement (same group) show very s ...
Periodic table
The periodic table is a tabular arrangement of the chemical elements, ordered by their atomic number (number of protons in the nucleus), electron configurations, and recurring chemical properties. The table also shows four rectangular blocks: s-, p- d- and f-block. In general, within one row (period) the elements are metals on the lefthand side, and non-metals on the righthand side.The rows of the table are called periods; the columns are called groups. Six groups (columns) have names as well as numbers: for example, group 17 elements are the halogens; and group 18, the noble gases. The periodic table can be used to derive relationships between the properties of the elements, and predict the properties of new elements yet to be discovered or synthesized. The periodic table provides a useful framework for analyzing chemical behavior, and is widely used in chemistry and other sciences.Although precursors exist, Dmitri Mendeleev is generally credited with the publication, in 1869, of the first widely recognized periodic table. He developed his table to illustrate periodic trends in the properties of the then-known elements. Mendeleev also predicted some properties of then-unknown elements that would be expected to fill gaps in this table. Most of his predictions were proved correct when the elements in question were subsequently discovered. Mendeleev's periodic table has since been expanded and refined with the discovery or synthesis of further new elements and the development of new theoretical models to explain chemical behavior.All elements from atomic numbers 1 (hydrogen) to 118 (ununoctium) have been discovered or reportedly synthesized, with elements 113, 115, 117, and 118 having yet to be confirmed. The first 94 elements exist naturally, although some are found only in trace amounts and were synthesized in laboratories before being found in nature. Elements with atomic numbers from 95 to 118 have only been synthesized in laboratories. It has been shown that einsteinium and fermium once occurred in nature but currently do not. Synthesis of elements having higher atomic numbers is being pursued. Numerous synthetic radionuclides of naturally occurring elements have also been produced in laboratories.