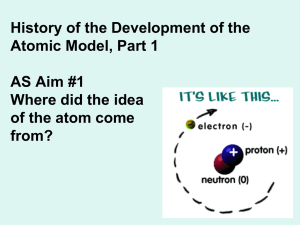
Name: Chemistry Notes: Chapter 1.1
... known naturally occurring elements. The smallest particle that makes up any type of e lement . All matter is made of atoms. Atoms are very very small. An atom is made up of 3 charged particles: 1. Protons—have a positive (+) charge 2. Neutrons—have no (o) charge (think: neutral) 3. ...
... known naturally occurring elements. The smallest particle that makes up any type of e lement . All matter is made of atoms. Atoms are very very small. An atom is made up of 3 charged particles: 1. Protons—have a positive (+) charge 2. Neutrons—have no (o) charge (think: neutral) 3. ...
The Atom and the Ion
... liquid element which is bromine. They have no luster, not malleable or ductile (brittle), they are bad conductors to heat and electricity, except graphite which is good conductor to electricity. Most of nonmetals contain 5,6 or 7 electrons in their outer shells. Nonmetals atoms are likely to gain el ...
... liquid element which is bromine. They have no luster, not malleable or ductile (brittle), they are bad conductors to heat and electricity, except graphite which is good conductor to electricity. Most of nonmetals contain 5,6 or 7 electrons in their outer shells. Nonmetals atoms are likely to gain el ...
Atomic Theory - WordPress.com
... Niels Bohr discovered that electrons orbit the nucleus like the planets around the sun in specific orbitals ...
... Niels Bohr discovered that electrons orbit the nucleus like the planets around the sun in specific orbitals ...
Key
... a) How many protons and neutrons are in the nucleus of an atom that has an atomic number of 27 and a mass number of 59? protons neutrons b) What is the symbol for the element? ...
... a) How many protons and neutrons are in the nucleus of an atom that has an atomic number of 27 and a mass number of 59? protons neutrons b) What is the symbol for the element? ...
File
... c. Although hydrogen is part of column 1, it is a non-metal. The reason why hydrogen is in this group is because it has one electron in its highest energy level. But since hydrogen has only one electron in total, it has properties that are different from other group 1 elements. Note: You will learn ...
... c. Although hydrogen is part of column 1, it is a non-metal. The reason why hydrogen is in this group is because it has one electron in its highest energy level. But since hydrogen has only one electron in total, it has properties that are different from other group 1 elements. Note: You will learn ...
File
... In the space in your answer booklet, show a numerical setup for calculating the atomic mass of copper. 83. Base your answer to the following question on the ...
... In the space in your answer booklet, show a numerical setup for calculating the atomic mass of copper. 83. Base your answer to the following question on the ...
2011 Chem Facts Key
... 48. Real gas particles have volume and are attracted to one another. They don"t always behave like ideal gases. Lighter gases (with weaker attractive forces) are often most ideal. Which of the following is the most ideal gas? He, Ne, Ar, Kr 49. Real gases behave more like ideal gases at low pressure ...
... 48. Real gas particles have volume and are attracted to one another. They don"t always behave like ideal gases. Lighter gases (with weaker attractive forces) are often most ideal. Which of the following is the most ideal gas? He, Ne, Ar, Kr 49. Real gases behave more like ideal gases at low pressure ...
Chapter 8 - Atomic Electron Configuration and Chemical Periodicity
... Effective Nuclear Charge, Z* Most electrons do not ‘feel’ the full positive charge of the nucleus. Other electrons in the atom (particularly those in lower energy orbitals) ‘shield’ some of this charge. The ___________ __________ __________ is the charge experienced by an electron in a multielectron ...
... Effective Nuclear Charge, Z* Most electrons do not ‘feel’ the full positive charge of the nucleus. Other electrons in the atom (particularly those in lower energy orbitals) ‘shield’ some of this charge. The ___________ __________ __________ is the charge experienced by an electron in a multielectron ...
Chemistry NYOS Dr. McPhee December 2015 Learning Objectives
... Given one of the families in the periodic table we learned, decide which orbitals are filling with electrons Given a suborbital shorthand electron configuration, determine the number of unpaired electrons Know how the Cr and Cu families differ in electron configuration from their neighbors Given an ...
... Given one of the families in the periodic table we learned, decide which orbitals are filling with electrons Given a suborbital shorthand electron configuration, determine the number of unpaired electrons Know how the Cr and Cu families differ in electron configuration from their neighbors Given an ...
Atoms, molecules and ions
... Chemical compound – when 2 or more different elements combine in a specific way to create a new material with different properties than elements alone. Na (soft, silver) Cl ( green gas) NaCl – table salt. Done by chemical reaction. Formula – list symbol each element in there. Subscript – tells the n ...
... Chemical compound – when 2 or more different elements combine in a specific way to create a new material with different properties than elements alone. Na (soft, silver) Cl ( green gas) NaCl – table salt. Done by chemical reaction. Formula – list symbol each element in there. Subscript – tells the n ...
Chapter 2: Matter is Made up of Atoms
... • In 1910 Thomson discovered that neon atoms have different masses. • In 1932, James Chadwick confirms existence of the neutron • Conclusion: there must be another particle that has no charge, called a neutron. ...
... • In 1910 Thomson discovered that neon atoms have different masses. • In 1932, James Chadwick confirms existence of the neutron • Conclusion: there must be another particle that has no charge, called a neutron. ...
worksheet #1 - chemistryrocks.net
... number of ______________________ in a neutral atom of that element. The atomic number gives the “identity “of an element as well as its location on the Periodic Table. No two different elements will have the ______________________ atomic number. [4] The ______________________ of an element is the av ...
... number of ______________________ in a neutral atom of that element. The atomic number gives the “identity “of an element as well as its location on the Periodic Table. No two different elements will have the ______________________ atomic number. [4] The ______________________ of an element is the av ...
1 Atomic Theory
... • In fact, it is impossible to determine the exact location of an electron. The probable location of an electron is based on how much energy the electron has. • According to the modern atomic model, at atom still has small positively charged nucleus surrounded by a large electron cloud region in whi ...
... • In fact, it is impossible to determine the exact location of an electron. The probable location of an electron is based on how much energy the electron has. • According to the modern atomic model, at atom still has small positively charged nucleus surrounded by a large electron cloud region in whi ...
3—3 Review and Reinforcement
... 8. 1 atomic mass unit (amu) is equal to one twelfth of the mass of a carbon-12 atom. 9. Atoms with the same number of protons but different numbers of electrons are called isotopes. ...
... 8. 1 atomic mass unit (amu) is equal to one twelfth of the mass of a carbon-12 atom. 9. Atoms with the same number of protons but different numbers of electrons are called isotopes. ...
Atomic Structure
... Valence electrons - The electrons in the outer most energy level Valence electrons determine the reactivity of an atom • Will it bond with other atoms? ...
... Valence electrons - The electrons in the outer most energy level Valence electrons determine the reactivity of an atom • Will it bond with other atoms? ...
Oxidation-Reduction (Redox) Reactions
... b. Balance O by adding H2O to the appropriate side. c. Balance H by adding the appropriate number of H+ ions to the side that needs H. Add H2O to the other side if needed. d. Balance net charge by adding electrons. Step 3: Multiply the half-reactions by integers that will allow for cancellation of e ...
... b. Balance O by adding H2O to the appropriate side. c. Balance H by adding the appropriate number of H+ ions to the side that needs H. Add H2O to the other side if needed. d. Balance net charge by adding electrons. Step 3: Multiply the half-reactions by integers that will allow for cancellation of e ...
Periodic table
The periodic table is a tabular arrangement of the chemical elements, ordered by their atomic number (number of protons in the nucleus), electron configurations, and recurring chemical properties. The table also shows four rectangular blocks: s-, p- d- and f-block. In general, within one row (period) the elements are metals on the lefthand side, and non-metals on the righthand side.The rows of the table are called periods; the columns are called groups. Six groups (columns) have names as well as numbers: for example, group 17 elements are the halogens; and group 18, the noble gases. The periodic table can be used to derive relationships between the properties of the elements, and predict the properties of new elements yet to be discovered or synthesized. The periodic table provides a useful framework for analyzing chemical behavior, and is widely used in chemistry and other sciences.Although precursors exist, Dmitri Mendeleev is generally credited with the publication, in 1869, of the first widely recognized periodic table. He developed his table to illustrate periodic trends in the properties of the then-known elements. Mendeleev also predicted some properties of then-unknown elements that would be expected to fill gaps in this table. Most of his predictions were proved correct when the elements in question were subsequently discovered. Mendeleev's periodic table has since been expanded and refined with the discovery or synthesis of further new elements and the development of new theoretical models to explain chemical behavior.All elements from atomic numbers 1 (hydrogen) to 118 (ununoctium) have been discovered or reportedly synthesized, with elements 113, 115, 117, and 118 having yet to be confirmed. The first 94 elements exist naturally, although some are found only in trace amounts and were synthesized in laboratories before being found in nature. Elements with atomic numbers from 95 to 118 have only been synthesized in laboratories. It has been shown that einsteinium and fermium once occurred in nature but currently do not. Synthesis of elements having higher atomic numbers is being pursued. Numerous synthetic radionuclides of naturally occurring elements have also been produced in laboratories.