Practice Problems in Biomedical Organic Chemistry
... This problem set was developed to assist undergraduate students taking a one semester or two semester, nonmajors course in organic chemistry. Students in these courses often come to organic chemistry from diverse backgrounds including biology, microbiology, and a variety of medical-related fields (e ...
... This problem set was developed to assist undergraduate students taking a one semester or two semester, nonmajors course in organic chemistry. Students in these courses often come to organic chemistry from diverse backgrounds including biology, microbiology, and a variety of medical-related fields (e ...
Document
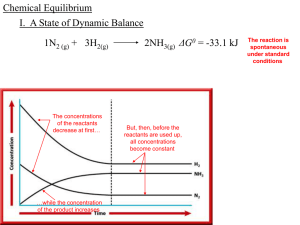
... The value of Kc for the reaction is 1.2 . The reaction is started with [H2 ]0 = 0.76 M, [N2]0 = 0.60 M and [NH3]0= 0.48 M. Which of the following is correct as the reaction comes to equilibrium? A) The concentration of N2will increase B) The concentration of H2will decrease C) The concentration of N ...
... The value of Kc for the reaction is 1.2 . The reaction is started with [H2 ]0 = 0.76 M, [N2]0 = 0.60 M and [NH3]0= 0.48 M. Which of the following is correct as the reaction comes to equilibrium? A) The concentration of N2will increase B) The concentration of H2will decrease C) The concentration of N ...
Document
... Suppose Si (having 4 valence electrons is doped with Ga (which has 3 valence electrons), 3 valence electrons are involved in bond formation with neighboring Si atom. A vacancy is left which can be filled by the transfer of a valence electron from a neighboring Si atom. The movement of electron into ...
... Suppose Si (having 4 valence electrons is doped with Ga (which has 3 valence electrons), 3 valence electrons are involved in bond formation with neighboring Si atom. A vacancy is left which can be filled by the transfer of a valence electron from a neighboring Si atom. The movement of electron into ...
Laboratory Works and Home Tasks in General Chemistry
... Normality (CN). Normality is defined as the number of moles of the equivalent of a solute (neq) in 1 liter of a solution; that is, CN = neq (solute) / V (solution). Thus, normality has the units of mol/L or N. In chemical reactions the molar ratio of reacting substances is not always 1:1. It is dete ...
... Normality (CN). Normality is defined as the number of moles of the equivalent of a solute (neq) in 1 liter of a solution; that is, CN = neq (solute) / V (solution). Thus, normality has the units of mol/L or N. In chemical reactions the molar ratio of reacting substances is not always 1:1. It is dete ...
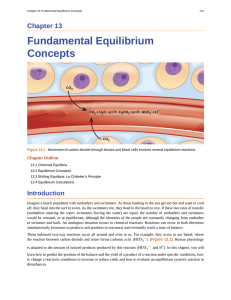
Fundamental Equilibrium Concepts
... In Example 13.2, it was mentioned that the common practice is to omit units when evaluating reaction quotients and equilibrium constants. It should be pointed out that using concentrations in these computations is a convenient but simplified approach that sometimes leads to results that seemingly co ...
... In Example 13.2, it was mentioned that the common practice is to omit units when evaluating reaction quotients and equilibrium constants. It should be pointed out that using concentrations in these computations is a convenient but simplified approach that sometimes leads to results that seemingly co ...
B.Sc Chemistry - Calicut University
... Here a detailed study is not expected, instead an introduction on the terms and concepts in chemistry is visualized. From a historical point of view Inorganic Chemistry is synonymous with general chemistry. An inorganic chemistry student is expected to be conversant with the chemistry of all the ele ...
... Here a detailed study is not expected, instead an introduction on the terms and concepts in chemistry is visualized. From a historical point of view Inorganic Chemistry is synonymous with general chemistry. An inorganic chemistry student is expected to be conversant with the chemistry of all the ele ...
Chemistry - Department of Education and Skills
... principles and concepts are more easily understood if they are demonstrated in the laboratory. The properties of particular substances are more fully appreciated if the student has the opportunity to ...
... principles and concepts are more easily understood if they are demonstrated in the laboratory. The properties of particular substances are more fully appreciated if the student has the opportunity to ...
AQA A-level Chemistry
... and products in a chemical reaction. The vertical (y) axis is enthalpy but not ∆H. The horizontal (x) axis is progress of reaction, reaction coordinate or extent of reaction. Two horizontal lines are drawn and labelled with the names or formulae of reactants and products. These represent the enthalp ...
... and products in a chemical reaction. The vertical (y) axis is enthalpy but not ∆H. The horizontal (x) axis is progress of reaction, reaction coordinate or extent of reaction. Two horizontal lines are drawn and labelled with the names or formulae of reactants and products. These represent the enthalp ...
LaBrake, Fundamentals Diagnostic Questions
... 16. Iced tea with lemon, ice, and sugar is an example of a) a heterogeneous mixture (correct) b) a homogenous mixture c) a mineral d) an element e) a compound 17. All of the following are considered subatomic particles, except: a) gamma rays (correct) b) electrons c) protons d) neutrons e) positron ...
... 16. Iced tea with lemon, ice, and sugar is an example of a) a heterogeneous mixture (correct) b) a homogenous mixture c) a mineral d) an element e) a compound 17. All of the following are considered subatomic particles, except: a) gamma rays (correct) b) electrons c) protons d) neutrons e) positron ...
1 Ag PO 7.5 10 1.79 10 418.57 mol x gL x M g
... Notice that the above expression is the product of a ratio of mole amounts and a volume3 term. The constant Kform does not change on dilution, but the volume term is changed by dilution. This means that the ratio of moles term in the above expression must change on dilution, in order to hold the pro ...
... Notice that the above expression is the product of a ratio of mole amounts and a volume3 term. The constant Kform does not change on dilution, but the volume term is changed by dilution. This means that the ratio of moles term in the above expression must change on dilution, in order to hold the pro ...
Equilibrium - pedagogics.ca
... In this case, increasing the temperature causes the position of equilibrium to be shifted to the left, i.e. there is less ammonia present at equilibrium at the higher temperature. The effect of a temperature change on a system at equilibrium can be now considered in terms of Le Chatelier’s principle ...
... In this case, increasing the temperature causes the position of equilibrium to be shifted to the left, i.e. there is less ammonia present at equilibrium at the higher temperature. The effect of a temperature change on a system at equilibrium can be now considered in terms of Le Chatelier’s principle ...
CHAPTER 4 REACTIONS IN AQUEOUS SOLUTIONS
... In dissolving a solid in a liquid, the solid is typically the solute, the substance present in a smaller amount, and the liquid is the solvent, the substance present in a larger amount. The two substances together form a solution, a homogeneous mixture of two or more substances. ...
... In dissolving a solid in a liquid, the solid is typically the solute, the substance present in a smaller amount, and the liquid is the solvent, the substance present in a larger amount. The two substances together form a solution, a homogeneous mixture of two or more substances. ...
Full-Text PDF
... activity coefficient γadd . Such an additive will be referred to as the equivalent additive. Equation (20) is quite general. It applies to any combination of additives be they liquids, solid suspensions, colloids, or any mixture thereof. Irrespective of the number and kind of additives, the values o ...
... activity coefficient γadd . Such an additive will be referred to as the equivalent additive. Equation (20) is quite general. It applies to any combination of additives be they liquids, solid suspensions, colloids, or any mixture thereof. Irrespective of the number and kind of additives, the values o ...
Chapter 4
... (c) is a weak electrolyte. A small amount of the compound dissociates into ions in water. When NaCl dissolves in water it dissociates into Na and Cl ions. When the ions are hydrated, the water molecules will be oriented so that the negative end of the water dipole interacts with the positive sodiu ...
... (c) is a weak electrolyte. A small amount of the compound dissociates into ions in water. When NaCl dissolves in water it dissociates into Na and Cl ions. When the ions are hydrated, the water molecules will be oriented so that the negative end of the water dipole interacts with the positive sodiu ...
21 More About Amines • Heterocyclic Compounds
... Notice that amine inversion takes place through a transition state in which the sp 3 nitrogen becomes an sp 2 nitrogen. The three groups bonded to the sp 2 nitrogen are coplanar in the transition state with bond angles of 120°, and the lone pair is in a p orbital. The “inverted” and “non-inverted” a ...
... Notice that amine inversion takes place through a transition state in which the sp 3 nitrogen becomes an sp 2 nitrogen. The three groups bonded to the sp 2 nitrogen are coplanar in the transition state with bond angles of 120°, and the lone pair is in a p orbital. The “inverted” and “non-inverted” a ...