states of Matter
... universe. Scientists and engineers have been tackling the possibility of fusion on earth to produce electricity for over 60 years. Fusion reactions involve relatively small nuclei, such as hydrogen or helium, that have sufficient kinetic energy that when they slam together, they overcome the mutual ...
... universe. Scientists and engineers have been tackling the possibility of fusion on earth to produce electricity for over 60 years. Fusion reactions involve relatively small nuclei, such as hydrogen or helium, that have sufficient kinetic energy that when they slam together, they overcome the mutual ...
Nuclear Fission and Fusion
... An atom of uranium or plutonium receives a neutron. The neutron causes it to be unstable and undergo nuclear fission. The large atom will split into 2 smaller atoms, gamma rays, kinetic energy and free neutrons. ...
... An atom of uranium or plutonium receives a neutron. The neutron causes it to be unstable and undergo nuclear fission. The large atom will split into 2 smaller atoms, gamma rays, kinetic energy and free neutrons. ...
have shown no evidence
... • Stability is favoured by even numbers of protons and neutrons • Not usually equal numbers • Plotting neutron number (A) against proton number (Z) for all known nuclei, shows area of stability • For very light elements N ≈ Z gives stable elements • 1:1 up to 4020Ca • Ratio gradually rises (A>Z) unt ...
... • Stability is favoured by even numbers of protons and neutrons • Not usually equal numbers • Plotting neutron number (A) against proton number (Z) for all known nuclei, shows area of stability • For very light elements N ≈ Z gives stable elements • 1:1 up to 4020Ca • Ratio gradually rises (A>Z) unt ...
Structure of the nucleus • It is now known that the nucleus consists of
... Diagram of a magnetic confinement fusion reactor. The plasma is confined and stopped from touching the walls of the reactor by the huge magnetic fields generated by the complex magnet system. ...
... Diagram of a magnetic confinement fusion reactor. The plasma is confined and stopped from touching the walls of the reactor by the huge magnetic fields generated by the complex magnet system. ...
Nuclear Chemistry - Xavier High School
... • Nuclear reactions involve the nucleus • The nucleus opens, and protons and neutrons are rearranged • The opening of the nucleus releases a tremendous amount of energy that holds the nucleus together – called binding energy • “Normal” Chemical Reactions involve electrons, not protons and neutrons ...
... • Nuclear reactions involve the nucleus • The nucleus opens, and protons and neutrons are rearranged • The opening of the nucleus releases a tremendous amount of energy that holds the nucleus together – called binding energy • “Normal” Chemical Reactions involve electrons, not protons and neutrons ...
Biology Fall Semester Test 1 Study Guide
... Cellular respiration and photosynthesis are responsible for recycling which two nutrients? A well-tested explanation that unifies a broad range of observations is a(an) The process by which organisms keep their internal conditions fairly constant is called In the metric system, the basic unit of len ...
... Cellular respiration and photosynthesis are responsible for recycling which two nutrients? A well-tested explanation that unifies a broad range of observations is a(an) The process by which organisms keep their internal conditions fairly constant is called In the metric system, the basic unit of len ...
Chapter 21 Powerpoint: Nuclear Chemistry
... 3. Identify the type of particle that has decayed and write it after the arrow. 4. Balance the mass number (top) and the atomic number (bottom). 5. Identify the new element. ...
... 3. Identify the type of particle that has decayed and write it after the arrow. 4. Balance the mass number (top) and the atomic number (bottom). 5. Identify the new element. ...
Nuclear Chemistry - Northwest ISD Moodle
... Subtract 2 from atomic number Identify new element from atomic number Add alpha particle ...
... Subtract 2 from atomic number Identify new element from atomic number Add alpha particle ...
Lawson criterion / plasma physics
... Only the He atoms are confined (neutrons escape the magnetic field) and therefore only 20% of the total fusion power is available for plasma heating ...
... Only the He atoms are confined (neutrons escape the magnetic field) and therefore only 20% of the total fusion power is available for plasma heating ...
Nuclear Chemistry
... • Why are some isotopes radioactive and others are not? – The proton : neutron ratio determines whether an isotope is radioactive • Elements with atomic # ≤ 20 prefer a 1 : 1 ratio • Elements with atomic # > 20 prefer a 1 : 1.5 ratio ...
... • Why are some isotopes radioactive and others are not? – The proton : neutron ratio determines whether an isotope is radioactive • Elements with atomic # ≤ 20 prefer a 1 : 1 ratio • Elements with atomic # > 20 prefer a 1 : 1.5 ratio ...
California Chemistry Standards Test
... molecules c. shared electron pairs d. electrostatic attraction between ions What is the purpose of a catalysts a. it permits reactants to start at lower nrg levels b. it lowers the energy barriers for a reaction to occur c. it has strong attraction for anions and cations d. it reacts independently o ...
... molecules c. shared electron pairs d. electrostatic attraction between ions What is the purpose of a catalysts a. it permits reactants to start at lower nrg levels b. it lowers the energy barriers for a reaction to occur c. it has strong attraction for anions and cations d. it reacts independently o ...
Example 27-3 The Binding Energy of 4He
... binding energy of the nucleus divided by the number of nucleons in the nucleus. ...
... binding energy of the nucleus divided by the number of nucleons in the nucleus. ...
Summative Assessment Review!
... • An alpha particle is simply a helium nuclei (He) which is ejected with high energy from an unstable nucleus ...
... • An alpha particle is simply a helium nuclei (He) which is ejected with high energy from an unstable nucleus ...
Chapter 7 - Bakersfield College
... D. Binding energy makes stable heavier nuclei possible (beyond hydrogen) which in turn accounts for the various elements and forms of matter found in the physical universe. 7-9. Nuclear Fission A. In 1939, uranium-235 was discovered to undergo nuclear fission when struck by a neutron. 1. A nucleus o ...
... D. Binding energy makes stable heavier nuclei possible (beyond hydrogen) which in turn accounts for the various elements and forms of matter found in the physical universe. 7-9. Nuclear Fission A. In 1939, uranium-235 was discovered to undergo nuclear fission when struck by a neutron. 1. A nucleus o ...
File - Dr. Wall`s Science
... • Microwaves cook using radiation • X-rays/MRIs/CT scans all use radiation ...
... • Microwaves cook using radiation • X-rays/MRIs/CT scans all use radiation ...
ib atomic and nuclear physics definitions and concepts
... RADIOACTIVE DECAY: A random and spontaneous process in which unstable nuclei emit a particle (disintegrate). The rate decreases exponentially with time. NATURAL RADIOACTIVE DECAY: The emission of an alpha or beta particle. NUCLEAR STRONG FORCE: The force that holds the particles of a nucleus togethe ...
... RADIOACTIVE DECAY: A random and spontaneous process in which unstable nuclei emit a particle (disintegrate). The rate decreases exponentially with time. NATURAL RADIOACTIVE DECAY: The emission of an alpha or beta particle. NUCLEAR STRONG FORCE: The force that holds the particles of a nucleus togethe ...
Fusion or Fission
... keep going is called the critical mass. The trouble with fission reactions is that radiation and nuclear waste products are created in the process. This is a problem, as many nuclear power plants use fission to produce energy, producing a lot of radioactive byproducts as a result. 4 Conversely, in f ...
... keep going is called the critical mass. The trouble with fission reactions is that radiation and nuclear waste products are created in the process. This is a problem, as many nuclear power plants use fission to produce energy, producing a lot of radioactive byproducts as a result. 4 Conversely, in f ...
1. Nucleons Protons and neutrons 2. Nuclide A atom in
... 1. Calculate the nuclear binding energy of a Sulfur– 32 atom. The measured atomic mass of Sulfur–32 is 31.972070 amu. 2. Calculate the nuclear binding energy of a Oxygen-16 atom. The measured atomic mass of Oxygen-16 is 15.994915 amu. 3. Calculate the binding energy per nucleon of a Manganese-55 ato ...
... 1. Calculate the nuclear binding energy of a Sulfur– 32 atom. The measured atomic mass of Sulfur–32 is 31.972070 amu. 2. Calculate the nuclear binding energy of a Oxygen-16 atom. The measured atomic mass of Oxygen-16 is 15.994915 amu. 3. Calculate the binding energy per nucleon of a Manganese-55 ato ...
Fission and Fusion Power Point
... In this example, a stray neutron strikes an atom of U-235. It absorbs the neutron and becomes an unstable atom of U-236. It then undergoes fission and splits into two daughter nuclides. ...
... In this example, a stray neutron strikes an atom of U-235. It absorbs the neutron and becomes an unstable atom of U-236. It then undergoes fission and splits into two daughter nuclides. ...
Activation energy
... • Activation energy is the energy needed to start a reaction and break chemical bonds in the reactants. – This is why a flammable material, like gasoline, does not burn without a spark or flame. – The spark supplies the activation energy to start the reaction. ...
... • Activation energy is the energy needed to start a reaction and break chemical bonds in the reactants. – This is why a flammable material, like gasoline, does not burn without a spark or flame. – The spark supplies the activation energy to start the reaction. ...
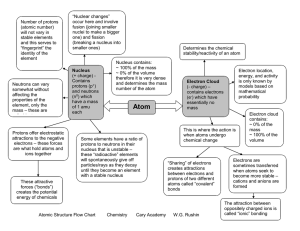
Atomic Structure Flow Chart Chemistry Cary Academy W.G. Rushin
... Protons offer electrostatic attractions to the negative electrons – these forces are what hold atoms and ions together ...
... Protons offer electrostatic attractions to the negative electrons – these forces are what hold atoms and ions together ...
Radioactive Reactions
... • When an atom emits part of its NUCLEUS (protons or neutrons) this is called radiation • This happens because the nucleus is unstable. • When an atom emits protons its identity changes • This can happen naturally (sun) or through man made isotopes in a lab ...
... • When an atom emits part of its NUCLEUS (protons or neutrons) this is called radiation • This happens because the nucleus is unstable. • When an atom emits protons its identity changes • This can happen naturally (sun) or through man made isotopes in a lab ...
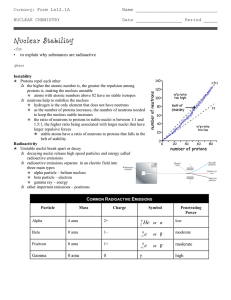
Nuclear Stability
... p as the number of protons increases, the number of neutrons needed to keep the nucleus stable increases p the ratio of neutrons to protons in stable nuclei is between 1:1 and 1.5:1, the higher ratio being associated with larger nuclei that have larger repulsive forces o stable atoms have a ratio of ...
... p as the number of protons increases, the number of neutrons needed to keep the nucleus stable increases p the ratio of neutrons to protons in stable nuclei is between 1:1 and 1.5:1, the higher ratio being associated with larger nuclei that have larger repulsive forces o stable atoms have a ratio of ...
Nuclear fusion

In nuclear physics, nuclear fusion is a nuclear reaction in which two or more atomic nuclei come very close and then collide at a very high speed and join to form a new nucleus. During this process, matter is not conserved because some of the matter of the fusing nuclei is converted to photons (energy). Fusion is the process that powers active or ""main sequence"" stars.The fusion of two nuclei with lower masses than Iron-56 (which, along with Nickel-62, has the largest binding energy per nucleon) generally releases energy, while the fusion of nuclei heavier than iron absorbs energy. The opposite is true for the reverse process, nuclear fission. This means that fusion generally occurs for lighter elements only, and likewise, that fission normally occurs only for heavier elements. There are extreme astrophysical events that can lead to short periods of fusion with heavier nuclei. This is the process that gives rise to nucleosynthesis, the creation of the heavy elements during events such as supernova.Following the discovery of quantum tunneling by Friedrich Hund, in 1929 Robert Atkinson and Fritz Houtermans used the measured masses of light elements to predict that large amounts of energy could be released by fusing small nuclei. Building upon the nuclear transmutation experiments by Ernest Rutherford, carried out several years earlier, the laboratory fusion of hydrogen isotopes was first accomplished by Mark Oliphant in 1932. During the remainder of that decade the steps of the main cycle of nuclear fusion in stars were worked out by Hans Bethe. Research into fusion for military purposes began in the early 1940s as part of the Manhattan Project. Fusion was accomplished in 1951 with the Greenhouse Item nuclear test. Nuclear fusion on a large scale in an explosion was first carried out on November 1, 1952, in the Ivy Mike hydrogen bomb test.Research into developing controlled thermonuclear fusion for civil purposes also began in earnest in the 1950s, and it continues to this day. The present article is about the theory of fusion. For details of the quest for controlled fusion and its history, see the article Fusion power.