Download: Worksheet - New York Science Teacher
... A. Nuclear reactions – the energy released during nuclear reactions is much greater than the energy released during chemical reactions 1. Radioactive decay a.) The stability of an isotope is based on the ratio of neutrons to protons in its nucleus or the binding energy per nucleon. b.) Nuclei that a ...
... A. Nuclear reactions – the energy released during nuclear reactions is much greater than the energy released during chemical reactions 1. Radioactive decay a.) The stability of an isotope is based on the ratio of neutrons to protons in its nucleus or the binding energy per nucleon. b.) Nuclei that a ...
Nuclear Reactions
... This nucleus has 79 protons and 118 neutrons, individually these have a mass of 79x1.0007276u ...
... This nucleus has 79 protons and 118 neutrons, individually these have a mass of 79x1.0007276u ...
Physical Science: Nuclear Chemistry Study Guide
... 28. What is the time required for half a sample of radioactive nuclei to decay called? 29. After three half-lives, what fraction of the original radioactive element remains? 30. To determine the age of fairly recent remains (in the tens of thousands of years as opposed to millions of years), scienti ...
... 28. What is the time required for half a sample of radioactive nuclei to decay called? 29. After three half-lives, what fraction of the original radioactive element remains? 30. To determine the age of fairly recent remains (in the tens of thousands of years as opposed to millions of years), scienti ...
Energy per nucleon
... a black hole (> 3 solar masses) or a neutron star (between 1.4 and 3 solar masses), where the atoms collapse into a single huge nucleus. Lighter stars become white dwarfs . ...
... a black hole (> 3 solar masses) or a neutron star (between 1.4 and 3 solar masses), where the atoms collapse into a single huge nucleus. Lighter stars become white dwarfs . ...
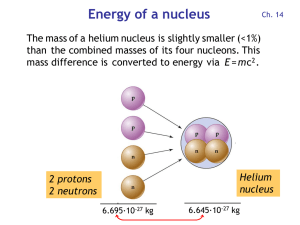
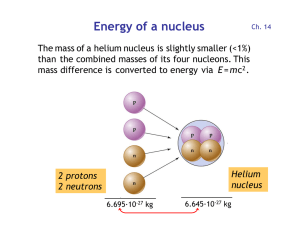
Energy of a nucleus
... a black hole (> 3 solar masses) or a neutron star (between 1.4 and 3 solar masses), where the atoms collapse into a single huge nucleus. Lighter stars become white dwarfs . • All elements heavier than iron/nickel are created during a supernova explosion, which has enough thermal energy to form nucle ...
... a black hole (> 3 solar masses) or a neutron star (between 1.4 and 3 solar masses), where the atoms collapse into a single huge nucleus. Lighter stars become white dwarfs . • All elements heavier than iron/nickel are created during a supernova explosion, which has enough thermal energy to form nucle ...
Fission vs Fusion Worksheet
... There are two main types of nuclear weapons: atomic bombs, which are powered by fission reactions similar to those in nuclear reactors [power plants], and hydrogen bombs, which derive their explosive power from fusion reactions. An atomic bomb slams together two pieces of fissionable material, usual ...
... There are two main types of nuclear weapons: atomic bombs, which are powered by fission reactions similar to those in nuclear reactors [power plants], and hydrogen bombs, which derive their explosive power from fusion reactions. An atomic bomb slams together two pieces of fissionable material, usual ...
Lecture 2: Basics / Lawson
... The force on an individual particle due to the electro-magnetic field (s is species index) ...
... The force on an individual particle due to the electro-magnetic field (s is species index) ...
Glossary of Key Terms in Chapter Two
... beta particle (9.1) an electron formed in the nucleus by the conversion of a neutron into a proton. binding energy (9.3) the energy required to break down the nucleus into its component parts. breeder reactor (9.4) a nuclear reactor that produces its own fuel in the process of providing electrical e ...
... beta particle (9.1) an electron formed in the nucleus by the conversion of a neutron into a proton. binding energy (9.3) the energy required to break down the nucleus into its component parts. breeder reactor (9.4) a nuclear reactor that produces its own fuel in the process of providing electrical e ...
TOPICS OBJECTIVES RESOURCES Temperature • Explain the
... Describe the way in which atoms make up matter. Describe the scientific contributions to the atomic theory. Describe the current model of the atom. State the location, charge, and relative mass of protons, neutrons, and electrons in an atom. Identify the nucleus and the electron cloud in a model of ...
... Describe the way in which atoms make up matter. Describe the scientific contributions to the atomic theory. Describe the current model of the atom. State the location, charge, and relative mass of protons, neutrons, and electrons in an atom. Identify the nucleus and the electron cloud in a model of ...
Chemistry 1 CP Concept 4 Nuclear Chemistry Study Guide
... 11. What device uses controlled nuclear fission to produce new radioactive substances and energy? _______________________________________ 12. Among atoms with low atomic numbers, what is the neutron-proton ratio of the most stable nuclei? ___________________ 13. What is a magic number? _____________ ...
... 11. What device uses controlled nuclear fission to produce new radioactive substances and energy? _______________________________________ 12. Among atoms with low atomic numbers, what is the neutron-proton ratio of the most stable nuclei? ___________________ 13. What is a magic number? _____________ ...
13.4 The nucleus 3 - Nuclear fission and nuclear fusion
... together two nuclei to create a new nucleus. The new nucleus will be an isotope of a new, heavier element. Nuclear fusion produces even more energy than nuclear fission. Nuclear fusion is the process that fuels the stars, including the sun (see Module 11.2). A series of nuclear fusion reactions resu ...
... together two nuclei to create a new nucleus. The new nucleus will be an isotope of a new, heavier element. Nuclear fusion produces even more energy than nuclear fission. Nuclear fusion is the process that fuels the stars, including the sun (see Module 11.2). A series of nuclear fusion reactions resu ...
Topic Review: Nuclear Chemistry 1. The stability of an isotope
... Remember the atomic mass and charge must add up on each side of the equation 7. Energy from fission and fusion comes from the very small fraction of mass that is lost – the reaction converts matter into energy. Einstein’s E=mc2 describes the relationship between energy and matter. 8. The energy ...
... Remember the atomic mass and charge must add up on each side of the equation 7. Energy from fission and fusion comes from the very small fraction of mass that is lost – the reaction converts matter into energy. Einstein’s E=mc2 describes the relationship between energy and matter. 8. The energy ...
1.6--NOTES--Detecting Radiation Nuclear Rxtns
... https://www.youtube.com/watch?v=t7FvxN_gkt4 Nuclear energy basics, issues ...
... https://www.youtube.com/watch?v=t7FvxN_gkt4 Nuclear energy basics, issues ...
Document
... must contain the same number of protons. They may contain varying numbers of neutrons. Isotopes of an element have the same Z but differing N and A values. Example: 11 12 13 14 ...
... must contain the same number of protons. They may contain varying numbers of neutrons. Isotopes of an element have the same Z but differing N and A values. Example: 11 12 13 14 ...
Nuclear Fission and Fusion Notes
... and p attract other n and p. *n have no charge and they do not repel each other or the protons. *p repel each other with the electric force and attract each other with the strong nuclear force. ...
... and p attract other n and p. *n have no charge and they do not repel each other or the protons. *p repel each other with the electric force and attract each other with the strong nuclear force. ...
Stoichiometry Introduction
... Fusion Reactions • 2 small nuclei release energy when they are fused together to form a single, larger nucleus • The process releases energy ...
... Fusion Reactions • 2 small nuclei release energy when they are fused together to form a single, larger nucleus • The process releases energy ...
Atomic Structure and Radioactivity
... - decay: the nucleus composition does not change. -decay occurs in large, unstable, nuclei. The heaviest stable isotope is 20983Bi. -decay transforms a neutron into a proton (n p+ + e ) Electron capture: p+ + e n and p+ n + e+ ...
... - decay: the nucleus composition does not change. -decay occurs in large, unstable, nuclei. The heaviest stable isotope is 20983Bi. -decay transforms a neutron into a proton (n p+ + e ) Electron capture: p+ + e n and p+ n + e+ ...
4.1 The Concepts of Force and Mass
... usually by beta emission, but they are stable against particle emission. Where are the limits of the driplines? How do the properties of these nuclei differ from those of ordinary nuclei? ...
... usually by beta emission, but they are stable against particle emission. Where are the limits of the driplines? How do the properties of these nuclei differ from those of ordinary nuclei? ...
physics - Keith E. Holbert
... A=N+Z: atomic mass number (the number of nucleons). Definitions and Distinctions • atomic mass number (A) [integer number] vs. atomic weight or atomic mass (M) [real value]: M≅A • isotope: nuclides with equal number of protons, but different numbers of neutrons • isotone: nuclides with equal number ...
... A=N+Z: atomic mass number (the number of nucleons). Definitions and Distinctions • atomic mass number (A) [integer number] vs. atomic weight or atomic mass (M) [real value]: M≅A • isotope: nuclides with equal number of protons, but different numbers of neutrons • isotone: nuclides with equal number ...
Lecture 1: The basics - University of Warwick
... or in the plasma state) Are based on a mixture of Deuterium and Tritium Both are related to the ...
... or in the plasma state) Are based on a mixture of Deuterium and Tritium Both are related to the ...
1 slide per page() - Wayne State University Physics and Astronomy
... Energy in a Fission Process Binding energy for heavy nuclei is about 7.2 MeV per nucleon Binding energy for intermediate nuclei is about 8.2 MeV per nucleon Therefore, the fission fragments have less mass than the nucleons in the original nuclei This decrease in mass per nucleon appears as ...
... Energy in a Fission Process Binding energy for heavy nuclei is about 7.2 MeV per nucleon Binding energy for intermediate nuclei is about 8.2 MeV per nucleon Therefore, the fission fragments have less mass than the nucleons in the original nuclei This decrease in mass per nucleon appears as ...
ppt
... Energy in a Fission Process Binding energy for heavy nuclei is about 7.2 MeV per nucleon Binding energy for intermediate nuclei is about 8.2 MeV per nucleon Therefore, the fission fragments have less mass than the nucleons in the original nuclei This decrease in mass per nucleon appears as ...
... Energy in a Fission Process Binding energy for heavy nuclei is about 7.2 MeV per nucleon Binding energy for intermediate nuclei is about 8.2 MeV per nucleon Therefore, the fission fragments have less mass than the nucleons in the original nuclei This decrease in mass per nucleon appears as ...
4 slides per page() - Wayne State University Physics and
... Neutrons are emitted when 235U undergoes fission These neutrons are then available to trigger fission in other nuclei nuclei This process is called a chain reaction If uncontrolled, a violent explosion can occur The principle behind the nuclear bomb, where 1 g of U can release release energy equ ...
... Neutrons are emitted when 235U undergoes fission These neutrons are then available to trigger fission in other nuclei nuclei This process is called a chain reaction If uncontrolled, a violent explosion can occur The principle behind the nuclear bomb, where 1 g of U can release release energy equ ...
Nuclear fusion

In nuclear physics, nuclear fusion is a nuclear reaction in which two or more atomic nuclei come very close and then collide at a very high speed and join to form a new nucleus. During this process, matter is not conserved because some of the matter of the fusing nuclei is converted to photons (energy). Fusion is the process that powers active or ""main sequence"" stars.The fusion of two nuclei with lower masses than Iron-56 (which, along with Nickel-62, has the largest binding energy per nucleon) generally releases energy, while the fusion of nuclei heavier than iron absorbs energy. The opposite is true for the reverse process, nuclear fission. This means that fusion generally occurs for lighter elements only, and likewise, that fission normally occurs only for heavier elements. There are extreme astrophysical events that can lead to short periods of fusion with heavier nuclei. This is the process that gives rise to nucleosynthesis, the creation of the heavy elements during events such as supernova.Following the discovery of quantum tunneling by Friedrich Hund, in 1929 Robert Atkinson and Fritz Houtermans used the measured masses of light elements to predict that large amounts of energy could be released by fusing small nuclei. Building upon the nuclear transmutation experiments by Ernest Rutherford, carried out several years earlier, the laboratory fusion of hydrogen isotopes was first accomplished by Mark Oliphant in 1932. During the remainder of that decade the steps of the main cycle of nuclear fusion in stars were worked out by Hans Bethe. Research into fusion for military purposes began in the early 1940s as part of the Manhattan Project. Fusion was accomplished in 1951 with the Greenhouse Item nuclear test. Nuclear fusion on a large scale in an explosion was first carried out on November 1, 1952, in the Ivy Mike hydrogen bomb test.Research into developing controlled thermonuclear fusion for civil purposes also began in earnest in the 1950s, and it continues to this day. The present article is about the theory of fusion. For details of the quest for controlled fusion and its history, see the article Fusion power.