Document
... a manner similar to a high-resolution in vitro NMR titration experiment. Hence, in vivo resonance signals of amino acids that define the binding interface of the NMR-visible component display characteristic chemical shift changes upon increasing amounts of the additionally expressed, unlabeled ligan ...
... a manner similar to a high-resolution in vitro NMR titration experiment. Hence, in vivo resonance signals of amino acids that define the binding interface of the NMR-visible component display characteristic chemical shift changes upon increasing amounts of the additionally expressed, unlabeled ligan ...
Organometallics II
... CHCOCH3 2 CH3MgBr + (CH3)2CHCOCH 1. diethyl ether 2. H3O+ OH (CH3)2CHCCH CHCCH3 CH3 ...
... CHCOCH3 2 CH3MgBr + (CH3)2CHCOCH 1. diethyl ether 2. H3O+ OH (CH3)2CHCCH CHCCH3 CH3 ...
Fulltext PDF
... tRNA gene but is added later as a post-transcriptional modification. It was not known what is present in place of thymidine in TC loop and which adenine was converted to 1-methyl adenine in M. smegmatis tRNAs. 1-methyl adenine is generally found in tRNA from eukaryotes but not in prokaryotes. These ...
... tRNA gene but is added later as a post-transcriptional modification. It was not known what is present in place of thymidine in TC loop and which adenine was converted to 1-methyl adenine in M. smegmatis tRNAs. 1-methyl adenine is generally found in tRNA from eukaryotes but not in prokaryotes. These ...
Boron and Metal Catalyzed CC and CH Bond Formation
... Dr. Kabalka. I would also like to acknowledge Dr. Min-Liang Yao. His insight and advice greatly helped me learn organic chemistry, on paper and in practice. I would also like to thank my committee members and instructors: Dr. John Bartmess, Dr. David Jenkins, and Dr. Kim Gwinn. In particular, specia ...
... Dr. Kabalka. I would also like to acknowledge Dr. Min-Liang Yao. His insight and advice greatly helped me learn organic chemistry, on paper and in practice. I would also like to thank my committee members and instructors: Dr. John Bartmess, Dr. David Jenkins, and Dr. Kim Gwinn. In particular, specia ...
4-Dimethylaminopyridine - Sigma
... DMAP has been used in natural-product chemistry for the acetylation of a tertiary hydroxyl group of an amino sugar,9 and for acylations of terpenes4,10 and acetylenes11,12 containing secondary and tertiary hydroxyl groups. Connors and Albert recommend using acetic anhydride/DMAP as a reagent for the ...
... DMAP has been used in natural-product chemistry for the acetylation of a tertiary hydroxyl group of an amino sugar,9 and for acylations of terpenes4,10 and acetylenes11,12 containing secondary and tertiary hydroxyl groups. Connors and Albert recommend using acetic anhydride/DMAP as a reagent for the ...
Microenvironment analysis and identification of magnesium binding
... Poisson Boltzmann (NLPB) equation provided further evidence for the locations of site-bound ions. We con®rmed the locations of experimentally determined sites and further differentiated between classes of ion binding. We also identi®ed potentially important, high scoring sites in the group I intron ...
... Poisson Boltzmann (NLPB) equation provided further evidence for the locations of site-bound ions. We con®rmed the locations of experimentally determined sites and further differentiated between classes of ion binding. We also identi®ed potentially important, high scoring sites in the group I intron ...
Rabbit Reticulocyte Lysate Technical Manual
... mRNA and thus reduce background translation to a minimum (1). The lysate contains the cellular components necessary for protein synthesis (tRNA, ribosomes, amino acids, initiation, elongation and termination factors). Rabbit Reticulocyte Lysate is optimized further for mRNA translation by the additi ...
... mRNA and thus reduce background translation to a minimum (1). The lysate contains the cellular components necessary for protein synthesis (tRNA, ribosomes, amino acids, initiation, elongation and termination factors). Rabbit Reticulocyte Lysate is optimized further for mRNA translation by the additi ...
Enol esters: Versatile substrates in synthesis of fine and specialty
... 2. Asymmetric hydroformylation of enol esters ................................................................................. 6 2.2. AHF/ Wittig tandem process involving enol esters ................................................................. 7 3. Asymmetric epoxidation of (Z)-enol esters ... ...
... 2. Asymmetric hydroformylation of enol esters ................................................................................. 6 2.2. AHF/ Wittig tandem process involving enol esters ................................................................. 7 3. Asymmetric epoxidation of (Z)-enol esters ... ...
APPROACHES TO CARBOHYDRATE-BASED CHEMICAL LIBRARIES: THE
... drug design is very simple. If one can define the three-dimensional structure of a target molecule of known biological importance, then it should be possible to "design" compounds which can interact with the target in a very specific manner, thereby producing a high-potency drug with minimal side ef ...
... drug design is very simple. If one can define the three-dimensional structure of a target molecule of known biological importance, then it should be possible to "design" compounds which can interact with the target in a very specific manner, thereby producing a high-potency drug with minimal side ef ...
SQA CfE Higher Chemistry Unit 2
... • an ester can be named given the names of the parent carboxylic acid and alcohol or from structural formulae; • structural formulae for esters can be drawn given the names of the parent alcohol and carboxylic acid or the names of esters; • esters have characteristic smells and are used as flavourin ...
... • an ester can be named given the names of the parent carboxylic acid and alcohol or from structural formulae; • structural formulae for esters can be drawn given the names of the parent alcohol and carboxylic acid or the names of esters; • esters have characteristic smells and are used as flavourin ...
Microsoft Word
... around the cap-binding pocket. Based on the crystallographic and NMR data the authors suggested that intrinsic and specific conformational flexibility of the S. mansoni eIF4E plays a crucial role in the TMG-cap binding, analogous to an “induced fit” mechanism. On the contrary, combined mutagenesis s ...
... around the cap-binding pocket. Based on the crystallographic and NMR data the authors suggested that intrinsic and specific conformational flexibility of the S. mansoni eIF4E plays a crucial role in the TMG-cap binding, analogous to an “induced fit” mechanism. On the contrary, combined mutagenesis s ...
Unit-7-Carboxylic-Acids-Phenols
... which play important roles in biochemistry. These include the carboxylic acids, phenols, amines and amides. We also look at a new type of stereoisomer that figures predominantly in biological chemistry; the optical isomer. ...
... which play important roles in biochemistry. These include the carboxylic acids, phenols, amines and amides. We also look at a new type of stereoisomer that figures predominantly in biological chemistry; the optical isomer. ...
Latest Publication (still not complete)
... cyclization processes, concentrating on the synthesis of three- to seven-membered ring containing products. However, in addition to this, the structure and bonding of carbene complexes is also discussed. In particular, an analysis of information gained through computational analysis is provided. Suc ...
... cyclization processes, concentrating on the synthesis of three- to seven-membered ring containing products. However, in addition to this, the structure and bonding of carbene complexes is also discussed. In particular, an analysis of information gained through computational analysis is provided. Suc ...
The Application of Hydrolytic Enzymes for Biotransformations of
... not defined. 5.3.1 Materials ................................................................ Error! Bookmark not defined. 5.3.1.1 Substrates ............................................................. Error! Bookmark not defined. 5.3.1.2 Enzymes ................................................... ...
... not defined. 5.3.1 Materials ................................................................ Error! Bookmark not defined. 5.3.1.1 Substrates ............................................................. Error! Bookmark not defined. 5.3.1.2 Enzymes ................................................... ...
Aldehydes, Ketones and Carboxylic Acids
... present in them. These compounds exist widely in nature and are responsible for the flavour and aroma of many foods. They are also important industrially both as reagents in synthesis and as solvents. ...
... present in them. These compounds exist widely in nature and are responsible for the flavour and aroma of many foods. They are also important industrially both as reagents in synthesis and as solvents. ...
Solving Spectroscopy Problems: Putting it All Together Once you`ve
... 3) Since we still have an oxygen left, it can attach to either the CH or the CH3 singlet. Look at the 1H‐NMR chemical shift and you will see that the CH3 singlet has the higher chemical shift, so it is probably attached to the oxygen. So, attach the CH(CH3)2 on one position on the benzene ring a ...
... 3) Since we still have an oxygen left, it can attach to either the CH or the CH3 singlet. Look at the 1H‐NMR chemical shift and you will see that the CH3 singlet has the higher chemical shift, so it is probably attached to the oxygen. So, attach the CH(CH3)2 on one position on the benzene ring a ...
Modifying the stereochemistry of an enzyme
... here is a growing demand in the chemical and biotechnology industries for reliable and efficient methods for the production of enantiomerically pure compounds. Increasingly, synthetic chemists are exploiting enzymes in the asymmetric and stereoselective synthesis of chiral building blocks (1). Howev ...
... here is a growing demand in the chemical and biotechnology industries for reliable and efficient methods for the production of enantiomerically pure compounds. Increasingly, synthetic chemists are exploiting enzymes in the asymmetric and stereoselective synthesis of chiral building blocks (1). Howev ...
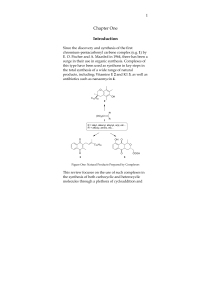
Bottromycin

Bottromycin is a macrocyclic peptide with antibiotic activity. It was first discovered in 1957 as a natural product isolated from Streptomyces bottropensis. It has been shown to inhibit methicillin-resistant Staphylococcus aureus (MRSA) and vancomycin-resistant Enterococci (VRE) among other Gram-positive bacteria and mycoplasma. Bottromycin is structurally distinct from both vancomycin, a glycopeptide antibiotic, and methicillin, a beta-lactam antibiotic.Bottromycin binds to the A site of the ribosome and blocks the binding of aminoacyl-tRNA, therefore inhibiting bacterial protein synthesis. Although bottromycin exhibits antibacterial activity in vitro, it has not yet been developed as a clinical antibiotic, potentially due to its poor stability in blood plasma. To increase its stability in vivo, some bottromycin derivatives have been explored.The structure of bottromycin contains a macrocyclic amidine as well as a thiazole ring. The absolute stereochemistry at several chiral centers has been determined as of 2009. In 2012, a three-dimensional solution structure of bottromycin was published. The solution structure revealed that several methyl groups are on the same face of the structure.Bottromycin falls within the ribosomally synthesized and post-translationally modified peptide class of natural product.