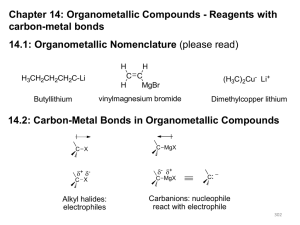
Organometallic Compounds - Reagents
... to a starting compound using known and reliable reactions. “it is a problem solving technique for transforming the structure of a synthetic target molecule (TM) to a sequence of progressively simpler structures along the pathway which ultimately leads to simple or commercially available starting mat ...
... to a starting compound using known and reliable reactions. “it is a problem solving technique for transforming the structure of a synthetic target molecule (TM) to a sequence of progressively simpler structures along the pathway which ultimately leads to simple or commercially available starting mat ...
HMDS+TMCS+Pyridine - Sigma
... Prepare a reagent blank (all components, solvents, etc., except sample), following the same procedure as used for the sample. 1. Weigh 1-10mg of sample into a 5mL reaction vessel. If appropriate, dissolve sample in solvent (see below). If sample is in aqueous solution, evaporate to dryness, then use ...
... Prepare a reagent blank (all components, solvents, etc., except sample), following the same procedure as used for the sample. 1. Weigh 1-10mg of sample into a 5mL reaction vessel. If appropriate, dissolve sample in solvent (see below). If sample is in aqueous solution, evaporate to dryness, then use ...
hydroxy- and oxoacids. heterofunctional compounds of benzene
... – to the left. Letters D and L are used before the name of substance designation of a configuration and a direction of rotation of a plane of polarization. Also before name of the substance must be written signs (+) and (-) corresponding to the rotation to the right or to the left. Mixture of identi ...
... – to the left. Letters D and L are used before the name of substance designation of a configuration and a direction of rotation of a plane of polarization. Also before name of the substance must be written signs (+) and (-) corresponding to the rotation to the right or to the left. Mixture of identi ...
Development of Catalytic Ester Condensations and Hydrolysis of
... washing the crude product with a small amount of water. However, sulfonic acids are easily deactivated by water since they have high hydrophlicity. Therefore, the author first considered that the solvent effect might play a key role in the high reactivity of ester condensation reactions catalyzed by ...
... washing the crude product with a small amount of water. However, sulfonic acids are easily deactivated by water since they have high hydrophlicity. Therefore, the author first considered that the solvent effect might play a key role in the high reactivity of ester condensation reactions catalyzed by ...
Enzyme Substrate Interactions Identification of Enzyme Catalytic Site
... • Enzyme concentration: – At saturating substrate concentration, the initial velocity is directly related to the enzyme concentration. E + S = ES = E + P. Thus, as long as S is not limiting, more E leads to more ES. ...
... • Enzyme concentration: – At saturating substrate concentration, the initial velocity is directly related to the enzyme concentration. E + S = ES = E + P. Thus, as long as S is not limiting, more E leads to more ES. ...
Educational Power Point on MRSA
... the dangerous forms of staph found in hospitals and nursing homes. "Until recently we rarely thought of it as a problem among healthy people in the community," said Dr. Rachel Gorwitz of the federal Centers for Disease Control and Prevention. Now, the germs causing outbreaks in schools, on sports te ...
... the dangerous forms of staph found in hospitals and nursing homes. "Until recently we rarely thought of it as a problem among healthy people in the community," said Dr. Rachel Gorwitz of the federal Centers for Disease Control and Prevention. Now, the germs causing outbreaks in schools, on sports te ...
Enzymatic Activities for the Synthesis of Chlorophyll in Pigment
... to the white sectors, almost no formation of PBG or of porphyrin was observed, whereas the green sectors formed significant amounts of PBG (data not shown). Therefore, we suspected that the blockage or the inactivation of PBG synthase was responsible for the pigment deficiency in the white sectors. ...
... to the white sectors, almost no formation of PBG or of porphyrin was observed, whereas the green sectors formed significant amounts of PBG (data not shown). Therefore, we suspected that the blockage or the inactivation of PBG synthase was responsible for the pigment deficiency in the white sectors. ...
the beginnings of synthetic organic chemistry: zinc alkyls and the
... historic, this being the first synthesis of a tertiary alcohol, and ironic. The existence of tertiary alcohols had been predicted by Kolbe (13), the most adamant opponent of the new structural theory, and this first synthesis was accomplished by Butlerov, its most ardent champion. This reaction, whi ...
... historic, this being the first synthesis of a tertiary alcohol, and ironic. The existence of tertiary alcohols had been predicted by Kolbe (13), the most adamant opponent of the new structural theory, and this first synthesis was accomplished by Butlerov, its most ardent champion. This reaction, whi ...
Pediatrician Perceptions of an Outpatient Antimicrobial Stewardship
... (1) "I did not read my audit reports because honestly, I didn't really care. To me, it was just another piece of paper. It didn't impress me at all. They [the study team! gave you a number describing your behavior but they had no deeper understanding of what was really going on." [El] (2) "I questio ...
... (1) "I did not read my audit reports because honestly, I didn't really care. To me, it was just another piece of paper. It didn't impress me at all. They [the study team! gave you a number describing your behavior but they had no deeper understanding of what was really going on." [El] (2) "I questio ...
Fragrance: The Most Chemically Complex Additive to Cosmetic
... to the formulation. The composition of a fragrance can reach well over one hundred compounds, which include essential oils and their isolates as well as other synthetically derived aroma chemicals. Although fragrances are designed for a wide variety of applications, they are not indefinitely stable. ...
... to the formulation. The composition of a fragrance can reach well over one hundred compounds, which include essential oils and their isolates as well as other synthetically derived aroma chemicals. Although fragrances are designed for a wide variety of applications, they are not indefinitely stable. ...
Use of a Sec signal peptide library from Bacillus subtilis for the
... © The Author(s) 2016. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the ori ...
... © The Author(s) 2016. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the ori ...
Immobilization and stability studies of a lipase from
... effect, while ethanol, methanol and propanol inhibited the enzyme activity. The effect of organic solvents might be attributed to the water content in reaction mix. Water plays a significant role in the activity of the enzymes (Klibanov, 1989). The inhibition of lipase in presence of ethanol, methan ...
... effect, while ethanol, methanol and propanol inhibited the enzyme activity. The effect of organic solvents might be attributed to the water content in reaction mix. Water plays a significant role in the activity of the enzymes (Klibanov, 1989). The inhibition of lipase in presence of ethanol, methan ...
Carboxylic acids, esters, and other acid derivatives
... • Because they don’t possess OH groups, esters cannot form Hbonds with other ester molecules. As a result, esters have lower boiling points than carboxylic acids and alcohols that have approximately the same molar mass. • They do have a C=O bond (polar) so their boiling points are between alcohols a ...
... • Because they don’t possess OH groups, esters cannot form Hbonds with other ester molecules. As a result, esters have lower boiling points than carboxylic acids and alcohols that have approximately the same molar mass. • They do have a C=O bond (polar) so their boiling points are between alcohols a ...
Hydrogen exchange mass spectrometry for the analysis of protein
... with more classical functional data, a more thorough understanding of protein function can be obtained. In many cases, protein dynamics are directly related to specific protein functions such as conformational changes during enzyme activation or protein movements during binding. The method is made p ...
... with more classical functional data, a more thorough understanding of protein function can be obtained. In many cases, protein dynamics are directly related to specific protein functions such as conformational changes during enzyme activation or protein movements during binding. The method is made p ...
Organic Chemistry
... Crossed Enolate Reactions using LDA • Equilibrium among enolate anions is established when the ketone is in slight excess, a condition under which it is possible for proton-transfer reactions to occur between an enolate and an a-hydrogen of an unreacted ketone. Thus, equilibrium is established betw ...
... Crossed Enolate Reactions using LDA • Equilibrium among enolate anions is established when the ketone is in slight excess, a condition under which it is possible for proton-transfer reactions to occur between an enolate and an a-hydrogen of an unreacted ketone. Thus, equilibrium is established betw ...
Bottromycin

Bottromycin is a macrocyclic peptide with antibiotic activity. It was first discovered in 1957 as a natural product isolated from Streptomyces bottropensis. It has been shown to inhibit methicillin-resistant Staphylococcus aureus (MRSA) and vancomycin-resistant Enterococci (VRE) among other Gram-positive bacteria and mycoplasma. Bottromycin is structurally distinct from both vancomycin, a glycopeptide antibiotic, and methicillin, a beta-lactam antibiotic.Bottromycin binds to the A site of the ribosome and blocks the binding of aminoacyl-tRNA, therefore inhibiting bacterial protein synthesis. Although bottromycin exhibits antibacterial activity in vitro, it has not yet been developed as a clinical antibiotic, potentially due to its poor stability in blood plasma. To increase its stability in vivo, some bottromycin derivatives have been explored.The structure of bottromycin contains a macrocyclic amidine as well as a thiazole ring. The absolute stereochemistry at several chiral centers has been determined as of 2009. In 2012, a three-dimensional solution structure of bottromycin was published. The solution structure revealed that several methyl groups are on the same face of the structure.Bottromycin falls within the ribosomally synthesized and post-translationally modified peptide class of natural product.