The therapeutic implications of ketone bodies
... particular relevance to brain metabolism, where Cahill and his colleagues have established that over 60% of the metabolic energy needs of brain can be supplied by ketone bodies, rather than by glucose [11]. They have further established that mild ketosis with blood levels of 5–7 mM is the normal phy ...
... particular relevance to brain metabolism, where Cahill and his colleagues have established that over 60% of the metabolic energy needs of brain can be supplied by ketone bodies, rather than by glucose [11]. They have further established that mild ketosis with blood levels of 5–7 mM is the normal phy ...
2-Phospho
... Comparing Fermentation with Anaerobic and Aerobic Respiration • All use glycolysis (net ATP = 2) to oxidize glucose and harvest chemical energy of food • In all three, NAD+ is the oxidizing agent that accepts electrons during glycolysis • The processes have different final electron acceptors: an or ...
... Comparing Fermentation with Anaerobic and Aerobic Respiration • All use glycolysis (net ATP = 2) to oxidize glucose and harvest chemical energy of food • In all three, NAD+ is the oxidizing agent that accepts electrons during glycolysis • The processes have different final electron acceptors: an or ...
Effects of biotin on pyruvate carboxylase, acetyl CoA
... Diabetic patients have been shown to have a lower serum biotin concentration than control groups. With biotin treatment, diabetic subjects should have a higher activity? ...
... Diabetic patients have been shown to have a lower serum biotin concentration than control groups. With biotin treatment, diabetic subjects should have a higher activity? ...
Cellular Respiration
... couples electron transport to ATP synthesis •NADH and FADH2 –Donate electrons to the electron transport chain, which powers ATP synthesis via oxidative phosphorylation ...
... couples electron transport to ATP synthesis •NADH and FADH2 –Donate electrons to the electron transport chain, which powers ATP synthesis via oxidative phosphorylation ...
No Slide Title
... Formed by phosphorylation of F6-P, catalyzed by PFK-2 Broken down by FBPase-2 PFK-2 and FBPase-2 are two distinct enzyme activities on 1 protein Balance of the 2 activities in the liver, which determines cellular level of F2,6BP, is regulated by glucagon Glucagon - released by pancreas to signal low ...
... Formed by phosphorylation of F6-P, catalyzed by PFK-2 Broken down by FBPase-2 PFK-2 and FBPase-2 are two distinct enzyme activities on 1 protein Balance of the 2 activities in the liver, which determines cellular level of F2,6BP, is regulated by glucagon Glucagon - released by pancreas to signal low ...
2-Phospho
... Comparing Fermentation with Anaerobic and Aerobic Respiration • All use glycolysis (net ATP = 2) to oxidize glucose and harvest chemical energy of food • In all three, NAD+ is the oxidizing agent that accepts electrons during glycolysis • The processes have different final electron acceptors: an or ...
... Comparing Fermentation with Anaerobic and Aerobic Respiration • All use glycolysis (net ATP = 2) to oxidize glucose and harvest chemical energy of food • In all three, NAD+ is the oxidizing agent that accepts electrons during glycolysis • The processes have different final electron acceptors: an or ...
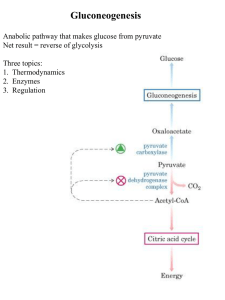
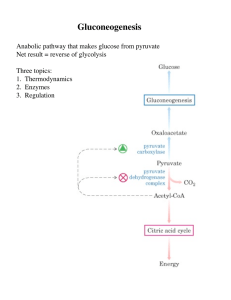
Gluconeogenesis - Creighton Chemistry Webserver
... Formed by phosphorylation of F6-P, catalyzed by PFK-2 Broken down by FBPase-2 PFK-2 and FBPase-2 are two distinct enzyme activities on 1 protein Balance of the 2 activities in the liver, which determines cellular level of F2,6BP, is regulated by glucagon Glucagon - released by pancreas to signal low ...
... Formed by phosphorylation of F6-P, catalyzed by PFK-2 Broken down by FBPase-2 PFK-2 and FBPase-2 are two distinct enzyme activities on 1 protein Balance of the 2 activities in the liver, which determines cellular level of F2,6BP, is regulated by glucagon Glucagon - released by pancreas to signal low ...
chapter 9 cellular respiration: harvesting chemical energy
... In the electron transport chain, the electrons move from molecule to molecule until they combine with molecular oxygen and hydrogen ions to form water. o As the electrons are passed along the chain, the energy released at each step in the chain is stored in a form the mitochondrion (or prokaryotic c ...
... In the electron transport chain, the electrons move from molecule to molecule until they combine with molecular oxygen and hydrogen ions to form water. o As the electrons are passed along the chain, the energy released at each step in the chain is stored in a form the mitochondrion (or prokaryotic c ...
Respiration: Occurs in two places in the cell Cytoplasm and
... The RXNs of Respiration: Electron Transport Chain Chemiosmosis and the proton (H+) motive force Linking e- transport and H+ shuttling to ATP synthesis NADH + H+ ...
... The RXNs of Respiration: Electron Transport Chain Chemiosmosis and the proton (H+) motive force Linking e- transport and H+ shuttling to ATP synthesis NADH + H+ ...
RESPIRATION PPT...Campbell Powerpoint presentation
... Comparing Fermentation with Anaerobic and Aerobic Respiration • All use glycolysis (net ATP = 2) to oxidize glucose and harvest chemical energy of food • In all three, NAD+ is the oxidizing agent that accepts electrons during glycolysis • The processes have different final electron acceptors: an or ...
... Comparing Fermentation with Anaerobic and Aerobic Respiration • All use glycolysis (net ATP = 2) to oxidize glucose and harvest chemical energy of food • In all three, NAD+ is the oxidizing agent that accepts electrons during glycolysis • The processes have different final electron acceptors: an or ...
Energy Metabolism - Georgia Institute of Technology
... Energy Metabolism • ATP synthesis – Outline the steps of glycolysis – Outline the steps of lipolysis – Citric acid cycle/Electron transport chain ...
... Energy Metabolism • ATP synthesis – Outline the steps of glycolysis – Outline the steps of lipolysis – Citric acid cycle/Electron transport chain ...
photosynthesis-and-cellular-respiration-worksheet
... Interactive Question 9.6 a. 2 ATP b. 2 glyceraldehyde-3-phosphate (we did not discuss this G3P at this point of the pathway, but it happens to be the same molecule that we see produced in the calvin cycle) c. 2 NAD+ d. 2 NADH e. 4 ATP (remember a total of 4 ATP are made in glycolysis, but it also re ...
... Interactive Question 9.6 a. 2 ATP b. 2 glyceraldehyde-3-phosphate (we did not discuss this G3P at this point of the pathway, but it happens to be the same molecule that we see produced in the calvin cycle) c. 2 NAD+ d. 2 NADH e. 4 ATP (remember a total of 4 ATP are made in glycolysis, but it also re ...
Adv. Bio. Ch 9 Glyco and Resp
... Glycolysis means “splitting of sugar” glucose (6C) 2 pyruvate (3C) 10 steps of glycolysis each with their own enzyme are broken down into two phases: energy ...
... Glycolysis means “splitting of sugar” glucose (6C) 2 pyruvate (3C) 10 steps of glycolysis each with their own enzyme are broken down into two phases: energy ...
- ISpatula
... The Insulin Receptor • After insulin has entered the circulation, it diffuses into tissues, where it is bound by specialized receptors that are found on the membranes of most tissues. • The biologic responses promoted by these insulin-receptor complexes have been identified in the primary target ti ...
... The Insulin Receptor • After insulin has entered the circulation, it diffuses into tissues, where it is bound by specialized receptors that are found on the membranes of most tissues. • The biologic responses promoted by these insulin-receptor complexes have been identified in the primary target ti ...
Ch_9 - Bartlett High School
... How is the ATP made? How do electrons get from glucose to O2? How does pyruvate get into the mitochondria for the Krebs Cycle? What happens during the Citric Acid Cycle? How many ATP so far? How many electron carriers so far? What happens during electron transport? Why do electrons NEED to “break th ...
... How is the ATP made? How do electrons get from glucose to O2? How does pyruvate get into the mitochondria for the Krebs Cycle? What happens during the Citric Acid Cycle? How many ATP so far? How many electron carriers so far? What happens during electron transport? Why do electrons NEED to “break th ...
Ch 9 Cellular respiration
... Fermentation generates ATP by substrate level phosphorylation as long as there is enough NAD+ (get enough because in anaerobic conditions,electrons are transferred from NADH to pyruvate) pyruvate is electron acceptor for oxidizing NADH back to NAD+ and can then be reused in glycolysis ...
... Fermentation generates ATP by substrate level phosphorylation as long as there is enough NAD+ (get enough because in anaerobic conditions,electrons are transferred from NADH to pyruvate) pyruvate is electron acceptor for oxidizing NADH back to NAD+ and can then be reused in glycolysis ...
Bio 226: Cell and Molecular Biology
... •Insensitive to Cyanide, Azide or CO •Sensitive to SHAM (salicylhydroxamic acid,) •Also found in fungi, trypanosomes & Plasmodium ...
... •Insensitive to Cyanide, Azide or CO •Sensitive to SHAM (salicylhydroxamic acid,) •Also found in fungi, trypanosomes & Plasmodium ...
Anaerobic Respiration
... • The process of lactic acid fermentation replaces the Krebs Cycle & the ETC so that the cell can have a continual source of energy, even in the absence of oxygen. • However this shift is only temporary and cells need oxygen for sustained activity. ...
... • The process of lactic acid fermentation replaces the Krebs Cycle & the ETC so that the cell can have a continual source of energy, even in the absence of oxygen. • However this shift is only temporary and cells need oxygen for sustained activity. ...
HONORS BIOLOGY CHAPTER 6 - Hudson City Schools / Homepage
... • Organic molecules with an abundance of C-H bonds are a source of e- with a potential to fall closer and closer to oxygen. • An e- loses its potential when it shifts from a less electronegative atom (doesn’t attract eas much) to a more electronegative atom (attracts e- more). ...
... • Organic molecules with an abundance of C-H bonds are a source of e- with a potential to fall closer and closer to oxygen. • An e- loses its potential when it shifts from a less electronegative atom (doesn’t attract eas much) to a more electronegative atom (attracts e- more). ...
Fatty acid
... water molecule • Enzymes are macromolecules that speed up the dehydration process • Polymers are disassembled to monomers by hydrolysis, a reaction that is essentially the reverse of the dehydration reaction Copyright © 2008 Pearson Education, Inc., publishing as Pearson Benjamin Cummings ...
... water molecule • Enzymes are macromolecules that speed up the dehydration process • Polymers are disassembled to monomers by hydrolysis, a reaction that is essentially the reverse of the dehydration reaction Copyright © 2008 Pearson Education, Inc., publishing as Pearson Benjamin Cummings ...
Lactic acid fermentation
... electrons to – NAD+, forming NADH. However, there is only a limited supply of NAD+ available in a cell. For glycolysis to continue, NADH must be oxidized – that is, have electrons taken away – to regenerate the NAD+. This is usually done through an electron transport chain in a process called oxidat ...
... electrons to – NAD+, forming NADH. However, there is only a limited supply of NAD+ available in a cell. For glycolysis to continue, NADH must be oxidized – that is, have electrons taken away – to regenerate the NAD+. This is usually done through an electron transport chain in a process called oxidat ...
III. 4. Test Respiració cel·lular
... 8) Why does the oxidation of organic compounds by molecular oxygen to produce CO2 and water release free energy? A) The covalent bonds in organic molecules are higher energy bonds than those in water and carbon dioxide. B) Electrons are being moved from atoms that have a lower affinity for electrons ...
... 8) Why does the oxidation of organic compounds by molecular oxygen to produce CO2 and water release free energy? A) The covalent bonds in organic molecules are higher energy bonds than those in water and carbon dioxide. B) Electrons are being moved from atoms that have a lower affinity for electrons ...
Adenylate Energy Charge during Batch Culture of
... Considerable attention is now being given to regulatory control exerted by cyclic AMP in prokaryotes (Rickenberg, 1974). Cyclic AMP levels are involved in the control of tricarboxylic acid cycle enzyme synthesis (Takahashi, I 975) and various membrane-associated phenomena (see Patrick & Dobrogosz, I ...
... Considerable attention is now being given to regulatory control exerted by cyclic AMP in prokaryotes (Rickenberg, 1974). Cyclic AMP levels are involved in the control of tricarboxylic acid cycle enzyme synthesis (Takahashi, I 975) and various membrane-associated phenomena (see Patrick & Dobrogosz, I ...
Slide 1
... other mitochondrial uncoupling proteins) which are activated by calcium coming in through the calcium uniporter. Also note that electron carriers can autooxidize directly to oxygen, creating oxygen radicals (Co-Q is the major site of autooxidation) with as much as 5% of resting oxygen use due to thi ...
... other mitochondrial uncoupling proteins) which are activated by calcium coming in through the calcium uniporter. Also note that electron carriers can autooxidize directly to oxygen, creating oxygen radicals (Co-Q is the major site of autooxidation) with as much as 5% of resting oxygen use due to thi ...
ch 9ppt
... serve as raw materials for building & fuels for energy controlled release of energy series of step-by-step enzyme-controlled reactions ...
... serve as raw materials for building & fuels for energy controlled release of energy series of step-by-step enzyme-controlled reactions ...
Glucose

Glucose is a sugar with the molecular formula C6H12O6. The name ""glucose"" (/ˈɡluːkoʊs/) comes from the Greek word γλευκος, meaning ""sweet wine, must"". The suffix ""-ose"" is a chemical classifier, denoting a carbohydrate. It is also known as dextrose or grape sugar. With 6 carbon atoms, it is classed as a hexose, a sub-category of monosaccharides. α-D-glucose is one of the 16 aldose stereoisomers. The D-isomer (D-glucose) occurs widely in nature, but the L-isomer (L-glucose) does not. Glucose is made during photosynthesis from water and carbon dioxide, using energy from sunlight. The reverse of the photosynthesis reaction, which releases this energy, is a very important source of power for cellular respiration. Glucose is stored as a polymer, in plants as starch and in animals as glycogen.