The instructions for how to create and run a living organism are
... To make proteins—the large, complex molecules necessary for structure and function—a cell must first make a copy of its DNA. The cell does that through transcription, which uses the DNA as a template to produce a corresponding strand of messenger RNA (mRNA). That mRNA then undergoes translation. In ...
... To make proteins—the large, complex molecules necessary for structure and function—a cell must first make a copy of its DNA. The cell does that through transcription, which uses the DNA as a template to produce a corresponding strand of messenger RNA (mRNA). That mRNA then undergoes translation. In ...
HonBio Chapter 3 notes
... organic – composed of carbon bonded to other elements. Functions of organic compounds: Main structural components of cells and tissues Participate in and regulate many chemical reactions Provide energy ...
... organic – composed of carbon bonded to other elements. Functions of organic compounds: Main structural components of cells and tissues Participate in and regulate many chemical reactions Provide energy ...
Announcements - Hiram College
... What does it do? How similar is it to something else? How does it fold? Where does it go in a cell? What does it interact with? How it is regulated? Level of confidence? ...
... What does it do? How similar is it to something else? How does it fold? Where does it go in a cell? What does it interact with? How it is regulated? Level of confidence? ...
Word Doc - Biochemistry
... residues appears to be the lower limit for a functional domain size. Protein sizes range from this lower limit to several hundred residues in multi-functional proteins. Very large aggregates can be formed from protein subunits, for example many thousand actin molecules assemble into a an actin filam ...
... residues appears to be the lower limit for a functional domain size. Protein sizes range from this lower limit to several hundred residues in multi-functional proteins. Very large aggregates can be formed from protein subunits, for example many thousand actin molecules assemble into a an actin filam ...
Protein structure
... in performing its biological function. In the context of these functional rearrangements, these tertiary or quaternary structures are usually referred to as chemical conformation, and transitions between them are called conformational changes. The primary structure is held together by covalent or pe ...
... in performing its biological function. In the context of these functional rearrangements, these tertiary or quaternary structures are usually referred to as chemical conformation, and transitions between them are called conformational changes. The primary structure is held together by covalent or pe ...
Protein Folding Questions only
... functional group. - Hydrophilic sidechains have various combinations of ____________. An exception to this observation is: ...
... functional group. - Hydrophilic sidechains have various combinations of ____________. An exception to this observation is: ...
6. 3-D structure of proteins
... amino acid side chains can stabilize or destabilize this structure. Five different kinds of constraints affect the stability of an α helix: 1. the electrostatic repulsion (or attraction) between successive amino acid residues with charged R group. 2. the bulkiness of adjacent R group 3. the interact ...
... amino acid side chains can stabilize or destabilize this structure. Five different kinds of constraints affect the stability of an α helix: 1. the electrostatic repulsion (or attraction) between successive amino acid residues with charged R group. 2. the bulkiness of adjacent R group 3. the interact ...
Proteins
... fold in such a way that hydrophylic amino acids will be exposed to aqueous environments, such as the cytoplasm, whereas hydrophobic amino acids tend be located away from water, for example, in the hydrophobic core of a protein, or within the bilipid layer of a cell membrane. Hydrophibicity is strong ...
... fold in such a way that hydrophylic amino acids will be exposed to aqueous environments, such as the cytoplasm, whereas hydrophobic amino acids tend be located away from water, for example, in the hydrophobic core of a protein, or within the bilipid layer of a cell membrane. Hydrophibicity is strong ...
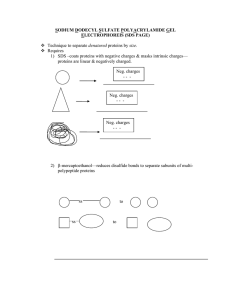
SDS Electrophoresis
... But then separated subunits/polypeptides will be linear & negative due to SDS treatment 3) Heat—to further denature proteins 4) polyacrylamide—gel matrix that acts as size sorter 5) electrophoresis, using electric field with positive anode and negative cathode, all proteins are attracted to bottom ...
... But then separated subunits/polypeptides will be linear & negative due to SDS treatment 3) Heat—to further denature proteins 4) polyacrylamide—gel matrix that acts as size sorter 5) electrophoresis, using electric field with positive anode and negative cathode, all proteins are attracted to bottom ...
Elise Young: Animal & Range Sciences
... Linking common factors in the phenomenon of protein clumping observed in several diseases Proteins perform many important functions at the cellular level. However, if proteins do not fold properly, they are prone to aggregating and sticking together, preventing them from performing their functions, ...
... Linking common factors in the phenomenon of protein clumping observed in several diseases Proteins perform many important functions at the cellular level. However, if proteins do not fold properly, they are prone to aggregating and sticking together, preventing them from performing their functions, ...
Let`s Get Pumped Up about Proteins!!!
... protein research/manufacturing • Typical cell produces ~2000 different proteins • 9 different categories depending on function (Table 2.1 pg. 57) ...
... protein research/manufacturing • Typical cell produces ~2000 different proteins • 9 different categories depending on function (Table 2.1 pg. 57) ...
Review For Final I - NAU jan.ucc.nau.edu web server
... • subtract this sum from the average you want to get • divide the difference by 40 for the percent score you need ...
... • subtract this sum from the average you want to get • divide the difference by 40 for the percent score you need ...
Amino acid side chains stabilise the enzyme shape Hydrogen bonds
... Many amino acids contain groups in the side chains that have a hydrogen atom attached to either an oxygen or nitrogen atom. Hydrogen bonding can occur between such groups. ...
... Many amino acids contain groups in the side chains that have a hydrogen atom attached to either an oxygen or nitrogen atom. Hydrogen bonding can occur between such groups. ...
Introduction to Proteins
... been flopped over 90 degrees to the right for illustration purposes.) Cysteine has an R group that is similar but includes a sulfur atom. Other amino acids have R groups that range from a single hydrogen atom up to a double ring of carbon and nitrogen atoms, as you can see in the next plate. Color t ...
... been flopped over 90 degrees to the right for illustration purposes.) Cysteine has an R group that is similar but includes a sulfur atom. Other amino acids have R groups that range from a single hydrogen atom up to a double ring of carbon and nitrogen atoms, as you can see in the next plate. Color t ...
How scientists are developing ways to help children with muscular
... new treatments for degenerative nerve diseases, such as Huntington’s disease, Alzheimer’s disease and Parkinson’s disease. Misfolded proteins in nerve cells (neurons) are a common factor in all of these diseases. These new compounds improve a cell’s ability to properly “fold” proteins. It activates ...
... new treatments for degenerative nerve diseases, such as Huntington’s disease, Alzheimer’s disease and Parkinson’s disease. Misfolded proteins in nerve cells (neurons) are a common factor in all of these diseases. These new compounds improve a cell’s ability to properly “fold” proteins. It activates ...
Chemistry 160 Protein Structure Homework
... 5. Describe 3 types of interactions that stabilize protein structure. 6. What drives protein folding? 7. Give two ways amino acid sequences are determined. 8. A small protein was cleaved in two separate experiments by chymotrypsin and by trypsin. The chymotrypsin fragments were: MAVKTMPW, ATF, AMERT ...
... 5. Describe 3 types of interactions that stabilize protein structure. 6. What drives protein folding? 7. Give two ways amino acid sequences are determined. 8. A small protein was cleaved in two separate experiments by chymotrypsin and by trypsin. The chymotrypsin fragments were: MAVKTMPW, ATF, AMERT ...
Intro to Nutrients
... All of the macronutrients start as a small unit – a monomer – which will link up with other monomers to form longer chains called ...
... All of the macronutrients start as a small unit – a monomer – which will link up with other monomers to form longer chains called ...
Determination of Proteins
... •Generally proteins are soluble in water, except the membrane proteins which are hydrophobic •Absorption maxima in the ultraviolet region •Proteins are charged molecules, but the charge depend on the pH of the buffer. •Move under an electric field and can be separated by ...
... •Generally proteins are soluble in water, except the membrane proteins which are hydrophobic •Absorption maxima in the ultraviolet region •Proteins are charged molecules, but the charge depend on the pH of the buffer. •Move under an electric field and can be separated by ...
Spectroscopy of Proteins
... • Exert all the biological functions of the organism: enzymes, antibodies, cytoskeletons, hormones, receptors ...
... • Exert all the biological functions of the organism: enzymes, antibodies, cytoskeletons, hormones, receptors ...
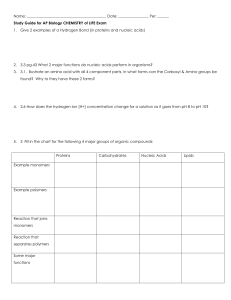
Name: Date: Per: ______ Study Guide for AP Biology CHEMISTRY
... 1. Give 2 examples of a Hydrogen Bond (in proteins and nucleic acids) ...
... 1. Give 2 examples of a Hydrogen Bond (in proteins and nucleic acids) ...
Biochemistry
... What makes Carbon Special? Why is Carbon so different from all the other elements on the periodic table? The answer derives from the ability of Carbon atoms to bond together to form long chains and rings. ...
... What makes Carbon Special? Why is Carbon so different from all the other elements on the periodic table? The answer derives from the ability of Carbon atoms to bond together to form long chains and rings. ...
Making Proteins
... 2. mRNA codons are matched to corresponding tRNA anticodons and appropriate amino acids are strung together. 3. Dehydration synthesis occurs between the amino acids, and they join, making a protein chain with peptide bonds in between 4. Ribosomes detach when they come across a “stop” codon (UAA, UAG ...
... 2. mRNA codons are matched to corresponding tRNA anticodons and appropriate amino acids are strung together. 3. Dehydration synthesis occurs between the amino acids, and they join, making a protein chain with peptide bonds in between 4. Ribosomes detach when they come across a “stop” codon (UAA, UAG ...
Structural Studies of Sgt2, a Component of the GET Pathway that
... diffusion technique. Crystals diffracted to 2.6-Å resolution on a home X-ray source, and belonged to the orthorhombic space group, C2221 with unit cell dimensions a= 72.378. Å, b= 81.413 Å, c= 109.349 Å, and α=β=γ=90°. The crystal structure was determined by molecular replacement and contained two m ...
... diffusion technique. Crystals diffracted to 2.6-Å resolution on a home X-ray source, and belonged to the orthorhombic space group, C2221 with unit cell dimensions a= 72.378. Å, b= 81.413 Å, c= 109.349 Å, and α=β=γ=90°. The crystal structure was determined by molecular replacement and contained two m ...
Cyclol

The cyclol hypothesis is the first structural model of a folded, globular protein. It was developed by Dorothy Wrinch in the late 1930s, and was based on three assumptions. Firstly, the hypothesis assumes that two peptide groups can be crosslinked by a cyclol reaction (Figure 1); these crosslinks are covalent analogs of non-covalent hydrogen bonds between peptide groups. These reactions have been observed in the ergopeptides and other compounds. Secondly, it assumes that, under some conditions, amino acids will naturally make the maximum possible number of cyclol crosslinks, resulting in cyclol molecules (Figure 2) and cyclol fabrics (Figure 3). These cyclol molecules and fabrics have never been observed. Finally, the hypothesis assumes that globular proteins have a tertiary structure corresponding to Platonic solids and semiregular polyhedra formed of cyclol fabrics with no free edges. Such ""closed cyclol"" molecules have not been observed either.Although later data demonstrated that this original model for the structure of globular proteins needed to be amended, several elements of the cyclol model were verified, such as the cyclol reaction itself and the hypothesis that hydrophobic interactions are chiefly responsible for protein folding. The cyclol hypothesis stimulated many scientists to research questions in protein structure and chemistry, and was a precursor of the more accurate models hypothesized for the DNA double helix and protein secondary structure. The proposal and testing of the cyclol model also provides an excellent illustration of empirical falsifiability acting as part of the scientific method.