Methods for Control of Microbial Growth
... Halogen Antimicrobials Denature Proteins • Chlorine compounds (gaseous Cl, bleach, chloramine); can be sporicidal • Iodine (tincture, Betadine) • Halogens can react with any organic matter – a surface should be clean before applying them! ...
... Halogen Antimicrobials Denature Proteins • Chlorine compounds (gaseous Cl, bleach, chloramine); can be sporicidal • Iodine (tincture, Betadine) • Halogens can react with any organic matter – a surface should be clean before applying them! ...
Chapter 2 ppt
... Alkalosis: Blood pH above 7.45 A person usually cannot survive if the pH drops to 6.9 or rises to 7.8 for more than a few hours. ...
... Alkalosis: Blood pH above 7.45 A person usually cannot survive if the pH drops to 6.9 or rises to 7.8 for more than a few hours. ...
Protein Study Guide
... Proteins are large complex molecules found in the cells of all living things. Proteins are critical components in muscle mass, and tissues including bones, blood and hormones. Our bodies are made up of 18-20% protein by weight. Our muscles are 22% protein and 70% water. Animal products, such as meat ...
... Proteins are large complex molecules found in the cells of all living things. Proteins are critical components in muscle mass, and tissues including bones, blood and hormones. Our bodies are made up of 18-20% protein by weight. Our muscles are 22% protein and 70% water. Animal products, such as meat ...
Document
... to reveal the monoglucosylated species recognized by the lectin sites of calnexin/calreticulin. In their ATP-bound state, calnexin bind to the monoglucosylated oligosaccharide and hydrophobic segments of the unfolded glycoprotein (via their polypeptide binding or chaperone sites). Glycoprotein disso ...
... to reveal the monoglucosylated species recognized by the lectin sites of calnexin/calreticulin. In their ATP-bound state, calnexin bind to the monoglucosylated oligosaccharide and hydrophobic segments of the unfolded glycoprotein (via their polypeptide binding or chaperone sites). Glycoprotein disso ...
Chapter 2
... 2° Secondary – polypeptide chain coils into alpha helix and beta pleated sheet due to H bonding. 3° Tertiary – overall shape assumed by each polypeptide chain-Protein 3-D structure determined by interactions among side chains. 1. H bonding between amino acids 2. Ionic bonding between side chains 3. ...
... 2° Secondary – polypeptide chain coils into alpha helix and beta pleated sheet due to H bonding. 3° Tertiary – overall shape assumed by each polypeptide chain-Protein 3-D structure determined by interactions among side chains. 1. H bonding between amino acids 2. Ionic bonding between side chains 3. ...
y-ion series=A, AA, LAA, SLAA
... • “Additional” proteins crashes specificity and discovery rate, increase shared tryptic peptides. • Additional” proteins greatly(!) increase time of analysis. • Need all available protein sequences from species of interest derived from trEMBL and SWISSPROT (Everything that is in NCBI, Sanger and EMB ...
... • “Additional” proteins crashes specificity and discovery rate, increase shared tryptic peptides. • Additional” proteins greatly(!) increase time of analysis. • Need all available protein sequences from species of interest derived from trEMBL and SWISSPROT (Everything that is in NCBI, Sanger and EMB ...
The Cell Membrane
... Composed of a phospholipid bilayer with a collage of many different proteins, lipids and carbohydrates. A Phospholipid is composed of 1 glycerol molecule, 2 fatty acids and 1 phosphate group. This structure causes hydrophilic and hydrophobic regions. ...
... Composed of a phospholipid bilayer with a collage of many different proteins, lipids and carbohydrates. A Phospholipid is composed of 1 glycerol molecule, 2 fatty acids and 1 phosphate group. This structure causes hydrophilic and hydrophobic regions. ...
Chapter 2
... functional domain such as a barrel or pocket. In this example, the coils of a globin chain form a pocket. 4) Some proteins have quaternary structure, in which two or more polypeptide chains associate as one molecule. Hemoglobin, shown here, consists of four globin chains (green and blue). Each globi ...
... functional domain such as a barrel or pocket. In this example, the coils of a globin chain form a pocket. 4) Some proteins have quaternary structure, in which two or more polypeptide chains associate as one molecule. Hemoglobin, shown here, consists of four globin chains (green and blue). Each globi ...
The Chemical and Physical Basis of Life
... Inorganic Compounds - Solutions, Acids, Bases, Salts, pH, Buffer Systems, Movement of Solute and Solvent in Solution I. Solutions A. Mixture B. True Solutions Properties of Water 1. Water serves as a medium for the chemical reactions and participates in the reactions. 2. Water has high heat capacity ...
... Inorganic Compounds - Solutions, Acids, Bases, Salts, pH, Buffer Systems, Movement of Solute and Solvent in Solution I. Solutions A. Mixture B. True Solutions Properties of Water 1. Water serves as a medium for the chemical reactions and participates in the reactions. 2. Water has high heat capacity ...
Hints on Column Chromatography
... Amino Acids, Peptides & Proteins Peptides & proteins: • Derived from amino acids through peptide or amide bonds. • The amine and acid ends of amino acids couple to form amide (peptide) bonds in peptides/proteins/enzymes. • Proteins fold into well-defined structures. The hydrophobic residues segrega ...
... Amino Acids, Peptides & Proteins Peptides & proteins: • Derived from amino acids through peptide or amide bonds. • The amine and acid ends of amino acids couple to form amide (peptide) bonds in peptides/proteins/enzymes. • Proteins fold into well-defined structures. The hydrophobic residues segrega ...
Lecture 4 - Sites@UCI
... Protein must “fold”into structure How does protein “fold?” First, understand protein composition Proteins made up of _____________ ...
... Protein must “fold”into structure How does protein “fold?” First, understand protein composition Proteins made up of _____________ ...
No Slide Title
... •Avoid sequences that look funny (i.e. avoid low complexity sequences). •Try to avoid prolines and cysteines. ...
... •Avoid sequences that look funny (i.e. avoid low complexity sequences). •Try to avoid prolines and cysteines. ...
Key concepts_chromatin
... left-handed turns about an octamer of histones, two each of four types: H2A, H2B, H3 and H4. H2A and H2B form heterodimers, while H3 and H4 form a tetramer. Some of the histones exist in variant forms, and all are subject to a number of possible posttranslational modifications. This allows an enormo ...
... left-handed turns about an octamer of histones, two each of four types: H2A, H2B, H3 and H4. H2A and H2B form heterodimers, while H3 and H4 form a tetramer. Some of the histones exist in variant forms, and all are subject to a number of possible posttranslational modifications. This allows an enormo ...
ESBA Go Lean Protein Evaluation
... SNAP-Ed Activity Evaluation Form 00/00/17 with [Educator]: Go Lean with Protein For each statement the middle, please place an “X” in one of the boxes on each side that best represents your perceptions before the workshop (left) and now, after the workshop (right). BEFORE this Workshop Disagree Unsu ...
... SNAP-Ed Activity Evaluation Form 00/00/17 with [Educator]: Go Lean with Protein For each statement the middle, please place an “X” in one of the boxes on each side that best represents your perceptions before the workshop (left) and now, after the workshop (right). BEFORE this Workshop Disagree Unsu ...
Cheese Lab - Protein Chemistry
... amino acids, it will cause that part of the protein to try to stay away from water. When a section of the protein is made of hydrophilic amino acids, it will cause that part of the protein to try to stay in water. The protein molecules in milk, called caseins, are very hydrophobic. They try to get a ...
... amino acids, it will cause that part of the protein to try to stay away from water. When a section of the protein is made of hydrophilic amino acids, it will cause that part of the protein to try to stay in water. The protein molecules in milk, called caseins, are very hydrophobic. They try to get a ...
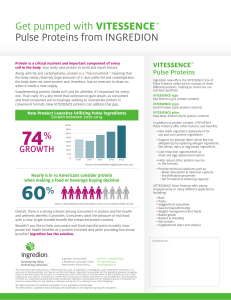
60% 74% - Ingredion
... cell in the body. Your body uses protein to build and repair tissues. Along with fat and carbohydrates, protein is a “macronutrient,” meaning that the body needs relatively large amounts of it. But unlike fat and carbohydrates, the body does not store protein and, therefore, has no reservoir to draw ...
... cell in the body. Your body uses protein to build and repair tissues. Along with fat and carbohydrates, protein is a “macronutrient,” meaning that the body needs relatively large amounts of it. But unlike fat and carbohydrates, the body does not store protein and, therefore, has no reservoir to draw ...
What happens to proteins key 14
... With the help of gastric juices and enzymes in your stomach and small intestine, proteins are broken down into amino acids and absorbed into your blood to be used by your cells. A limited supply of amino acids exist in pools in your body, which act as reservoir for the synthesis of protein as needed ...
... With the help of gastric juices and enzymes in your stomach and small intestine, proteins are broken down into amino acids and absorbed into your blood to be used by your cells. A limited supply of amino acids exist in pools in your body, which act as reservoir for the synthesis of protein as needed ...
The Structure and Function of Macromolecules
... Primary function = Energy Storage One gram of fat stores twice the energy of a gram of polysaccharide Advantageous to animals that have to move around – unlike plants that can have unlimited bulk without concern for mobility. Cells that store fat – adipose cells ...
... Primary function = Energy Storage One gram of fat stores twice the energy of a gram of polysaccharide Advantageous to animals that have to move around – unlike plants that can have unlimited bulk without concern for mobility. Cells that store fat – adipose cells ...
2.24 MB - KFUPM Resources v3
... chains of nucleotides twisted together into a double helix with links of hydrogen bonds Contains all inherited information necessary to build/maintain an organism, coded in the order of the nucleotide bases, with each three base “letters” forming a genetic code “word” All living organisms have D ...
... chains of nucleotides twisted together into a double helix with links of hydrogen bonds Contains all inherited information necessary to build/maintain an organism, coded in the order of the nucleotide bases, with each three base “letters” forming a genetic code “word” All living organisms have D ...
Ch.5
... (acidic proteins have pI < 7) If pH < pI, the protein is positively charged (basic proteins have pI > 7) ...
... (acidic proteins have pI < 7) If pH < pI, the protein is positively charged (basic proteins have pI > 7) ...
Proteins
... Because of their a.a`composition, proteins can bear +ve and –ve charges (amphoteric nature). The pH at which an a.a` or protein has no net charge is known as its isoelectric point. This characteristic is used for separation and quantitation of proteins such as electrophoresis. Solubility: ...
... Because of their a.a`composition, proteins can bear +ve and –ve charges (amphoteric nature). The pH at which an a.a` or protein has no net charge is known as its isoelectric point. This characteristic is used for separation and quantitation of proteins such as electrophoresis. Solubility: ...
protein pwrpt - Malibu High School
... How does my body use protein? • To build new cells • Maintain tissues • Make enzymes that do specific jobs such as digest foods. • Create neurotransmitters. ...
... How does my body use protein? • To build new cells • Maintain tissues • Make enzymes that do specific jobs such as digest foods. • Create neurotransmitters. ...
Cyclol

The cyclol hypothesis is the first structural model of a folded, globular protein. It was developed by Dorothy Wrinch in the late 1930s, and was based on three assumptions. Firstly, the hypothesis assumes that two peptide groups can be crosslinked by a cyclol reaction (Figure 1); these crosslinks are covalent analogs of non-covalent hydrogen bonds between peptide groups. These reactions have been observed in the ergopeptides and other compounds. Secondly, it assumes that, under some conditions, amino acids will naturally make the maximum possible number of cyclol crosslinks, resulting in cyclol molecules (Figure 2) and cyclol fabrics (Figure 3). These cyclol molecules and fabrics have never been observed. Finally, the hypothesis assumes that globular proteins have a tertiary structure corresponding to Platonic solids and semiregular polyhedra formed of cyclol fabrics with no free edges. Such ""closed cyclol"" molecules have not been observed either.Although later data demonstrated that this original model for the structure of globular proteins needed to be amended, several elements of the cyclol model were verified, such as the cyclol reaction itself and the hypothesis that hydrophobic interactions are chiefly responsible for protein folding. The cyclol hypothesis stimulated many scientists to research questions in protein structure and chemistry, and was a precursor of the more accurate models hypothesized for the DNA double helix and protein secondary structure. The proposal and testing of the cyclol model also provides an excellent illustration of empirical falsifiability acting as part of the scientific method.