Solutes
... Conjugate Acid – the base with the transferred proton (H+) Conjugate Acid – Base Pair – two substances related to each other by the donating and accepting of a single ...
... Conjugate Acid – the base with the transferred proton (H+) Conjugate Acid – Base Pair – two substances related to each other by the donating and accepting of a single ...
Bifunctional Asymmetric Catalysis: Cooperative Lewis Acid/Base
... action of Lewis acidic and Lewis basic catalysts by the simultaneous activation of both electrophilic and nucleophilic reaction partners. For our part, we have contributed three separate bifunctional methods that combine achiral Lewis acids with chiral cinchona alkaloid nucleophiles, for example, be ...
... action of Lewis acidic and Lewis basic catalysts by the simultaneous activation of both electrophilic and nucleophilic reaction partners. For our part, we have contributed three separate bifunctional methods that combine achiral Lewis acids with chiral cinchona alkaloid nucleophiles, for example, be ...
Chemical Reactions
... 1. Give the student the formula of barium hydroxide and sulfuric acid and ask them to predict the product of the reaction. 2. To a 250-mL beaker add 30 mL of 0.1 M sulfuric acid. 3. To the beaker add 50 mL of water and 2 drops of phenolphthalein. 4. Ask the student for their opinion as why the water ...
... 1. Give the student the formula of barium hydroxide and sulfuric acid and ask them to predict the product of the reaction. 2. To a 250-mL beaker add 30 mL of 0.1 M sulfuric acid. 3. To the beaker add 50 mL of water and 2 drops of phenolphthalein. 4. Ask the student for their opinion as why the water ...
12-Nucleophilic Reactions
... If the carbocation structure can thus be stabilized, then the transition state structure will also be stabilized and the rate of the reaction will be faster ...
... If the carbocation structure can thus be stabilized, then the transition state structure will also be stabilized and the rate of the reaction will be faster ...
Practice Exam-1A Fall 2016
... (e) CH3COOH, carbonic acid Hint: Follow the rules of naming the compounds. Be able to differentiate the rules between ionic and molecular compounds. 14. The atomic masses of 10B and 11B are 10.0129 amu (natural abundance 19.78% = 0.1978) and 11.0093 amu (natural abundance 80.22% = 0.8022), respectiv ...
... (e) CH3COOH, carbonic acid Hint: Follow the rules of naming the compounds. Be able to differentiate the rules between ionic and molecular compounds. 14. The atomic masses of 10B and 11B are 10.0129 amu (natural abundance 19.78% = 0.1978) and 11.0093 amu (natural abundance 80.22% = 0.8022), respectiv ...
Document
... d. Define Brønsted–Lowry acid and Brønsted–Lowry base. e. Write the chemical equation of a Brønsted–Lowry base in aqueous solution f. Write the chemical equation of an acid in aqueous solution using the hydronium ion. g. Learn the common strong acids and strong ...
... d. Define Brønsted–Lowry acid and Brønsted–Lowry base. e. Write the chemical equation of a Brønsted–Lowry base in aqueous solution f. Write the chemical equation of an acid in aqueous solution using the hydronium ion. g. Learn the common strong acids and strong ...
Answers - Scioly.org
... The lower Kf value for tetraamminecadmium(II) indicates that a larger concentration of ammonia molecules is present in the equilibrium mixture. This implies that the degree of dissociation (the reverse reaction of formation) must be greater for the tetraamminecadmium(II), so the coordinate covalent ...
... The lower Kf value for tetraamminecadmium(II) indicates that a larger concentration of ammonia molecules is present in the equilibrium mixture. This implies that the degree of dissociation (the reverse reaction of formation) must be greater for the tetraamminecadmium(II), so the coordinate covalent ...
Molecular Formulas - Hatboro
... outside of the classroom. The ultimate objective, of course, is to prepare you to take the AP Chemistry test on May 4, 2015, and in order to accomplish this, topics are covered very quickly. For this reason, most students take AP Chemistry after they already completed a year of high school chemistry ...
... outside of the classroom. The ultimate objective, of course, is to prepare you to take the AP Chemistry test on May 4, 2015, and in order to accomplish this, topics are covered very quickly. For this reason, most students take AP Chemistry after they already completed a year of high school chemistry ...
FINAL EXAM REVIEW PROBLEMS
... a. 0.10 M Na2CO3 b. 0.010 M Al2(SO4)3 54. Calculate the number of moles of Cl- ions in 1.75 L of 1.0 x 10-3 M AlCl3. 55. What volume of 12 M HCl must be taken to prepare 0.75 L of 0.45 M HCl? 56. When aqueous Na2SO4 and Pb(NO3)2 are mixed, PbSO4 precipitates. Calculate the PbSO4 formed when 1.25 L o ...
... a. 0.10 M Na2CO3 b. 0.010 M Al2(SO4)3 54. Calculate the number of moles of Cl- ions in 1.75 L of 1.0 x 10-3 M AlCl3. 55. What volume of 12 M HCl must be taken to prepare 0.75 L of 0.45 M HCl? 56. When aqueous Na2SO4 and Pb(NO3)2 are mixed, PbSO4 precipitates. Calculate the PbSO4 formed when 1.25 L o ...
Chapter 4. Aqueous Reactions and Solution Stoichiometry
... We usually think that an oxidation necessarily involves a reaction with oxygen and/or addition of an atom of oxygen to the formula. We often think that all atoms of the same element must have the same oxidation number and that this number is uniquely related to the atom’s location in the periodic ta ...
... We usually think that an oxidation necessarily involves a reaction with oxygen and/or addition of an atom of oxygen to the formula. We often think that all atoms of the same element must have the same oxidation number and that this number is uniquely related to the atom’s location in the periodic ta ...
Bio 2 alkanes+isomerism
... Suppose we want to add one more C atom to propane. We have two different kinds of H we can replace, a methylene H or a methyl H. If we replace the methyl H, we have butane. If we replace the methylene H, we have a skeletal isomer of butane. This isomer of butane has a common or trivial name, isobuta ...
... Suppose we want to add one more C atom to propane. We have two different kinds of H we can replace, a methylene H or a methyl H. If we replace the methyl H, we have butane. If we replace the methylene H, we have a skeletal isomer of butane. This isomer of butane has a common or trivial name, isobuta ...
Unit C3 - Chemistry in Action
... 2) Marble chips are made of calcium carbonate (CaCO3). What volume of carbon dioxide will be released when 500g of CaCO3 is reacted with dilute hydrochloric acid? CaCO3 + 2HCl ...
... 2) Marble chips are made of calcium carbonate (CaCO3). What volume of carbon dioxide will be released when 500g of CaCO3 is reacted with dilute hydrochloric acid? CaCO3 + 2HCl ...
Unit C3 - Chemistry In Action
... 2) Marble chips are made of calcium carbonate (CaCO3). What volume of carbon dioxide will be released when 500g of CaCO3 is reacted with dilute hydrochloric acid? CaCO3 + 2HCl ...
... 2) Marble chips are made of calcium carbonate (CaCO3). What volume of carbon dioxide will be released when 500g of CaCO3 is reacted with dilute hydrochloric acid? CaCO3 + 2HCl ...
Acid + Base Class # 1
... 20. Strong acids & strong bases have lots of ions in solution. Strong acids & strong bases are good electrolytes, they will conduct electricity well because of all the loose ions they have in solution. 21. All acids and all bases are electrolytes. 22. Their electrolyte strengths (their ability to c ...
... 20. Strong acids & strong bases have lots of ions in solution. Strong acids & strong bases are good electrolytes, they will conduct electricity well because of all the loose ions they have in solution. 21. All acids and all bases are electrolytes. 22. Their electrolyte strengths (their ability to c ...
Question paper - Edexcel
... Answer ALL the questions in this section. You should aim to spend no more than 20 minutes on this section. For each question, select one answer from A to D and put a cross in the box . and then mark your new answer with a If you change your mind, put a line through the box cross . 1 In which of the ...
... Answer ALL the questions in this section. You should aim to spend no more than 20 minutes on this section. For each question, select one answer from A to D and put a cross in the box . and then mark your new answer with a If you change your mind, put a line through the box cross . 1 In which of the ...
10.4 Alcohols - SCIS Teachers
... •The physical properties of alcohols are similar to those of both water and hydrocarbons •The shorter chain alcohols such as methanol and ethanol are similar to water, in general they •have higher boiling points than hydrocarbons but lower than water •dissolve in water to some degree •are more polar ...
... •The physical properties of alcohols are similar to those of both water and hydrocarbons •The shorter chain alcohols such as methanol and ethanol are similar to water, in general they •have higher boiling points than hydrocarbons but lower than water •dissolve in water to some degree •are more polar ...
by John Mu
... and shelter. As the cultural development of humans continued, they looked for a greater variety of materials to cater for their needs. The twentieth century saw an explosion in both the use of traditional materials and in the research for development of a wider range of materials to satisfy technolo ...
... and shelter. As the cultural development of humans continued, they looked for a greater variety of materials to cater for their needs. The twentieth century saw an explosion in both the use of traditional materials and in the research for development of a wider range of materials to satisfy technolo ...
chemical reactions
... CHEMISTRY AND LIFE One unromantic but productive way of viewing life is to see it as a set of coordinated chemical reactions. ...
... CHEMISTRY AND LIFE One unromantic but productive way of viewing life is to see it as a set of coordinated chemical reactions. ...
Nucleophilic Additions to Carbonyl Group
... group bonded to the carbonyl group with another heteroatom or group. The replacement groups include halogens (mostly Cl and Br), which form acyl halides; carboxylate (RCOO—) groups, which form carboxylic anhydrides; alkoxy (RO—) groups, which form esters; and amino (—NR2) groups, which form amides. ...
... group bonded to the carbonyl group with another heteroatom or group. The replacement groups include halogens (mostly Cl and Br), which form acyl halides; carboxylate (RCOO—) groups, which form carboxylic anhydrides; alkoxy (RO—) groups, which form esters; and amino (—NR2) groups, which form amides. ...
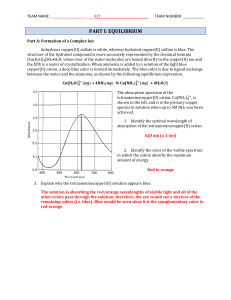
Chem 12 UNIT TWO CHEMICAL EQUILIBRIUM 7.1 REVERSIBLE
... (in text they give an example of Kits Beach - sodium and chloride ions will not form sodium chloride and precipitate out of solution from the ocean water. Ions in solution have higher entropy than ions in crystal lattice structure) ...
... (in text they give an example of Kits Beach - sodium and chloride ions will not form sodium chloride and precipitate out of solution from the ocean water. Ions in solution have higher entropy than ions in crystal lattice structure) ...
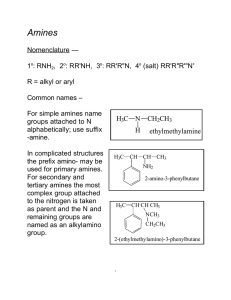
Amines
... - combining molecules with an acid (COOH) group on each end are reacted with two chemicals that contain amine (NH2) groups on each end. - Form nylon 6,6, made of hexamethylene diamine with six carbon atoms and acidipic acid, as well as six carbon atoms. • The second approach: - a compound has an aci ...
... - combining molecules with an acid (COOH) group on each end are reacted with two chemicals that contain amine (NH2) groups on each end. - Form nylon 6,6, made of hexamethylene diamine with six carbon atoms and acidipic acid, as well as six carbon atoms. • The second approach: - a compound has an aci ...
226 amines lec
... -NH2 is considered a substituent, like chloro or nitro, and is called amino. It is located by number in the usual way. For secondary and tertiary amines, the largest alkyl group is taken as the ...
... -NH2 is considered a substituent, like chloro or nitro, and is called amino. It is located by number in the usual way. For secondary and tertiary amines, the largest alkyl group is taken as the ...
Derivatization Reagents - Sigma
... requires strongly basic catalysts (sodium or potassium methoxide). More acidic OH groups (phenols, carboxylic acids) require less basic catalysts (hydrogen chloride, boron trifluoride). SUPELCO Bulletin 909 ...
... requires strongly basic catalysts (sodium or potassium methoxide). More acidic OH groups (phenols, carboxylic acids) require less basic catalysts (hydrogen chloride, boron trifluoride). SUPELCO Bulletin 909 ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.