Chapter 4. Hard and Soft Acid/Base Theory based on Lewis Acids
... Definitions of Lewis Acid-Base Theory. Lewis acid/base theory (sometimes called donoracceptor theory) is a broad, widely applicable approach to the classification of chemical substances and the analysis of chemical reactions. According to this theory, a base is defined as an electron pair donor, and ...
... Definitions of Lewis Acid-Base Theory. Lewis acid/base theory (sometimes called donoracceptor theory) is a broad, widely applicable approach to the classification of chemical substances and the analysis of chemical reactions. According to this theory, a base is defined as an electron pair donor, and ...
Saturday Study Session 1 1st Class Reactions
... The dehydrated sample absorbed moisture after heating. c. The amount of the hydrate sample used was too small. d. The crucible was not heated to constant mass before use. e. Excess heating caused the dehydrated sample to decompose. ...
... The dehydrated sample absorbed moisture after heating. c. The amount of the hydrate sample used was too small. d. The crucible was not heated to constant mass before use. e. Excess heating caused the dehydrated sample to decompose. ...
Chapter 3 – part I Sections 1-3
... ions) are these. You will learn later that strong acids and bases are strong electrolytes. • Weak electrolytes are weak conductors, ionic compounds that are insoluble are these. Insoluble = only a few dissolve into ions. You will learn later that weak acids and bases are weak electrolytes. • Non-ele ...
... ions) are these. You will learn later that strong acids and bases are strong electrolytes. • Weak electrolytes are weak conductors, ionic compounds that are insoluble are these. Insoluble = only a few dissolve into ions. You will learn later that weak acids and bases are weak electrolytes. • Non-ele ...
Unit 11 acids and bases part 1
... Each salt listed in this table can be formed by the reaction between an acid and a base. ...
... Each salt listed in this table can be formed by the reaction between an acid and a base. ...
Learning Guide for Chapter 16
... esters - also have C=O band alcohols - also have OH band anhydrides - also have 2 C=O bands NMR Spectra of Ethers What chemical shift are the H's next to the O in an ether? 3-4 ppm H ...
... esters - also have C=O band alcohols - also have OH band anhydrides - also have 2 C=O bands NMR Spectra of Ethers What chemical shift are the H's next to the O in an ether? 3-4 ppm H ...
Past AP FRQ`s Linked to Text Chapters
... Consider three unlabeled bottles, each containing small pieces of one of the following metals. - Magnesium - Sodium - Silver ...
... Consider three unlabeled bottles, each containing small pieces of one of the following metals. - Magnesium - Sodium - Silver ...
Biology 251 Fall 2015 1 TOPIC 23: ACID BASE BALANCE I
... that in mind as you read these notes, because I have not explicitly included this enzyme in most of the following discussion. ...
... that in mind as you read these notes, because I have not explicitly included this enzyme in most of the following discussion. ...
The alcohols
... Alcohols are named according to standard IUPAC rules • select the longest chain of C atoms containing the O-H group; • remove the e and add ol after the basic name • number the chain starting from the end nearer the O-H group • the number is placed after the an and before the ol ... e.g butan-2-ol • ...
... Alcohols are named according to standard IUPAC rules • select the longest chain of C atoms containing the O-H group; • remove the e and add ol after the basic name • number the chain starting from the end nearer the O-H group • the number is placed after the an and before the ol ... e.g butan-2-ol • ...
No Slide Title
... Alcohols are named according to standard IUPAC rules • select the longest chain of C atoms containing the O-H group; • remove the e and add ol after the basic name • number the chain starting from the end nearer the O-H group • the number is placed after the an and before the ol ... e.g butan-2-ol • ...
... Alcohols are named according to standard IUPAC rules • select the longest chain of C atoms containing the O-H group; • remove the e and add ol after the basic name • number the chain starting from the end nearer the O-H group • the number is placed after the an and before the ol ... e.g butan-2-ol • ...
KS4-Chemical-Reactions
... Reversible and Irreversible Reaction • Most Chemical reactions are considered irreversible in that the new products are not readily changed back into reactants. Eg. Once you have reacted magnesium with hydrochloric acid it is very hard to get the magnesium back. ...
... Reversible and Irreversible Reaction • Most Chemical reactions are considered irreversible in that the new products are not readily changed back into reactants. Eg. Once you have reacted magnesium with hydrochloric acid it is very hard to get the magnesium back. ...
Types of Chemical Reactions Name_________________________
... How does the aluminum bromide precipitate become deposited on the watch glass covering the beaker since it does not come in contact with the bromine liquid? ...
... How does the aluminum bromide precipitate become deposited on the watch glass covering the beaker since it does not come in contact with the bromine liquid? ...
Synthesis of Heterocycles from Anthranilic acid
... of anthranilic acid starts when Carl Julius Fritzsche (1808-1871) experimented in his laboratory in St. Petersburg with degradation of the ancient dye indigo in the late 1830s.1 The chemical structure of indigo was not known at the time, but it was of interest to the flourishing textile industry whi ...
... of anthranilic acid starts when Carl Julius Fritzsche (1808-1871) experimented in his laboratory in St. Petersburg with degradation of the ancient dye indigo in the late 1830s.1 The chemical structure of indigo was not known at the time, but it was of interest to the flourishing textile industry whi ...
Synthesis of Alum Lab
... 2Al(s) + 2K+(aq) + 2OH-(aq) + 6H2O(l) 2[Al(OH)4]-(aq) + 2K+(aq) + 3H2(g) The oxidation and reduction half reactions: ...
... 2Al(s) + 2K+(aq) + 2OH-(aq) + 6H2O(l) 2[Al(OH)4]-(aq) + 2K+(aq) + 3H2(g) The oxidation and reduction half reactions: ...
aldehyde group - Imperial Valley College Faculty Websites
... carbon as shown here. The -hydrogen is a hydrogen atom attached to an -carbon atom. -Hydrogen atoms are slightly acidic and are more easily released than , , or H atoms during an aldol reaction. ...
... carbon as shown here. The -hydrogen is a hydrogen atom attached to an -carbon atom. -Hydrogen atoms are slightly acidic and are more easily released than , , or H atoms during an aldol reaction. ...
OC 2/e Ch 15
... of water (hydration) to the carbonyl group of an aldehyde or ketone gives a gem-diol, commonly referred to as a hydrate • when formaldehyde is dissolved in water at 20°C, the carbonyl group is more than 99% hydrated O HCH + H2 O ...
... of water (hydration) to the carbonyl group of an aldehyde or ketone gives a gem-diol, commonly referred to as a hydrate • when formaldehyde is dissolved in water at 20°C, the carbonyl group is more than 99% hydrated O HCH + H2 O ...
1 -
... bond as the basis for the parent name. The position of the triple bond takes the lowest possible number. If both double and triple bonds are present, the ending becomes –enyne and numbering is such as to give the lowest possible numbers to the double and triple bonds, irrespective of whether – ...
... bond as the basis for the parent name. The position of the triple bond takes the lowest possible number. If both double and triple bonds are present, the ending becomes –enyne and numbering is such as to give the lowest possible numbers to the double and triple bonds, irrespective of whether – ...
odd - WWW2
... have the argon condense out, since it has a boiling point higher than dinitrogen or dihydrogen. However, the energy requirements for this procedure would probably be prohibitive. Alternatively, the used gases could be periodically siphoned off and the dihydrogen burned as an energy source. ...
... have the argon condense out, since it has a boiling point higher than dinitrogen or dihydrogen. However, the energy requirements for this procedure would probably be prohibitive. Alternatively, the used gases could be periodically siphoned off and the dihydrogen burned as an energy source. ...
Chemistry
... relate bond energy with bond strength; define bond length; explain ionic character of covalent bond; predict the nature of bonding on the basis of electro negativity; describe the change in bond length of hetronuclear molecules due to the difference of E.N in bonded atoms explain dipole moment; pred ...
... relate bond energy with bond strength; define bond length; explain ionic character of covalent bond; predict the nature of bonding on the basis of electro negativity; describe the change in bond length of hetronuclear molecules due to the difference of E.N in bonded atoms explain dipole moment; pred ...
General Sciences Sample First Exercise Propanoic Acid Solution
... C2H5COOH is the conjugate acid and they form a pair. II-1) Case of dilution: nS1 = nS2 so V = (100×10-3) ÷ 10-1 = 1 mL. Fold F = C1 / C2 = 100. II-2-a) S1 is more concentrated than S2, so C1> C2 and consequently [H3O+]S1 > [H3O+]S2 But pH is inversely proportional to [H3O+] then pH1 < pH2 Thus the s ...
... C2H5COOH is the conjugate acid and they form a pair. II-1) Case of dilution: nS1 = nS2 so V = (100×10-3) ÷ 10-1 = 1 mL. Fold F = C1 / C2 = 100. II-2-a) S1 is more concentrated than S2, so C1> C2 and consequently [H3O+]S1 > [H3O+]S2 But pH is inversely proportional to [H3O+] then pH1 < pH2 Thus the s ...
9. The Copigmentation Interactions between Strawberry
... parameters of the reaction shows that the activation energy of the first reaction channel is lower and the frequency factor is higher supporting a higher reaction rate. The red raspberry extracts were selected as pigments and five phenolic acids include caffeic acid as copigments(Sun et al., 2010). ...
... parameters of the reaction shows that the activation energy of the first reaction channel is lower and the frequency factor is higher supporting a higher reaction rate. The red raspberry extracts were selected as pigments and five phenolic acids include caffeic acid as copigments(Sun et al., 2010). ...
The aim of this exercise is to study the acid... prepare a buffer solution. General Sciences Sample
... C2H5COOH is the conjugate acid and they form a pair. II-1) Case of dilution: nS1 = nS2 so V = (100×10-3) ÷ 10-1 = 1 mL. Fold F = C1 / C2 = 100. II-2-a) S1 is more concentrated than S2, so C1> C2 and consequently [H3O+]S1 > [H3O+]S2 But pH is inversely proportional to [H3O+] then pH1 < pH2 Thus the s ...
... C2H5COOH is the conjugate acid and they form a pair. II-1) Case of dilution: nS1 = nS2 so V = (100×10-3) ÷ 10-1 = 1 mL. Fold F = C1 / C2 = 100. II-2-a) S1 is more concentrated than S2, so C1> C2 and consequently [H3O+]S1 > [H3O+]S2 But pH is inversely proportional to [H3O+] then pH1 < pH2 Thus the s ...
Document
... The bulb in Figure 4.6(a) is only dimly lit because acetic acid is a weak acid and therefore a weak electrolyte [recall Figure 4.3(c)]. The situation in (b) is similar because ammonia is a weak base and therefore also ionizes only slightly. When the two solutions are mixed, which is what has been do ...
... The bulb in Figure 4.6(a) is only dimly lit because acetic acid is a weak acid and therefore a weak electrolyte [recall Figure 4.3(c)]. The situation in (b) is similar because ammonia is a weak base and therefore also ionizes only slightly. When the two solutions are mixed, which is what has been do ...
STUDY GUIDE
... rearranged. Ask What gaseous product would form by reacting sulfuric acid with sodium cyanide? (hydrogen cyanide, HCN) Ask What gaseous product would form from the reaction of sulfuric acid and sodium sulfide? (hydrogen sulfide, H2S) Explain that both HCN and H2S are gases that can be lethal if inha ...
... rearranged. Ask What gaseous product would form by reacting sulfuric acid with sodium cyanide? (hydrogen cyanide, HCN) Ask What gaseous product would form from the reaction of sulfuric acid and sodium sulfide? (hydrogen sulfide, H2S) Explain that both HCN and H2S are gases that can be lethal if inha ...
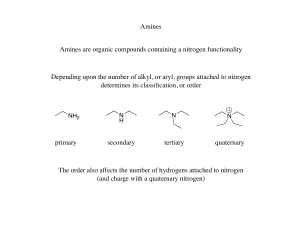
Chapter 19
... Amine salts are also used to catalyze a variety of organic reactions that feature two components that are soluble in different liquid phases (e.g. organic and aqueous) ...
... Amine salts are also used to catalyze a variety of organic reactions that feature two components that are soluble in different liquid phases (e.g. organic and aqueous) ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.