File
... 2 Na(s) + SrBr2(aq) NR CrI3(aq) + 3 KCl(aq) CrCl3(s) + 3 KI(aq) (DR – ppt) Zn(s) + H2SO3(aq) ZnSO3(aq) + H2(g) (SR – metal + acid) K2CO3(aq) + 2 HI(aq) 2 KI(aq) + H2CO3(aq) (DR – gas) ...
... 2 Na(s) + SrBr2(aq) NR CrI3(aq) + 3 KCl(aq) CrCl3(s) + 3 KI(aq) (DR – ppt) Zn(s) + H2SO3(aq) ZnSO3(aq) + H2(g) (SR – metal + acid) K2CO3(aq) + 2 HI(aq) 2 KI(aq) + H2CO3(aq) (DR – gas) ...
Ch. 11 Notes with Answers
... • 2º alcohols are way slower • 1º alcohols can’t react at all via this mechanism, because 1º R+ are too unstable. • Ditto for vinyl or aryl alcohols 5. HBr can also react with 1º ROH to give 1º RBr, although it is not often the method of choice • The mechanism is different, but rather interesting (n ...
... • 2º alcohols are way slower • 1º alcohols can’t react at all via this mechanism, because 1º R+ are too unstable. • Ditto for vinyl or aryl alcohols 5. HBr can also react with 1º ROH to give 1º RBr, although it is not often the method of choice • The mechanism is different, but rather interesting (n ...
Lecture - Ch 19
... of Aldehydes and Ketones • Nucleophilic additions to aldehydes and ketones have two general variations – Product is a direct result of the tetrahedral intermediate being protonated by water or acid – Carbonyl oxygen atom is protonated and eliminated as HO- or H2O to give a product with a C=Nu double ...
... of Aldehydes and Ketones • Nucleophilic additions to aldehydes and ketones have two general variations – Product is a direct result of the tetrahedral intermediate being protonated by water or acid – Carbonyl oxygen atom is protonated and eliminated as HO- or H2O to give a product with a C=Nu double ...
Unit 10
... Extracting metal – getting metal from ores. What are the metal compounds from mineral ores? Are they soluble in water? Insoluble metal oxides, carbonates and sulphides – found in ores Which metals are found free in nature? Unreactive metals such as gold and platinum found free (as elements) in natur ...
... Extracting metal – getting metal from ores. What are the metal compounds from mineral ores? Are they soluble in water? Insoluble metal oxides, carbonates and sulphides – found in ores Which metals are found free in nature? Unreactive metals such as gold and platinum found free (as elements) in natur ...
Lecture 10 Carbon-Nitrogen Bonds Formation I
... These reactions are analogous to the base-catalyzed hydrolysis of the carbonyl compounds. The order of reactivity of the compounds having different leaving groups is: acid halide > anhydride > ester > amide. ...
... These reactions are analogous to the base-catalyzed hydrolysis of the carbonyl compounds. The order of reactivity of the compounds having different leaving groups is: acid halide > anhydride > ester > amide. ...
WELCOME TO AP CHEMISTRY
... I am very much looking forward to meeting you in September and teaching you second-year Chemistry in preparation for the AP Test in May, 2014. In September we will start out running and then proceed to a gallop as the months go by! The summer assignment is attached. The purpose of the assignment is ...
... I am very much looking forward to meeting you in September and teaching you second-year Chemistry in preparation for the AP Test in May, 2014. In September we will start out running and then proceed to a gallop as the months go by! The summer assignment is attached. The purpose of the assignment is ...
6CH02 - MPPE
... SECTION A Answer ALL the questions in this section. You should aim to spend no more than 20 minutes on this section. For each question, select one answer from A to D and put a cross in the box . and then mark your new answer with If you change your mind, put a line through the box a cross . 1 The c ...
... SECTION A Answer ALL the questions in this section. You should aim to spend no more than 20 minutes on this section. For each question, select one answer from A to D and put a cross in the box . and then mark your new answer with If you change your mind, put a line through the box a cross . 1 The c ...
Tr-dT, 2-cyanoethanol
... of the reaction. The mixture was diluted by 54 ml of water (t = 1°C) and immediatly applied to a column (25 x 170 mm) with Molselect-DEAE 25 ion-exchange resin in HC07 form with the flow rate 70 ml/h. A glass filter was used to remove DCC and dicyclohexylurea (DCU) precipitates. The elution was perf ...
... of the reaction. The mixture was diluted by 54 ml of water (t = 1°C) and immediatly applied to a column (25 x 170 mm) with Molselect-DEAE 25 ion-exchange resin in HC07 form with the flow rate 70 ml/h. A glass filter was used to remove DCC and dicyclohexylurea (DCU) precipitates. The elution was perf ...
Language of chemistry
... Carbonate - CO3 2Bicarbonate - HCO3 Nitrate - NO3 Sulfate – SO4 2Sulfite – SO3 2Chloride – Cl Ammonium– NH4+ Salt – It is a compound formed when an acid reacts with a base. Examples: NaCl sodium chloride KCl potassium chloride NH4Cl ammonium chloride MgCO3 magnesium carbonate NH4NO3 ammonium nitrate ...
... Carbonate - CO3 2Bicarbonate - HCO3 Nitrate - NO3 Sulfate – SO4 2Sulfite – SO3 2Chloride – Cl Ammonium– NH4+ Salt – It is a compound formed when an acid reacts with a base. Examples: NaCl sodium chloride KCl potassium chloride NH4Cl ammonium chloride MgCO3 magnesium carbonate NH4NO3 ammonium nitrate ...
amines amide - TangHua2012-2013
... How to tell amines and amides? • If we only know the chemical formulas… • Tell the functional group which substituents attach to • Amines---NH2 group • Amides---CONH2 group ...
... How to tell amines and amides? • If we only know the chemical formulas… • Tell the functional group which substituents attach to • Amines---NH2 group • Amides---CONH2 group ...
AQA Additional Sci C2 Revision Guide
... When elements react, their atoms join with other atoms to form compounds. This involves giving, taking or sharing electrons in order to gain full outer shells. This gives the most stable arrangement of electrons. Ionic bonding Compounds formed from metals and non-metals consist of ions. Metal atoms ...
... When elements react, their atoms join with other atoms to form compounds. This involves giving, taking or sharing electrons in order to gain full outer shells. This gives the most stable arrangement of electrons. Ionic bonding Compounds formed from metals and non-metals consist of ions. Metal atoms ...
equilibrium questions - Southington Public Schools
... Answer the following questions that relate to solubility of salts of lead and barium. (a) A saturated solution is prepared by adding excess PbI2(s) to distilled water to form 1.0 L of solution at 25˚C. The concentration of Pb2+(aq) in the saturated solution is found to be 1.3 10–3 M. The chemical ...
... Answer the following questions that relate to solubility of salts of lead and barium. (a) A saturated solution is prepared by adding excess PbI2(s) to distilled water to form 1.0 L of solution at 25˚C. The concentration of Pb2+(aq) in the saturated solution is found to be 1.3 10–3 M. The chemical ...
Industrial Chemistry - Deans Community High School
... possible routes. One is a direct,single-step process, (adding HCl (aq) directly to the solid NaOH) and secondly a two-step process (dissolve the solid NaOH in water first, then add the HCl(aq)) All steps are exothermic. If Hess’s Law applies, the enthalpy change for route 1 must be the same for the ...
... possible routes. One is a direct,single-step process, (adding HCl (aq) directly to the solid NaOH) and secondly a two-step process (dissolve the solid NaOH in water first, then add the HCl(aq)) All steps are exothermic. If Hess’s Law applies, the enthalpy change for route 1 must be the same for the ...
Chemical Reactions Chapter 11
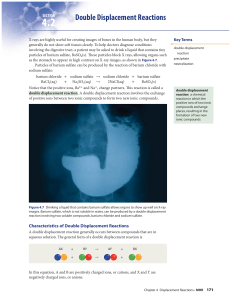
... 4. Double-replacement reaction is a chemical change involving an exchange of positive ions between two compounds. – you can recognize a double-replacement reaction because both the reactants and the products are two compounds – They generally take place in aqueous solutions, and often produce a prec ...
... 4. Double-replacement reaction is a chemical change involving an exchange of positive ions between two compounds. – you can recognize a double-replacement reaction because both the reactants and the products are two compounds – They generally take place in aqueous solutions, and often produce a prec ...
AP Chemistry Summer Packet ANSWERS
... b. Solid zinc metal reacts with sulfuric acid to form hydrogen gas and an aqueous solution of zinc sulfate. c. When liquid phosphorous trichloride is added to water, it reacts to form aqueous phosphorous acid, and hydrochloric acid. d. When hydrogen sulfide gas is passed over solid hot iron (III) hy ...
... b. Solid zinc metal reacts with sulfuric acid to form hydrogen gas and an aqueous solution of zinc sulfate. c. When liquid phosphorous trichloride is added to water, it reacts to form aqueous phosphorous acid, and hydrochloric acid. d. When hydrogen sulfide gas is passed over solid hot iron (III) hy ...
Here are the answers and work for your summer packet.
... b. Solid zinc metal reacts with sulfuric acid to form hydrogen gas and an aqueous solution of zinc sulfate. c. When liquid phosphorous trichloride is added to water, it reacts to form aqueous phosphorous acid, and hydrochloric acid. d. When hydrogen sulfide gas is passed over solid hot iron (III) hy ...
... b. Solid zinc metal reacts with sulfuric acid to form hydrogen gas and an aqueous solution of zinc sulfate. c. When liquid phosphorous trichloride is added to water, it reacts to form aqueous phosphorous acid, and hydrochloric acid. d. When hydrogen sulfide gas is passed over solid hot iron (III) hy ...
TANNIC ACID
... gallotannins may be obtained from nutgalls, the excrescences which form on young twigs of various Quercus species, e.g., Q. infectoria; these include Chinese and Aleppo tannins. They may also be obtained from various Sumac species, e.g. Rhus corieria, R. galabra, R. thypia; these include Sicilian an ...
... gallotannins may be obtained from nutgalls, the excrescences which form on young twigs of various Quercus species, e.g., Q. infectoria; these include Chinese and Aleppo tannins. They may also be obtained from various Sumac species, e.g. Rhus corieria, R. galabra, R. thypia; these include Sicilian an ...
CH - UTH e
... • Can’t make Grignards with acidic or electro‐philic f functional groups present in the molecule: i l i h l l ...
... • Can’t make Grignards with acidic or electro‐philic f functional groups present in the molecule: i l i h l l ...
HONG KONG DIPLOMA OF SECONDARY EDUCATION
... Mars is the fourth planet from the Sun. Mars has a very different atmosphere from that of the Earth. The atmosphere is relatively thin with an average pressure of 0.6 kilopascals (kPa) (compared to Earth’s 101.3 kPa). It is actually less than 1% the atmospheric density of Earth. Therefore, there is ...
... Mars is the fourth planet from the Sun. Mars has a very different atmosphere from that of the Earth. The atmosphere is relatively thin with an average pressure of 0.6 kilopascals (kPa) (compared to Earth’s 101.3 kPa). It is actually less than 1% the atmospheric density of Earth. Therefore, there is ...
Mole Equation Homework Hint: Start equations with the numbers
... Hint: Start equations with the numbers given, and pay close attention to what the question is asking you to find. Usually, the first step in most stoichiometry problems (calculation of quantities in chemical equations) is to convert the given numbers to moles. SHOW YOUR WORK!!!!!!!!!!!!!!!!!!!!!!!!! ...
... Hint: Start equations with the numbers given, and pay close attention to what the question is asking you to find. Usually, the first step in most stoichiometry problems (calculation of quantities in chemical equations) is to convert the given numbers to moles. SHOW YOUR WORK!!!!!!!!!!!!!!!!!!!!!!!!! ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.