Instructions for AP/IB 2 Chem Summer Assignment Note
... Learn the general formula for each type of reaction. If the reaction occurs in water solution, you must give the net ionic equation. If it doesn't occur in aqueous solution, the atoms/molecules do not exist as ions. ...
... Learn the general formula for each type of reaction. If the reaction occurs in water solution, you must give the net ionic equation. If it doesn't occur in aqueous solution, the atoms/molecules do not exist as ions. ...
Chapter 19 - people.vcu.edu
... In fact, it’s so active that you can halogenate without the metal Lewis-acid catalyst. NH2 ...
... In fact, it’s so active that you can halogenate without the metal Lewis-acid catalyst. NH2 ...
Unit 2
... • Thermal cracking is where heat is used to split large molecules into smaller ones. • Catalytic cracking is where a catalyst is used to split large molecules into smaller ones. ...
... • Thermal cracking is where heat is used to split large molecules into smaller ones. • Catalytic cracking is where a catalyst is used to split large molecules into smaller ones. ...
3.5 The Alcohols
... 1) Classify the following alcohols as primary, secondary or tertiary: a. CH3CH2CH2OH b. CH3CH(OH)CH2CH3 c. CH3CH(OH)CH(CH3)2 d. Pentan – 2 – ol e. Butan – 1 – ol f. 2 – Methyl propan – 2 – ol 2) Draw the oxidation products of the alcohols in question 1: a. ...
... 1) Classify the following alcohols as primary, secondary or tertiary: a. CH3CH2CH2OH b. CH3CH(OH)CH2CH3 c. CH3CH(OH)CH(CH3)2 d. Pentan – 2 – ol e. Butan – 1 – ol f. 2 – Methyl propan – 2 – ol 2) Draw the oxidation products of the alcohols in question 1: a. ...
Brominations and Alkene Synthesis CHM 233 Review
... alkene to make an alcohol. In conc. sulfuric acid medium, the acid helps to REMOVE water from an alcohol to make an alkene (the sulfuric acid DEHYDRATES the alcohol) ...
... alkene to make an alcohol. In conc. sulfuric acid medium, the acid helps to REMOVE water from an alcohol to make an alkene (the sulfuric acid DEHYDRATES the alcohol) ...
CHM 331 : General Organic Chemistry
... alkene to make an alcohol. In conc. sulfuric acid medium, the acid helps to REMOVE water from an alcohol to make an alkene (the sulfuric acid DEHYDRATES the alcohol) Alkenes I ...
... alkene to make an alcohol. In conc. sulfuric acid medium, the acid helps to REMOVE water from an alcohol to make an alkene (the sulfuric acid DEHYDRATES the alcohol) Alkenes I ...
Environmental Chemistry
... 2NO2 + H2O HNO3 + HNO2 (nitric and nitrous acid) These non-metal oxides are all gases Their acidic products all contribute to the acidity of rain. Note: strong acids completely dissociate to their ions, so that a monoprotic acid will have the same hydronium ion concentration as the original concen ...
... 2NO2 + H2O HNO3 + HNO2 (nitric and nitrous acid) These non-metal oxides are all gases Their acidic products all contribute to the acidity of rain. Note: strong acids completely dissociate to their ions, so that a monoprotic acid will have the same hydronium ion concentration as the original concen ...
Chem 216 H W13 Notes - Dr. Masato Koreeda Thin
... adsorbent layer. When illuminated with an ultraviolet (UV) lamp, the absorbent then glows the pale green or blue colored fluorescent light. However, dark spots show up at the places where UVabsorbing organic compounds are located on the TLC plate because they quench the fluorescence. To be detectabl ...
... adsorbent layer. When illuminated with an ultraviolet (UV) lamp, the absorbent then glows the pale green or blue colored fluorescent light. However, dark spots show up at the places where UVabsorbing organic compounds are located on the TLC plate because they quench the fluorescence. To be detectabl ...
Organometallics II
... but species formed is unstable and dissociates under the reaction conditions to form a ketone ...
... but species formed is unstable and dissociates under the reaction conditions to form a ketone ...
makeup2
... cm. If the density of silver is 10.5 g/cm3, how long a strip should be cut to obtain 1.00 g of the metal? (A) 9.52 x 10¯2 cm (B) 1.19 cm (C) 8.00 cm (D) 12.5 cm 2. A car traveling at 10 miles per hour emits about 0.15 kg of carbon monoxide (CO) gas per mile. How many moles of CO are emitted per mile ...
... cm. If the density of silver is 10.5 g/cm3, how long a strip should be cut to obtain 1.00 g of the metal? (A) 9.52 x 10¯2 cm (B) 1.19 cm (C) 8.00 cm (D) 12.5 cm 2. A car traveling at 10 miles per hour emits about 0.15 kg of carbon monoxide (CO) gas per mile. How many moles of CO are emitted per mile ...
CHEM1102 2014-J-8 June 2014 • Complete the following table
... Ethanol is the weakest acid as its conjugate base is not resonance stabilised. Phenol is a stronger acid as its conjugate base is resonance stabilised – the negative charge can be delocalised into the aromatic ring as shown below. ...
... Ethanol is the weakest acid as its conjugate base is not resonance stabilised. Phenol is a stronger acid as its conjugate base is resonance stabilised – the negative charge can be delocalised into the aromatic ring as shown below. ...
Fatty Acid-Asphaltene Interactions at Oil/Water
... Asphaltenes have been shown to stabilize the water-in-oil emulsions by forming a viscoelastic interfacial film via molecular aggregation at the oil/water interfaces. Natural carboxylic acids and their anions present in crude oil are able to compete with asphaltenes to adsorb at the crude oil/water i ...
... Asphaltenes have been shown to stabilize the water-in-oil emulsions by forming a viscoelastic interfacial film via molecular aggregation at the oil/water interfaces. Natural carboxylic acids and their anions present in crude oil are able to compete with asphaltenes to adsorb at the crude oil/water i ...
Chapter 1 Chemical Bonding and Chemical Structure
... • A phenyl group can be represented a few different ways ...
... • A phenyl group can be represented a few different ways ...
GRIGNARD REAGENTS
... two-headed arrow are resonance forms of the same chemical entity; they differ only the location of electrons, and can be interconverted by the movement of curly arrows. A carbonyl group is a HYBRID of the two resonance forms shown. ...
... two-headed arrow are resonance forms of the same chemical entity; they differ only the location of electrons, and can be interconverted by the movement of curly arrows. A carbonyl group is a HYBRID of the two resonance forms shown. ...
standard sample test
... 14. 2.55 g of sulfuric acid (an acid) is reacted with 1.88 g of sodium hydroxide (a base). The reaction takes place as a solution in water. After the reaction is completed the acidity or basicity of the solution is tested. Which of the following statements is true? (a) The solution was found to be a ...
... 14. 2.55 g of sulfuric acid (an acid) is reacted with 1.88 g of sodium hydroxide (a base). The reaction takes place as a solution in water. After the reaction is completed the acidity or basicity of the solution is tested. Which of the following statements is true? (a) The solution was found to be a ...
Name - rwebbchem
... 1. Would a precipitate form from a reaction of aluminum chloride and sodium hydroxide? If yes, write and balance the equation that illustrates the reaction. ...
... 1. Would a precipitate form from a reaction of aluminum chloride and sodium hydroxide? If yes, write and balance the equation that illustrates the reaction. ...
Second Semester Final Review Guide
... grams of iron, 210.385 grams of oxygen. The compounds molar mass is 159.697 grams/ mole. a. What percentage of the compound is aluminum?____________ b. What percentage of the compound is oxygen?_____________ c. What percentage of the compound is sulfur?_____________ d. What is the empirical formula ...
... grams of iron, 210.385 grams of oxygen. The compounds molar mass is 159.697 grams/ mole. a. What percentage of the compound is aluminum?____________ b. What percentage of the compound is oxygen?_____________ c. What percentage of the compound is sulfur?_____________ d. What is the empirical formula ...
Practice Test 2
... What volume (in liters) of hydrogen gas at STP is released by mixing 5.34 g of iron with 5.00 mL of 1.250 M HCl? (15 pts) ...
... What volume (in liters) of hydrogen gas at STP is released by mixing 5.34 g of iron with 5.00 mL of 1.250 M HCl? (15 pts) ...
45.1 Inter-conversions between the functional groups
... 2. Rate of reactions Many organic reactions are slow. Reactions can be speeded up by using catalysts and a high temperature. Lead to a higher production cost. ...
... 2. Rate of reactions Many organic reactions are slow. Reactions can be speeded up by using catalysts and a high temperature. Lead to a higher production cost. ...
- Thieme Connect
... The amide functionality is ubiquitous, and amides are also versatile educts in organic synthesis; thus there is continuing interest in their synthesis.1 Of particular interest are possibilities of direct conversion of simple substrates into amides which bypass conventional routes, such as reaction o ...
... The amide functionality is ubiquitous, and amides are also versatile educts in organic synthesis; thus there is continuing interest in their synthesis.1 Of particular interest are possibilities of direct conversion of simple substrates into amides which bypass conventional routes, such as reaction o ...
Atoms, Ions, and Molecules File
... Inorganic Nomenclature Inorganic compounds may have traditional names such as H2O - water NH3 - ammonia CaSO4 - gypsum ...Or names related to their chemical formulas such as: NaCl - sodium chloride CO2 - carbon dioxide Na2CO3 - sodium carbonate ...
... Inorganic Nomenclature Inorganic compounds may have traditional names such as H2O - water NH3 - ammonia CaSO4 - gypsum ...Or names related to their chemical formulas such as: NaCl - sodium chloride CO2 - carbon dioxide Na2CO3 - sodium carbonate ...
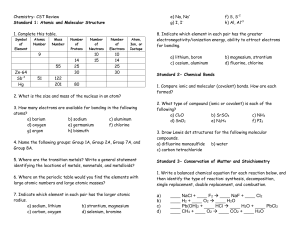
Chemistry- CST Review
... 8. If a sample of gas occupies 6.55 L at 300 °C, what will be its volume at 25 °C if the pressure does not change? 9. A gas at 790 mm Hg and 25 °C occupies a container with an initial volume of 1.20 L. By changing the volume, the pressure of the gas increases to 1500 mm Hg as the temperature is rais ...
... 8. If a sample of gas occupies 6.55 L at 300 °C, what will be its volume at 25 °C if the pressure does not change? 9. A gas at 790 mm Hg and 25 °C occupies a container with an initial volume of 1.20 L. By changing the volume, the pressure of the gas increases to 1500 mm Hg as the temperature is rais ...
cape chemistry unit ii module i
... Sometimes it is necessary to modify the phenol first to make the reaction faster. For example, benzoyl chloride has the formula C6H5COCl. The -COCl group is attached directly to a benzene ring. It is much less reactive than simple acyl chlorides like ethanoyl chloride. In order to get a reasonably q ...
... Sometimes it is necessary to modify the phenol first to make the reaction faster. For example, benzoyl chloride has the formula C6H5COCl. The -COCl group is attached directly to a benzene ring. It is much less reactive than simple acyl chlorides like ethanoyl chloride. In order to get a reasonably q ...
Alcohols and Phenols
... • NaBH4 is not sensitive to moisture and it does not reduce other common functional groups • Lithium aluminum hydride (LiAlH4) is more powerful, less specific, and very reactive with water • Both add the equivalent of “H-” ...
... • NaBH4 is not sensitive to moisture and it does not reduce other common functional groups • Lithium aluminum hydride (LiAlH4) is more powerful, less specific, and very reactive with water • Both add the equivalent of “H-” ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.