Chapter 19 - U of L Class Index
... This is a great way to make 3° alcohols in which two of the alkyl substituents are the same (both derived from the Grignard reagent). The mechanism for this process is completely analogous to that of hydride reaction above. Just replace "H - " with "Et-" and you have it. ...
... This is a great way to make 3° alcohols in which two of the alkyl substituents are the same (both derived from the Grignard reagent). The mechanism for this process is completely analogous to that of hydride reaction above. Just replace "H - " with "Et-" and you have it. ...
Topic 8 specification content - A
... I can explain that ethanol is produced industrially by fermentation of glucose, I can give the conditions for this process and I can explain that the ethanol produced industrially by fermentation is separated by fractional distillation and can then be used as a biofuel ...
... I can explain that ethanol is produced industrially by fermentation of glucose, I can give the conditions for this process and I can explain that the ethanol produced industrially by fermentation is separated by fractional distillation and can then be used as a biofuel ...
Ch 10- Alcohols and Ethers
... – Usually occurs without presence of carbocation – Preferred method of synthesis of Alkyl Bromide from alcohol ...
... – Usually occurs without presence of carbocation – Preferred method of synthesis of Alkyl Bromide from alcohol ...
Mild Conversion of Alcohols to Alkyl Halides Using Halide
... hexanes was sufficient to achieve the separation of nbromobutane, without further purification. The 1-n-butyl-3methylimidazolium cation was recycled in the form of an ionic liquid (presumably bmiHSO4). This transformation is significant from the viewpoint of pollution avoidance. A widely used method ...
... hexanes was sufficient to achieve the separation of nbromobutane, without further purification. The 1-n-butyl-3methylimidazolium cation was recycled in the form of an ionic liquid (presumably bmiHSO4). This transformation is significant from the viewpoint of pollution avoidance. A widely used method ...
RULES OF CHEMICAL NOMENCLATURE I. Elements (periodic
... left of the staircase; non-metals are to the right in periodic table.) (oxygen becomes “oxide,” chlorine becomes “chloride,” sulfur becomes “sulfide,” etc.) C. Most metals have a variable valence and this must be indicated in the name of the compound. 1. The stock system: the valence (+) of the meta ...
... left of the staircase; non-metals are to the right in periodic table.) (oxygen becomes “oxide,” chlorine becomes “chloride,” sulfur becomes “sulfide,” etc.) C. Most metals have a variable valence and this must be indicated in the name of the compound. 1. The stock system: the valence (+) of the meta ...
10.4 Alcohols 10.4 Alcohols
... •Alcohols have the general formula: CnH2n+1OH •The physical properties of alcohols are similar to those of both water and hydrocarbons •The shorter chain alcohols such as methanol and ethanol are similar to water, in general they •have higher boiling points than hydrocarbons but ...
... •Alcohols have the general formula: CnH2n+1OH •The physical properties of alcohols are similar to those of both water and hydrocarbons •The shorter chain alcohols such as methanol and ethanol are similar to water, in general they •have higher boiling points than hydrocarbons but ...
Origins of Medicinal Chemistry
... Why does this OH group react with phenol to form an ester? While the other reacts with acetic acid to give an ester? O ...
... Why does this OH group react with phenol to form an ester? While the other reacts with acetic acid to give an ester? O ...
Document
... Lower members of amines are _____________ with a smell of ___________; while the higher ones are liquids with a distinctive ____________ smell. Amines have ___________ boiling points than less polar compounds due to its ______________ bonding; but they have __________ boiling points than alcohol ...
... Lower members of amines are _____________ with a smell of ___________; while the higher ones are liquids with a distinctive ____________ smell. Amines have ___________ boiling points than less polar compounds due to its ______________ bonding; but they have __________ boiling points than alcohol ...
Bk3BP08EE
... The reaction of chloroethane with ammonia proceeds via a nucleophilic substitution mechanism as shown below: ...
... The reaction of chloroethane with ammonia proceeds via a nucleophilic substitution mechanism as shown below: ...
幻灯片 1
... Hydrocarbons having no double or triple bond functional groups are classified as alkanes or cycloalkanes, depending on whether the carbon atoms of the molecule are arranged only in chains or also in rings. Although these hydrocarbons have no functional groups, they constitute the framework on which ...
... Hydrocarbons having no double or triple bond functional groups are classified as alkanes or cycloalkanes, depending on whether the carbon atoms of the molecule are arranged only in chains or also in rings. Although these hydrocarbons have no functional groups, they constitute the framework on which ...
CHM2045 Final Exam Review, Spring 2017
... 1. The combustion of 40.10 g of a compound which contains only C, H, Cl and O yields 58.57 g of CO2 and 14.98 g of H2O. Another sample of the compound with a mass of 75.00 g is found to contain 22.06 g of Cl. What is the empirical formula of the compound? If the molar mass of the compound is approxi ...
... 1. The combustion of 40.10 g of a compound which contains only C, H, Cl and O yields 58.57 g of CO2 and 14.98 g of H2O. Another sample of the compound with a mass of 75.00 g is found to contain 22.06 g of Cl. What is the empirical formula of the compound? If the molar mass of the compound is approxi ...
Exam only.
... 2. Which of the following (non–cyclic) hydrocarbons has at least two π bonds? a. b. c. d. e. ...
... 2. Which of the following (non–cyclic) hydrocarbons has at least two π bonds? a. b. c. d. e. ...
No Slide Title
... Addition of aqueous sodium hydroxide liberates the free base from its salt C6H5NH3+Cl¯(aq) ...
... Addition of aqueous sodium hydroxide liberates the free base from its salt C6H5NH3+Cl¯(aq) ...
aminepp - Knockhardy
... Addition of aqueous sodium hydroxide liberates the free base from its salt C6H5NH3+Cl¯(aq) ...
... Addition of aqueous sodium hydroxide liberates the free base from its salt C6H5NH3+Cl¯(aq) ...
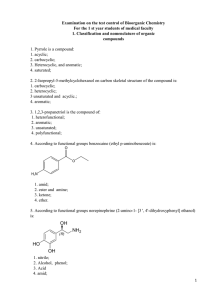
2. 2-Isopropyl-5-methylcyclohexanol on carbon skeletal
... 1. OH- group consisting of carboxylic acid functional groups; 2. hydroxyl group with sp 3-hybrid oxygen; 3. N-H acidic center; 4. OH group bonded directly to the heterocycle; 39. The most strong acidic properties of the compound shown in: 1. acetic acid; 2. propanoic acid; 3. 2-methylpropanoic acid; ...
... 1. OH- group consisting of carboxylic acid functional groups; 2. hydroxyl group with sp 3-hybrid oxygen; 3. N-H acidic center; 4. OH group bonded directly to the heterocycle; 39. The most strong acidic properties of the compound shown in: 1. acetic acid; 2. propanoic acid; 3. 2-methylpropanoic acid; ...
Ch. 09 Alcohols, Ethers, Epoxides
... Substitution and Elimination Reactions of Alcohols • Treatment of alcohols with a strong acid protonates the O converting the bad leaving group ¯OH into H2O, a good leaving group. • The pKa of (ROH2)+ is ~ -2, so protonation of alcohols only occurs with very strong acids. • This makes it possible t ...
... Substitution and Elimination Reactions of Alcohols • Treatment of alcohols with a strong acid protonates the O converting the bad leaving group ¯OH into H2O, a good leaving group. • The pKa of (ROH2)+ is ~ -2, so protonation of alcohols only occurs with very strong acids. • This makes it possible t ...
A Classification of AP Chemistry Reactions
... There are four types of redox with oxygen compounds, classified by the oxidizing agent: oxidizing acids (HNO3 and H2SO4), manganese compounds (MnO4- and MnO2), chromium compounds (Cr2O72-), and hydrogen peroxide (H2O2). Oxidizing acids Oxidizing acids are strong acids with anions that can be reduced ...
... There are four types of redox with oxygen compounds, classified by the oxidizing agent: oxidizing acids (HNO3 and H2SO4), manganese compounds (MnO4- and MnO2), chromium compounds (Cr2O72-), and hydrogen peroxide (H2O2). Oxidizing acids Oxidizing acids are strong acids with anions that can be reduced ...
ELECTROLYTES: NONELECTROLYTES
... Covalent acids form ions in solution, with the help of the water molecules. For instance, hydrogen chloride molecules, which are polar, give up their hydrogens to water, forming chloride ions (Cl-) and hydronium ions (H3O+). ...
... Covalent acids form ions in solution, with the help of the water molecules. For instance, hydrogen chloride molecules, which are polar, give up their hydrogens to water, forming chloride ions (Cl-) and hydronium ions (H3O+). ...
ch5_f08
... • Solid magnesium and aqueous iron(III) chloride • Solid nickel and aqueous sodium chloride ...
... • Solid magnesium and aqueous iron(III) chloride • Solid nickel and aqueous sodium chloride ...
Main Menu - MsReenChemistry
... Alcohols react with carboxylic acids to form esters in a condensation reaction in which water is also produced. This is an equilibrium reaction that must be catalysed by ...
... Alcohols react with carboxylic acids to form esters in a condensation reaction in which water is also produced. This is an equilibrium reaction that must be catalysed by ...
AS Paper 1 Practice Paper 4 - A
... The student decided to use a measuring cylinder to obtain 25.0 cm3 of the supplier’s solution. This was poured into a 250 cm3 graduated flask and the liquid level was made up to the mark with de-ionised water. The student filled a burette with the acid solution. The following results were obtained w ...
... The student decided to use a measuring cylinder to obtain 25.0 cm3 of the supplier’s solution. This was poured into a 250 cm3 graduated flask and the liquid level was made up to the mark with de-ionised water. The student filled a burette with the acid solution. The following results were obtained w ...
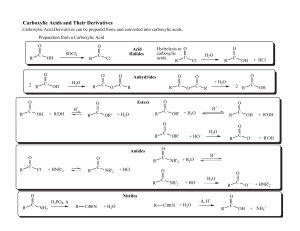
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.