13. Alcohols
... methylated spirits (meths): 90% ethanol and 10% methanol. Denatured alcohol is a useful portable fuel (e.g. for camping stoves) as, unlike LPG, it does not need to be transported in heavy, specialized containers. It is also used as a solvent. 25 of 33 ...
... methylated spirits (meths): 90% ethanol and 10% methanol. Denatured alcohol is a useful portable fuel (e.g. for camping stoves) as, unlike LPG, it does not need to be transported in heavy, specialized containers. It is also used as a solvent. 25 of 33 ...
NOTES CHEMICAL REACTIONS:
... reaction (right side of arrow) • REACTANT: starting substance(s) in a chemical reaction (left side of arrow) • Law of Conservation of Mass must be satisfied! ...
... reaction (right side of arrow) • REACTANT: starting substance(s) in a chemical reaction (left side of arrow) • Law of Conservation of Mass must be satisfied! ...
1A - The changing atom History of the atom • The model of the atom
... Put the 2 before the molecule with the element you are scaling up. You now have the same number of atoms on each side. Step 4 All that remains is to add the state symbols: Iron + Hydrochloric acid Iron (II) chloride + Hydrogen Fe(s) + 2HCl(aq) ...
... Put the 2 before the molecule with the element you are scaling up. You now have the same number of atoms on each side. Step 4 All that remains is to add the state symbols: Iron + Hydrochloric acid Iron (II) chloride + Hydrogen Fe(s) + 2HCl(aq) ...
PDF carboxylic acids
... An examination of Table 1 reveals that acids 1 to 4 are simple monocarboxylic acids i.e. they have only one COOH functional group. Compounds 5 and 6 possess hydroxyl as well as carboxylic groups. In such cases they are named as hydroxyl derivatives of carboxylic acids rather than carboxyl derivativ ...
... An examination of Table 1 reveals that acids 1 to 4 are simple monocarboxylic acids i.e. they have only one COOH functional group. Compounds 5 and 6 possess hydroxyl as well as carboxylic groups. In such cases they are named as hydroxyl derivatives of carboxylic acids rather than carboxyl derivativ ...
S4 Standard Grade Revision Booklet
... When acids are electrolysed, hydrogen gas is produced at the negative electrode. When acids or alkalis are diluted their pH moves towards 7. The test for hydrogen gas is - a lighted splint goes “pop”. Examples of non-metal oxides are:- carbon dioxide (CO2), sulphur dioxide (SO2) and nitrogen dioxide ...
... When acids are electrolysed, hydrogen gas is produced at the negative electrode. When acids or alkalis are diluted their pH moves towards 7. The test for hydrogen gas is - a lighted splint goes “pop”. Examples of non-metal oxides are:- carbon dioxide (CO2), sulphur dioxide (SO2) and nitrogen dioxide ...
Final
... E = hc / λ = hν Write a set of quantum #’s for the last electron added or the first electron removed Write an electron configuration for: elements anomalous elements ions ...
... E = hc / λ = hν Write a set of quantum #’s for the last electron added or the first electron removed Write an electron configuration for: elements anomalous elements ions ...
C:\Users\mrh70950\Documents\My Files\WordPerfect
... d. an intermediate haloalkene is generated; this alkene is the major product if bases weaker than NaNH2 (eg, alkoxides) are used 2. elimination of 2 HX from vicinal,1,2-dihaloalkanes: double dehydrohalogenation using very strong bases a. same comments as above 3. alkylation of terminal alkynes a. Cs ...
... d. an intermediate haloalkene is generated; this alkene is the major product if bases weaker than NaNH2 (eg, alkoxides) are used 2. elimination of 2 HX from vicinal,1,2-dihaloalkanes: double dehydrohalogenation using very strong bases a. same comments as above 3. alkylation of terminal alkynes a. Cs ...
WADE7Lecture10a
... • The LiAlH4 (or LAH) will add two hydrides to the ester to form the primary alkyl halide. • The mechanism is similar to the attack of Grignards on esters. Chapter 10 ...
... • The LiAlH4 (or LAH) will add two hydrides to the ester to form the primary alkyl halide. • The mechanism is similar to the attack of Grignards on esters. Chapter 10 ...
Click Here To File
... aqueous solution to give H+ions. Therefore HF is a weaker acid than HCl. (b) In solid state, PCl5 consists of ions [PCl4]+[PCl6]−. On melting these ions become free to move and hence conducts electricity in the molten state. (c) In SF6, S is sterically protected by six F atoms and hence does not all ...
... aqueous solution to give H+ions. Therefore HF is a weaker acid than HCl. (b) In solid state, PCl5 consists of ions [PCl4]+[PCl6]−. On melting these ions become free to move and hence conducts electricity in the molten state. (c) In SF6, S is sterically protected by six F atoms and hence does not all ...
CP Chemistry Final Review – Chap. 10-19
... 3. Characterize reactions as endothermic or exothermic and create a graph from the sign of H. ChemFinal10-19 ...
... 3. Characterize reactions as endothermic or exothermic and create a graph from the sign of H. ChemFinal10-19 ...
A2 2, Analytical, Transition Metals, Electrochemistry and
... Permission to reproduce all copyright material has been applied for. In some cases, efforts to contact copyright holders may have been unsuccessful and CCEA will be happy to rectify any omissions of acknowledgement in future if notified. ...
... Permission to reproduce all copyright material has been applied for. In some cases, efforts to contact copyright holders may have been unsuccessful and CCEA will be happy to rectify any omissions of acknowledgement in future if notified. ...
Chapter 13…States of Matter
... 4. Calculate the amount of energy required to heat a 150 g chunk of aluminum from 20C to 40C. (Cp of aluminum = 0.220 cal/gC) H=mCpT (150g)(.22)(20) = 660 cal Chapters 17& 18…Reaction Rates & Equilibrium Define: 1. Equilibrium: the reaction occurs simultaneously in both directions. 2. Activate ...
... 4. Calculate the amount of energy required to heat a 150 g chunk of aluminum from 20C to 40C. (Cp of aluminum = 0.220 cal/gC) H=mCpT (150g)(.22)(20) = 660 cal Chapters 17& 18…Reaction Rates & Equilibrium Define: 1. Equilibrium: the reaction occurs simultaneously in both directions. 2. Activate ...
chemistry 232 elementary organic chemistry ii
... Acid-Base Protonation/Deprotonation Reactions (Ch. 7 & 10) Protonation/Deprotonation of Alcohols Deprotonation of Alkynes Acid-Catalyzed Rearrangements (Ch. 9 & 10) via SN1 Reaction Pathway (step-wise) The Pinacol Rearrangement Nucleophilic Addition to Carbonyl Compounds (Ch. 15, 16, & 17) Organomet ...
... Acid-Base Protonation/Deprotonation Reactions (Ch. 7 & 10) Protonation/Deprotonation of Alcohols Deprotonation of Alkynes Acid-Catalyzed Rearrangements (Ch. 9 & 10) via SN1 Reaction Pathway (step-wise) The Pinacol Rearrangement Nucleophilic Addition to Carbonyl Compounds (Ch. 15, 16, & 17) Organomet ...
+ Br2, FeBr3 + Br2, FeBr3
... (b) What is the reactive electrophile in the above reaction? NO2+, nitronium ion. (c) If we used only pure (fuming) sulfuric acid, what would be the product(s)? mostly sulfonation of Cl benzene, both o and p, because SO3H+ becomes the superelectrophile and there is not as much protons for the dehydr ...
... (b) What is the reactive electrophile in the above reaction? NO2+, nitronium ion. (c) If we used only pure (fuming) sulfuric acid, what would be the product(s)? mostly sulfonation of Cl benzene, both o and p, because SO3H+ becomes the superelectrophile and there is not as much protons for the dehydr ...
CHEM_2nd_Semester_Final_R eview
... 27. Describe the dissociation (ionization) of strong acids and bases versus weak acids and bases. 28. List the 6 strong acids and state the rule for strong bases. 29. What are the pH values for acids? Bases? 30. What is more acidic, a solution with a pH of 2 or 5? What is more basic, a solution with ...
... 27. Describe the dissociation (ionization) of strong acids and bases versus weak acids and bases. 28. List the 6 strong acids and state the rule for strong bases. 29. What are the pH values for acids? Bases? 30. What is more acidic, a solution with a pH of 2 or 5? What is more basic, a solution with ...
Chemistry 2nd Semester Final Exam Review Chemical Bonds Give
... 27. Describe the dissociation (ionization) of strong acids and bases versus weak acids and bases. 28. List the 6 strong acids and state the rule for strong bases. 29. What are the pH values for acids? Bases? 30. What is more acidic, a solution with a pH of 2 or 5? What is more basic, a solution with ...
... 27. Describe the dissociation (ionization) of strong acids and bases versus weak acids and bases. 28. List the 6 strong acids and state the rule for strong bases. 29. What are the pH values for acids? Bases? 30. What is more acidic, a solution with a pH of 2 or 5? What is more basic, a solution with ...
2nd Semester Final Review
... 27. Describe the dissociation (ionization) of strong acids and bases versus weak acids and bases. 28. List the 6 strong acids and state the rule for strong bases. 29. What are the pH values for acids? Bases? 30. What is more acidic, a solution with a pH of 2 or 5? What is more basic, a solution with ...
... 27. Describe the dissociation (ionization) of strong acids and bases versus weak acids and bases. 28. List the 6 strong acids and state the rule for strong bases. 29. What are the pH values for acids? Bases? 30. What is more acidic, a solution with a pH of 2 or 5? What is more basic, a solution with ...
Synthesis Reactions occur when two of more reactants combine to
... All chlorides, bromides, and iodides except mercury, silver, and lead(II). All sulfates except mercury, silver, lead(II), calcium, barium, and strontium. Mainly Water Insoluble (will NOT dissolve into ions in water; will remain as a solid “precipitate”) All carbonates, phosphates, and sulfites excep ...
... All chlorides, bromides, and iodides except mercury, silver, and lead(II). All sulfates except mercury, silver, lead(II), calcium, barium, and strontium. Mainly Water Insoluble (will NOT dissolve into ions in water; will remain as a solid “precipitate”) All carbonates, phosphates, and sulfites excep ...
Chemical Reactions and Equations
... Starting with elements that only occur in one substance on each side of the equation, make sure that each side of the equation has an equal # of that element. Proceed with all elements. Remember that changing the # of one element may alter elements that have already been ...
... Starting with elements that only occur in one substance on each side of the equation, make sure that each side of the equation has an equal # of that element. Proceed with all elements. Remember that changing the # of one element may alter elements that have already been ...
Carboxylic acids from primary alcohols and aldehydes by a
... quantitative yields. Benzylic (entries 1, 4–7 and 10), aliphatic (entries 2 and 12) as well as homobenzylic (entries 3, 8, 9, and 11) alcohols oxidized smoothly in a short amount of time. Electron-poor (entries 5 and 6) as well as electron-rich (entry 4 and 7) benzyl alcohols were oxidized without a ...
... quantitative yields. Benzylic (entries 1, 4–7 and 10), aliphatic (entries 2 and 12) as well as homobenzylic (entries 3, 8, 9, and 11) alcohols oxidized smoothly in a short amount of time. Electron-poor (entries 5 and 6) as well as electron-rich (entry 4 and 7) benzyl alcohols were oxidized without a ...
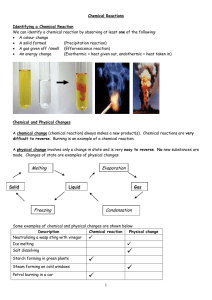
Chemical Reactions
... A word equation is a simple way of showing a chemical reaction. The reactants are shown on the left hand side and the products are shown on the right hand side. The reactants are separated from each other by a plus sign and the products are separated from each other by a plus sign. There should be a ...
... A word equation is a simple way of showing a chemical reaction. The reactants are shown on the left hand side and the products are shown on the right hand side. The reactants are separated from each other by a plus sign and the products are separated from each other by a plus sign. There should be a ...
Prof. Kamakaka`s Lecture 1 Notes
... You can measure pKa of an acid by performing a titration curve. Take and acid, measure pH--- say 1.0 Add a strong base such as NaOH and after each addition of NaOH, you measure pH The pH at mid-point of the titration is the pKa for that acid at that temperature pH=pKa=50% ionized acid It is possibl ...
... You can measure pKa of an acid by performing a titration curve. Take and acid, measure pH--- say 1.0 Add a strong base such as NaOH and after each addition of NaOH, you measure pH The pH at mid-point of the titration is the pKa for that acid at that temperature pH=pKa=50% ionized acid It is possibl ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.