Nuggets of Knowledge for Chapter 10 – Alkyl Halides II Chem 2310 I
... ◦ NaNH2 has a conjugate acid pKa of 36, while tert-butoxide has a conjugate acid pKa of 18. This makes it 1018 more basic, which is a whole different level of “strong base.” ◦ Using these much stronger bases, vinyl halides can form alkynes, and aryl halides can form benzynes. Alkynes are stable prod ...
... ◦ NaNH2 has a conjugate acid pKa of 36, while tert-butoxide has a conjugate acid pKa of 18. This makes it 1018 more basic, which is a whole different level of “strong base.” ◦ Using these much stronger bases, vinyl halides can form alkynes, and aryl halides can form benzynes. Alkynes are stable prod ...
File
... reactions. _____ 15. Carbon dioxide and water are the products of the combustion of hexane (C6H14). _____ 16. A nonmetal can replace another nonmetal from a compound in a single-replacement reaction. ...
... reactions. _____ 15. Carbon dioxide and water are the products of the combustion of hexane (C6H14). _____ 16. A nonmetal can replace another nonmetal from a compound in a single-replacement reaction. ...
Chemical Equations TrackStar Assignment
... 2. What is a reversible reaction and how is it indicated? 3. Write the reaction for a silver spoon tarnishing. What type of reaction is this? 4. Write the reaction for the burning of Methane gas (the gas used in Chemistry lab). What type of reaction is this? 5. Write the reaction of the neutralizati ...
... 2. What is a reversible reaction and how is it indicated? 3. Write the reaction for a silver spoon tarnishing. What type of reaction is this? 4. Write the reaction for the burning of Methane gas (the gas used in Chemistry lab). What type of reaction is this? 5. Write the reaction of the neutralizati ...
Oxidation Reduction PowerPoint
... to form one or more oxygen-containing compounds, often including water. ...
... to form one or more oxygen-containing compounds, often including water. ...
Fall.2008.Week9.Lesson.1 - reich
... • Combustion means burning and fire. What two things does fire require? O2 and something to burn. We normally burn hydrocarbons (Hydro=H, Carbon = C therefore stuff made up of H and C). • The products are always CO2 and H2O. • Methane and Oxygen burn write the equation. • ___CH4+ ___O2 ___ CO2 + _ ...
... • Combustion means burning and fire. What two things does fire require? O2 and something to burn. We normally burn hydrocarbons (Hydro=H, Carbon = C therefore stuff made up of H and C). • The products are always CO2 and H2O. • Methane and Oxygen burn write the equation. • ___CH4+ ___O2 ___ CO2 + _ ...
L1 - Amines
... • Organic bases. • Generally have strong, unpleasant odors. • Are found extensively in biological systems. • Found in both controlled and medicinal compounds 1,5 - diaminopentane H2N – CH2-CH2-CH2-CH2-CH2 – NH2 Cadaverine ...
... • Organic bases. • Generally have strong, unpleasant odors. • Are found extensively in biological systems. • Found in both controlled and medicinal compounds 1,5 - diaminopentane H2N – CH2-CH2-CH2-CH2-CH2 – NH2 Cadaverine ...
Pre-Board Examination2016 Class : XII MM: 70 Subject : Chemistry
... (b) Out of C and CO, which is a better reducing agent at the lower temperature range in the blast furnace to extract iron from the oxide ore? 13. (a) Which metal in the first transition series (3d series) exhibits +1 oxidation state most frequently and why? (b) Which of the following cations are col ...
... (b) Out of C and CO, which is a better reducing agent at the lower temperature range in the blast furnace to extract iron from the oxide ore? 13. (a) Which metal in the first transition series (3d series) exhibits +1 oxidation state most frequently and why? (b) Which of the following cations are col ...
Objectives - Dixie State University
... 1. Give the definitions of Lewis acids and bases and Bronsted-Lowry acids and bases, and explain the relationship between these definitions. 2. Explain happens in an association, dissociation, and displacement reaction, and correctly label reactions as one of these. 3. Identify the nucleophile and e ...
... 1. Give the definitions of Lewis acids and bases and Bronsted-Lowry acids and bases, and explain the relationship between these definitions. 2. Explain happens in an association, dissociation, and displacement reaction, and correctly label reactions as one of these. 3. Identify the nucleophile and e ...
New AQA C3 revison guide
... Water is the most common chemical on the planet. One of its most important properties is as a solvent. It is an extremely good solvent for a wide variety of chemicals, and for this reason it is difficult to obtain pure. Sea water is formed when rainwater runs through rocks & soils, dissolving out mi ...
... Water is the most common chemical on the planet. One of its most important properties is as a solvent. It is an extremely good solvent for a wide variety of chemicals, and for this reason it is difficult to obtain pure. Sea water is formed when rainwater runs through rocks & soils, dissolving out mi ...
ppt
... The ether oxygen is sp3-hybridized and tetrahedral. In general, the C-O bonds of ethers have low reactivity. 16.3: Physical Properties of Ethers the O-H group of alcohols act as both an H-bond donor (Lewis acid) and H-bond acceptor (Lewis base). Ethers are only H-bond acceptors (Lewis base) 16.4: Cr ...
... The ether oxygen is sp3-hybridized and tetrahedral. In general, the C-O bonds of ethers have low reactivity. 16.3: Physical Properties of Ethers the O-H group of alcohols act as both an H-bond donor (Lewis acid) and H-bond acceptor (Lewis base). Ethers are only H-bond acceptors (Lewis base) 16.4: Cr ...
11U CHEMISTRY EXAM REVIEW QUESTIONS June 2010
... tin (s) + potassium iodide (aq) fluorine (aq) + sodium chloride (aq) sodium (s) + calcium sulfate (aq) ...
... tin (s) + potassium iodide (aq) fluorine (aq) + sodium chloride (aq) sodium (s) + calcium sulfate (aq) ...
SCH3U Exam Review 1 11U CHEMISTRY EXAM
... tin (s) + potassium iodide (aq) fluorine (aq) + sodium chloride (aq) sodium (s) + calcium sulfate (aq) ...
... tin (s) + potassium iodide (aq) fluorine (aq) + sodium chloride (aq) sodium (s) + calcium sulfate (aq) ...
Examples
... More active metals will replace less active metals from their compound in a solution A less active element will have no reaction when added to a more active element! Active metals replace hydrogen in water Active metals replace hydrogen in acids ...
... More active metals will replace less active metals from their compound in a solution A less active element will have no reaction when added to a more active element! Active metals replace hydrogen in water Active metals replace hydrogen in acids ...
F Practice Test #2 Solutions
... A) No precipitate forms. B) NiS will precipitate from solution. C) No reaction will occur. D) Both KNO3 and NiS precipitate from solution. E) KNO3 will precipitate from solution. 10. Which of the following statements concerning equilibrium is not true? A) The equilibrium constant is independent of t ...
... A) No precipitate forms. B) NiS will precipitate from solution. C) No reaction will occur. D) Both KNO3 and NiS precipitate from solution. E) KNO3 will precipitate from solution. 10. Which of the following statements concerning equilibrium is not true? A) The equilibrium constant is independent of t ...
Final Review Answers
... a. an increase in temperature (a) more collisions between particles causing an increase in dissolving particles (b) decreases solubility, as T increases more dissolved gas escapes b. shaking, agitation (a) more collisions between particles causing an increase in dissolving particles (b) increases so ...
... a. an increase in temperature (a) more collisions between particles causing an increase in dissolving particles (b) decreases solubility, as T increases more dissolved gas escapes b. shaking, agitation (a) more collisions between particles causing an increase in dissolving particles (b) increases so ...
Types of Chemical Reactions
... The ability of one metal to displace another depends on their relative ease of oxidation—a more active metal (one that is more easily oxidized) displaces a less active metal. In the first reaction above, lead is more active than copper. The relative activities of metals can be tabulated in an activi ...
... The ability of one metal to displace another depends on their relative ease of oxidation—a more active metal (one that is more easily oxidized) displaces a less active metal. In the first reaction above, lead is more active than copper. The relative activities of metals can be tabulated in an activi ...
IOSR Journal of Applied Chemistry (IOSR-JAC) e-ISSN: 2278-5736.
... On exposure of CdS particles, an electron from the valence band is excited to conduction band (e-CB) leaving behind a hole in the valence band (h +VB). This hole creates electron deficiency (h +s) at semiconductor surface [14], which can dissociate water into hydroxyl radical and a proton [15]. This ...
... On exposure of CdS particles, an electron from the valence band is excited to conduction band (e-CB) leaving behind a hole in the valence band (h +VB). This hole creates electron deficiency (h +s) at semiconductor surface [14], which can dissociate water into hydroxyl radical and a proton [15]. This ...
7-1 EXPERIMENT 7: Reduction of Carbonyl Compounds – Achiral
... In this experiment, a ketoester will be reduced with and without chiral reagents. The milder reducing agent, NaBH4, will be used to produce the racemic mixture since LiAlH4 would cause reductions at both carbonyl groups (a ketone and an ester) of the starting material. This reaction involves the for ...
... In this experiment, a ketoester will be reduced with and without chiral reagents. The milder reducing agent, NaBH4, will be used to produce the racemic mixture since LiAlH4 would cause reductions at both carbonyl groups (a ketone and an ester) of the starting material. This reaction involves the for ...
Chem 3.5 #6 Amines
... Write an equation which shows a reaction that could be used to produce CH3CH2NH2 from CH3CH2Cl. ...
... Write an equation which shows a reaction that could be used to produce CH3CH2NH2 from CH3CH2Cl. ...
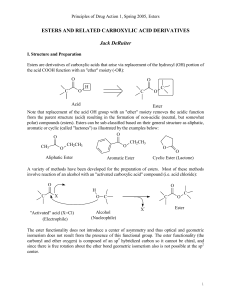
esters and related carboxylic acid derivatives
... There are a number of other functional groups that are derivatives of traditional esters or "esterlike" including amides and amide derivatives (carbamates, ureas, etc.), carbonates, anhydrides and phosphate esters. The amide and amide derivatives are discussed in a separate chapter: O ...
... There are a number of other functional groups that are derivatives of traditional esters or "esterlike" including amides and amide derivatives (carbamates, ureas, etc.), carbonates, anhydrides and phosphate esters. The amide and amide derivatives are discussed in a separate chapter: O ...
Chapter 24. Amines
... is chiral (in principle but not in practice): the lone pair of electrons is the fourth substituent Most amines that have 3 different substituents on N are not resolved because the molecules interconvert by pyramidal inversion ...
... is chiral (in principle but not in practice): the lone pair of electrons is the fourth substituent Most amines that have 3 different substituents on N are not resolved because the molecules interconvert by pyramidal inversion ...
Chapter 24. Amines - Houston Community College System
... is chiral (in principle but not in practice): the lone pair of electrons is the fourth substituent Most amines that have 3 different substituents on N are not resolved because the molecules interconvert by pyramidal inversion ...
... is chiral (in principle but not in practice): the lone pair of electrons is the fourth substituent Most amines that have 3 different substituents on N are not resolved because the molecules interconvert by pyramidal inversion ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.