Notes
... Two compounds react to form two new compounds. All double replacement reactions must have a "driving force" that removes a pair of ions from solution. Ions keep their same charges as reactants and products. Formation of a precipitate: A precipitate is an insoluble substance formed by the reaction of ...
... Two compounds react to form two new compounds. All double replacement reactions must have a "driving force" that removes a pair of ions from solution. Ions keep their same charges as reactants and products. Formation of a precipitate: A precipitate is an insoluble substance formed by the reaction of ...
Final Exam Review- no solutions
... a. Hg(l) + MnSO4(aq) HgSO4(s) + Mn(s) b. 2 Ag (s) + H2SO4(aq) Ag2SO4(aq) + H2(g) c. Ca(s) + 2 H2O(l) Ca(OH)2(aq) + H2(g) 11. Titration reveals that 11.6 mL of 3.0 M sulfuric acid are required to neutralize the sodium hydroxide in 25.00 mL of NaOH solution. What is the molarity of the NaOH solu ...
... a. Hg(l) + MnSO4(aq) HgSO4(s) + Mn(s) b. 2 Ag (s) + H2SO4(aq) Ag2SO4(aq) + H2(g) c. Ca(s) + 2 H2O(l) Ca(OH)2(aq) + H2(g) 11. Titration reveals that 11.6 mL of 3.0 M sulfuric acid are required to neutralize the sodium hydroxide in 25.00 mL of NaOH solution. What is the molarity of the NaOH solu ...
CHAPTER 20 CARBOXYLIC ACIDS Organic Chemistry
... 20.6 Preparation of Carboxylic Acids 1- Oxidation of arenes Oxidation of a substituted alkylbenzene with KMnO4 or Na2Cr2O7 or H2Cr2O7 gives a substituted benzoic acid (see Section 16.10) 1° and 2° alkyl groups can be oxidized, but tertiary groups are not ( the alkylbenzene must contain at least o ...
... 20.6 Preparation of Carboxylic Acids 1- Oxidation of arenes Oxidation of a substituted alkylbenzene with KMnO4 or Na2Cr2O7 or H2Cr2O7 gives a substituted benzoic acid (see Section 16.10) 1° and 2° alkyl groups can be oxidized, but tertiary groups are not ( the alkylbenzene must contain at least o ...
Chapter 8 Alkenes and Alkynes II: Addition Reactions Alkenes are
... A cyclic intermediate results when an alkene reacts with MnO4A syn addition of the oxygens occurs resulting in a cis-diol after the oxygen-metal bonds are cleaved with the base. ...
... A cyclic intermediate results when an alkene reacts with MnO4A syn addition of the oxygens occurs resulting in a cis-diol after the oxygen-metal bonds are cleaved with the base. ...
Organic Chemistry Structures of Organic Compounds
... The overall reaction with Cl2 faster than Br2 If excess halogen, e.g., Cl2, more chlorination ie. CH4 → ...
... The overall reaction with Cl2 faster than Br2 If excess halogen, e.g., Cl2, more chlorination ie. CH4 → ...
Reactions of Alcohols
... The ZnCl2 coordinates to the hydroxyl oxygen, and this generates a far superior leaving group. Primary alcohols react in a similar fashion except the free cation is not generated, and the substitution is of S N2 ...
... The ZnCl2 coordinates to the hydroxyl oxygen, and this generates a far superior leaving group. Primary alcohols react in a similar fashion except the free cation is not generated, and the substitution is of S N2 ...
Pre-Lab Questions - Dr. Hornbuckle`s Home Page
... V) In the absence of pinane, what is formed (50% of the product) from the decomposition of pinane hydroperoxide? VI) Yields of 33% 3-chloropyridine has been formed from vapors of chloroform and pyrrole passing through a _________________ at temp. of __________. VII) 1,3,5-tribromobenzene reverts to ...
... V) In the absence of pinane, what is formed (50% of the product) from the decomposition of pinane hydroperoxide? VI) Yields of 33% 3-chloropyridine has been formed from vapors of chloroform and pyrrole passing through a _________________ at temp. of __________. VII) 1,3,5-tribromobenzene reverts to ...
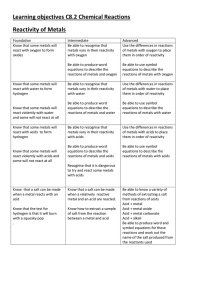
Learning objectives C8.2 Chemical Reactions Reactivity of Metals
... methods of extracting a salt from reactions of acids Acid + metal Acid + metal oxide Acid + metal carbonate Acid + alkali Be able to produce word and symbol equations for these reactions and work out the name of the salt produced from the reactants used ...
... methods of extracting a salt from reactions of acids Acid + metal Acid + metal oxide Acid + metal carbonate Acid + alkali Be able to produce word and symbol equations for these reactions and work out the name of the salt produced from the reactants used ...
Basic Organic Chemistry Laboratory Course
... darkens. This reaction is not specific to alkenes, as for example oxygen containing compounds form soluble oxonium salts. ...
... darkens. This reaction is not specific to alkenes, as for example oxygen containing compounds form soluble oxonium salts. ...
Chapter-16B
... Characteristic Reactions • in the general reaction, we showed the nucleophile as an anion; this need not be the case • neutral molecules such as water, alcohols, ammonia, and amines can also serve as nucleophiles • in the general reaction, we showed the leaving group as an anion to illustrate an im ...
... Characteristic Reactions • in the general reaction, we showed the nucleophile as an anion; this need not be the case • neutral molecules such as water, alcohols, ammonia, and amines can also serve as nucleophiles • in the general reaction, we showed the leaving group as an anion to illustrate an im ...
Chemical Reactions
... b. First balance the atoms of elements that only appear once on each side of the equation. c. Next balance all other atoms. d. Balance H and O atoms LAST. ...
... b. First balance the atoms of elements that only appear once on each side of the equation. c. Next balance all other atoms. d. Balance H and O atoms LAST. ...
Nitrogen and its compounds - kcpe-kcse
... The drying agent used for ammonia is quick lime. Other drying agents such as concentrated sulphuric acid or phosphorus (V) oxide or fused calcium chloride cannot dry an alkaline gas like ammonia. Sulphuric acid and phosphorus (V) oxide are both acidic. They react with ammonia, forming their respecti ...
... The drying agent used for ammonia is quick lime. Other drying agents such as concentrated sulphuric acid or phosphorus (V) oxide or fused calcium chloride cannot dry an alkaline gas like ammonia. Sulphuric acid and phosphorus (V) oxide are both acidic. They react with ammonia, forming their respecti ...
View Article - Asian Journal of Chemistry
... sodium hydroxide solution. Liquid bromine ( 4.2 mL, 80 mmol) was added dropwise in 2 h when the mixture maintained at 100 - 130 ºC heated by oil bath, excess time was prolonged for reaction until the colour of the reaction mixture turned to light yellow, after that the oil bath was removed and ethyl ...
... sodium hydroxide solution. Liquid bromine ( 4.2 mL, 80 mmol) was added dropwise in 2 h when the mixture maintained at 100 - 130 ºC heated by oil bath, excess time was prolonged for reaction until the colour of the reaction mixture turned to light yellow, after that the oil bath was removed and ethyl ...
Chemistry - SchoolNotes.com
... CaCO3(s) CaO(s) + CO2(g). What mass of lime can be produced from 1.5 x 103 kg of limestone? 8.4 x 105 g 23) If 30.0 g of sodium chloride reacts with excess sulfuric acid, how many grams of hydrogen chloride are produced? NaCl + H2SO4 --- HCl + Na2SO4 18.7 g HCl Stoichiometry – Limiting Reagent 24 ...
... CaCO3(s) CaO(s) + CO2(g). What mass of lime can be produced from 1.5 x 103 kg of limestone? 8.4 x 105 g 23) If 30.0 g of sodium chloride reacts with excess sulfuric acid, how many grams of hydrogen chloride are produced? NaCl + H2SO4 --- HCl + Na2SO4 18.7 g HCl Stoichiometry – Limiting Reagent 24 ...
1. This is a question about trends in chemistry In
... CH3CH2*CH(OH)CH2CH2CH3 and can exist as two different optical isomers (enantiomers), which are nonsuperimposable mirror images. A molecule which contains just one chiral centre will always exist in two enantiomeric forms. However, some molecules with two or more chiral centres can be achiral, i.e. t ...
... CH3CH2*CH(OH)CH2CH2CH3 and can exist as two different optical isomers (enantiomers), which are nonsuperimposable mirror images. A molecule which contains just one chiral centre will always exist in two enantiomeric forms. However, some molecules with two or more chiral centres can be achiral, i.e. t ...
Oxidation-Reduction Reactions
... • Planck’s Quantization of Energy (1900) According to Max Planck, when solids are heated, they emit electromagnetic radiation over a wide range of wavelengths. He proposed that an atom could emit or absorb energy only in discrete quantities, like small packages, and quantum is the smallest quantity ...
... • Planck’s Quantization of Energy (1900) According to Max Planck, when solids are heated, they emit electromagnetic radiation over a wide range of wavelengths. He proposed that an atom could emit or absorb energy only in discrete quantities, like small packages, and quantum is the smallest quantity ...
11_Phenolics - WordPress.com
... Cannabinoids, the active constituents of Cannabis. Cresols from coal tar and creosote. Guaiacol (2-methoxyphenol) from roasted coffee, whisky, and smoke that has a smoky flavor. Salicylic acid, a plant hormone and analgesic, antipyretic, and antiinflammatory drug, precursor compound to Aspirin. Aspi ...
... Cannabinoids, the active constituents of Cannabis. Cresols from coal tar and creosote. Guaiacol (2-methoxyphenol) from roasted coffee, whisky, and smoke that has a smoky flavor. Salicylic acid, a plant hormone and analgesic, antipyretic, and antiinflammatory drug, precursor compound to Aspirin. Aspi ...
organic functional group analysis
... 3. Solubility in 5% HCl: Diagnosis of a High Molecular Weight Organic Base Add ~1 mL of 5% HCI dropwise, with agitation, to ~0.5 mL drops of the known compound. High molecular weight amines will be soluble in 5% HCl because of the formation of the ammonium chloride salt High molecular weight neutral ...
... 3. Solubility in 5% HCl: Diagnosis of a High Molecular Weight Organic Base Add ~1 mL of 5% HCI dropwise, with agitation, to ~0.5 mL drops of the known compound. High molecular weight amines will be soluble in 5% HCl because of the formation of the ammonium chloride salt High molecular weight neutral ...
Orbitals
... Hydride Reagents: Alcohol Formation Grignard reagent reacts with aldehyde or ketone C=O to give tetrahedral magnesium alkoxide intermediate Intermediate is hydrolyzed to produce neutral alcohol Grignard reagent reacts like a carbon anion, or carbanion • Reaction mechanism involves radicals but can b ...
... Hydride Reagents: Alcohol Formation Grignard reagent reacts with aldehyde or ketone C=O to give tetrahedral magnesium alkoxide intermediate Intermediate is hydrolyzed to produce neutral alcohol Grignard reagent reacts like a carbon anion, or carbanion • Reaction mechanism involves radicals but can b ...
General Chemistry Review Problems
... w. aluminum bromide reacts with potassium sulfate to yield potassium bromide and aluminum sulfate. x. methane combusts with oxygen to form carbon dioxide and water. y. acetic acid reacts with copper to produce copper(II) acetate and hydrogen gas. z. aluminum reacts with iron(III) oxide to produce al ...
... w. aluminum bromide reacts with potassium sulfate to yield potassium bromide and aluminum sulfate. x. methane combusts with oxygen to form carbon dioxide and water. y. acetic acid reacts with copper to produce copper(II) acetate and hydrogen gas. z. aluminum reacts with iron(III) oxide to produce al ...
158KB - NZQA
... Whereas HCOOH is a weak acid, it does not readily dissociate in water. HCOOH(aq) + H2O() H3O+(aq) + HCOO–(aq) [H3O+] = 0.00398mol L–1 In the resulting solutions, HCl has a higher concentration of H 3O+, and therefore a lower pH (1) than HCOOH, which has a lower concentration of H3O+, and therefor ...
... Whereas HCOOH is a weak acid, it does not readily dissociate in water. HCOOH(aq) + H2O() H3O+(aq) + HCOO–(aq) [H3O+] = 0.00398mol L–1 In the resulting solutions, HCl has a higher concentration of H 3O+, and therefore a lower pH (1) than HCOOH, which has a lower concentration of H3O+, and therefor ...
Functional groups and homologous series
... Using the scheme on the left which summarizes the organic reactions in the text, it is possible to devise reaction pathways. These should involve no more than two steps and should include the reagents, conditions and relevant equations. ...
... Using the scheme on the left which summarizes the organic reactions in the text, it is possible to devise reaction pathways. These should involve no more than two steps and should include the reagents, conditions and relevant equations. ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.