untitled file - Blue Earth Area Schools
... either lactic acid or ethanol and CO2 • Then NAD+ is recycled and glycolysis can ...
... either lactic acid or ethanol and CO2 • Then NAD+ is recycled and glycolysis can ...
Thursday, September 4 Bell Work: Predict the outcome of slight
... Lipids are the one class of large biological molecules that does not include true polymers The unifying feature of lipids is that they mix poorly, if at all, with water Lipids are hydrophobic because they consist mostly of hydrocarbons, which form nonpolar covalent bonds The most biological ...
... Lipids are the one class of large biological molecules that does not include true polymers The unifying feature of lipids is that they mix poorly, if at all, with water Lipids are hydrophobic because they consist mostly of hydrocarbons, which form nonpolar covalent bonds The most biological ...
The Chemical Basis of Life Atoms, Molecules, Ions, and Bonds
... (three hydrogens bonded to the last carbon). You can remember this by thinking that each carbon is “saturated” with hydrogen. ...
... (three hydrogens bonded to the last carbon). You can remember this by thinking that each carbon is “saturated” with hydrogen. ...
DiagnosticTest
... c. proteins amino acids d. proteins fatty acids 4. Which process occurs at I? a. Photosynthesis b. Respiration c. Feeding d. Excretion 5. Respiration occurs only at a. I only b. II only c. II and III only d. II and IV only 6. Which chemical change takes place in green plants, but not in mammals? a. ...
... c. proteins amino acids d. proteins fatty acids 4. Which process occurs at I? a. Photosynthesis b. Respiration c. Feeding d. Excretion 5. Respiration occurs only at a. I only b. II only c. II and III only d. II and IV only 6. Which chemical change takes place in green plants, but not in mammals? a. ...
Lucky Lady Slots Online - How Does Shot Roulette Work
... 3. What element is found in proteins but not in carbohydrates and lipids? ________ 4.Proteins have many functions what are the two classes of proteins? ...
... 3. What element is found in proteins but not in carbohydrates and lipids? ________ 4.Proteins have many functions what are the two classes of proteins? ...
10/31
... Nitrogen required for proteins, nucleic acids and other important cell constituents Most microorganisms are incapable of using nitrogen gas as a nitrogen source They must therefore incorporate either ammonia (NH3) or nitrate (NO3) ...
... Nitrogen required for proteins, nucleic acids and other important cell constituents Most microorganisms are incapable of using nitrogen gas as a nitrogen source They must therefore incorporate either ammonia (NH3) or nitrate (NO3) ...
Nutritional requirements
... It has no nutritive value in man but the presence of cellulose in food helps to avoid constipation. Excess glucose is converted into ...
... It has no nutritive value in man but the presence of cellulose in food helps to avoid constipation. Excess glucose is converted into ...
Nutritional Requirements
... It has no nutritive value in man but the presence of cellulose in food helps to avoid constipation. Excess glucose is converted into ...
... It has no nutritive value in man but the presence of cellulose in food helps to avoid constipation. Excess glucose is converted into ...
Respiration
... digested into organic molecules which are transported by the bloodstream to our cells where these substances can be oxidized during cellular respiration and their energy converted into ATP energy. Alternately, the organic molecules, products of digestion, can be used by the cell for the biosynthesis ...
... digested into organic molecules which are transported by the bloodstream to our cells where these substances can be oxidized during cellular respiration and their energy converted into ATP energy. Alternately, the organic molecules, products of digestion, can be used by the cell for the biosynthesis ...
Stabilization of Low Affinity Protein-Protein Interactions by
... The introduction of new chemical functionalities into proteins represents a promising approach for investigating and manipulating diverse biological processes. Among a number of different approaches, the expansion of the genetic code has emerged as an eminent tool for in vivo site-specific incorpora ...
... The introduction of new chemical functionalities into proteins represents a promising approach for investigating and manipulating diverse biological processes. Among a number of different approaches, the expansion of the genetic code has emerged as an eminent tool for in vivo site-specific incorpora ...
Chemical reactions take place inside cells.
... carbohydrates, simple lipids are made of atoms of carbon, oxygen, and hydrogen and can be used by cells for energy and for making structures. However, the atoms in all lipids are arranged differently from the atoms in carbohydrates. Many common lipids consist of a molecule called glycerol bonded to ...
... carbohydrates, simple lipids are made of atoms of carbon, oxygen, and hydrogen and can be used by cells for energy and for making structures. However, the atoms in all lipids are arranged differently from the atoms in carbohydrates. Many common lipids consist of a molecule called glycerol bonded to ...
1 Review I: Protein Structure Amino Acids Amino Acids (contd
... Repetitive secondary structure 3.6 residues per turn; pitch (rise per turn) = 5.4 Å C′=O of i forms H bonds with NH of residue i+4 Intra-strand H bonding C′=O groups are parallel to the axis; side chains point away from the axis All NH and C′O are H-bonded, except first NH and last C′O ...
... Repetitive secondary structure 3.6 residues per turn; pitch (rise per turn) = 5.4 Å C′=O of i forms H bonds with NH of residue i+4 Intra-strand H bonding C′=O groups are parallel to the axis; side chains point away from the axis All NH and C′O are H-bonded, except first NH and last C′O ...
Protein Synthesis and Function: Chapter 3
... Consists of a stack of flattened sacs called cisternae Closely associated with ER Transitional vesicles from the ER containing proteins go to the Golgi apparatus for modification and maturation Condensing vesicles transport proteins to organelles or secretory proteins to the outside ...
... Consists of a stack of flattened sacs called cisternae Closely associated with ER Transitional vesicles from the ER containing proteins go to the Golgi apparatus for modification and maturation Condensing vesicles transport proteins to organelles or secretory proteins to the outside ...
Biomolecules
... All biologically known protein are polymers of a set of twenty known amino acids. All biologically known amino acids are α L amino acids. They are composed of carboxylic end COOH and amino end NH2 and α carbon attached to both of them and special side chain (R) attached to this α carbon . This side ...
... All biologically known protein are polymers of a set of twenty known amino acids. All biologically known amino acids are α L amino acids. They are composed of carboxylic end COOH and amino end NH2 and α carbon attached to both of them and special side chain (R) attached to this α carbon . This side ...
The Role of Leucine-doc
... dietary protein for weight loss. Diets with a reduced ratio of carbohydrates/protein are reported to be beneficial for weight loss, although diet studies appear to lack a fundamental hypothesis to support higher protein intakes. Presently, needs for dietary proteins are established by the recommende ...
... dietary protein for weight loss. Diets with a reduced ratio of carbohydrates/protein are reported to be beneficial for weight loss, although diet studies appear to lack a fundamental hypothesis to support higher protein intakes. Presently, needs for dietary proteins are established by the recommende ...
INTRODUCTORY BIOCHEMISTRY BI 28 Second Midterm
... showed that the production of CO2 by the extract increased when succinate was added. In fact, for every mole of succinate added, many extra moles of CO2 were produced. Explain this effect in terms of the known catabolic pathways. Ans: Succinate is an intermediate in the citric acid cycle that is not ...
... showed that the production of CO2 by the extract increased when succinate was added. In fact, for every mole of succinate added, many extra moles of CO2 were produced. Explain this effect in terms of the known catabolic pathways. Ans: Succinate is an intermediate in the citric acid cycle that is not ...
Say It With DNA - District 196 e
... Say It with DNA! Introduction: Having studied the process by which DNA directs the synthesis of proteins, the student will decode a "secret message". To do this, the student will follow the procedure of protein synthesis listed below, much like what happens within one's cells. Complete the following ...
... Say It with DNA! Introduction: Having studied the process by which DNA directs the synthesis of proteins, the student will decode a "secret message". To do this, the student will follow the procedure of protein synthesis listed below, much like what happens within one's cells. Complete the following ...
1 - contentextra
... Carriers having different electronegativities allow multiple transfers of electrons down an energy gradient. The molecules NADH and FADH2 from the previous stages of cellular respiration provide the electrons (hydrogen) for this chain to occur. Oxygen is the final electron (hydrogen) acceptor formin ...
... Carriers having different electronegativities allow multiple transfers of electrons down an energy gradient. The molecules NADH and FADH2 from the previous stages of cellular respiration provide the electrons (hydrogen) for this chain to occur. Oxygen is the final electron (hydrogen) acceptor formin ...
(pt=4) Label the following diagram with the following terms: ATP
... evolved to minimize the chance of mutations in the DNA molecule. Why? (one reason) ________________________________________________________________________________ ________________________________________________________________________________ What is present in the cell to minimize the chances of ...
... evolved to minimize the chance of mutations in the DNA molecule. Why? (one reason) ________________________________________________________________________________ ________________________________________________________________________________ What is present in the cell to minimize the chances of ...
Amino Acids
... When you ingest a protein your body breaks it down into the individual aminos, reorders them, re-folds them, and turns them into whatever is needed by the body at that time. From only 20 amino acids, the body is able to make thousands of unique proteins with different functions. ...
... When you ingest a protein your body breaks it down into the individual aminos, reorders them, re-folds them, and turns them into whatever is needed by the body at that time. From only 20 amino acids, the body is able to make thousands of unique proteins with different functions. ...
- Circle of Docs
... 39. Glutathione peroxidase is an enzyme in various redox reactions which serves to destroy peroxides and free radicals and requires which mineral as a cofactor? a. Zinc b. Selenium c. Iron d. Chromium ...
... 39. Glutathione peroxidase is an enzyme in various redox reactions which serves to destroy peroxides and free radicals and requires which mineral as a cofactor? a. Zinc b. Selenium c. Iron d. Chromium ...
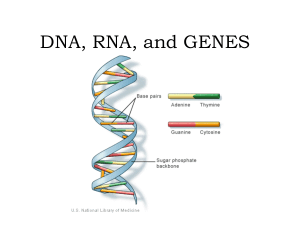
DNA, RNA, and GENES
... • DNA is a Chemical code that contains information that an organism needs to grow and function (nucleic acids) ...
... • DNA is a Chemical code that contains information that an organism needs to grow and function (nucleic acids) ...
Take notes on this document while you are watching the recorded
... 1. The lipids are a large and diverse group of naturally occurring organic compounds that are related by their solubility (will dissolve) in nonpolar5 organic solvents (e.g. ether, chloroform, acetone & benzene) and general insolubility in water (do not dissolve in water - repel water; hydrophobic). ...
... 1. The lipids are a large and diverse group of naturally occurring organic compounds that are related by their solubility (will dissolve) in nonpolar5 organic solvents (e.g. ether, chloroform, acetone & benzene) and general insolubility in water (do not dissolve in water - repel water; hydrophobic). ...
Energy metabolism
... of fat in liver leading to hyperlipidemia or fatty liver. Since CAC is inhibited, availability of oxalloacetate is also limited, this leads to the inhibition of gluconeogenesis leading to hypoglycemia. Excess of NADH accelerates lactate dh to produce lactic acid leading to ...
... of fat in liver leading to hyperlipidemia or fatty liver. Since CAC is inhibited, availability of oxalloacetate is also limited, this leads to the inhibition of gluconeogenesis leading to hypoglycemia. Excess of NADH accelerates lactate dh to produce lactic acid leading to ...
Biochemistry
_and_Carl_Ferdinand_Cori.jpg?width=300)
Biochemistry, sometimes called biological chemistry, is the study of chemical processes within and relating to living organisms. By controlling information flow through biochemical signaling and the flow of chemical energy through metabolism, biochemical processes give rise to the complexity of life. Over the last decades of the 20th century, biochemistry has become so successful at explaining living processes that now almost all areas of the life sciences from botany to medicine to genetics are engaged in biochemical research. Today, the main focus of pure biochemistry is in understanding how biological molecules give rise to the processes that occur within living cells, which in turn relates greatly to the study and understanding of whole organisms.Biochemistry is closely related to molecular biology, the study of the molecular mechanisms by which genetic information encoded in DNA is able to result in the processes of life. Depending on the exact definition of the terms used, molecular biology can be thought of as a branch of biochemistry, or biochemistry as a tool with which to investigate and study molecular biology.Much of biochemistry deals with the structures, functions and interactions of biological macromolecules, such as proteins, nucleic acids, carbohydrates and lipids, which provide the structure of cells and perform many of the functions associated with life. The chemistry of the cell also depends on the reactions of smaller molecules and ions. These can be inorganic, for example water and metal ions, or organic, for example the amino acids which are used to synthesize proteins. The mechanisms by which cells harness energy from their environment via chemical reactions are known as metabolism. The findings of biochemistry are applied primarily in medicine, nutrition, and agriculture. In medicine, biochemists investigate the causes and cures of disease. In nutrition, they study how to maintain health and study the effects of nutritional deficiencies. In agriculture, biochemists investigate soil and fertilizers, and try to discover ways to improve crop cultivation, crop storage and pest control.