2. Structure and bonding of carbohydrates, proteins and lipids
... 2.2. Summary and conclusion Carbohydrates and proteins can occur as polymers, which can be broken down to monomers. Lipids do not have polymer forms but form clusters in the watery milieu of the organism. Compound structures In carbohydrates the polymer forms have mostly not more than two different ...
... 2.2. Summary and conclusion Carbohydrates and proteins can occur as polymers, which can be broken down to monomers. Lipids do not have polymer forms but form clusters in the watery milieu of the organism. Compound structures In carbohydrates the polymer forms have mostly not more than two different ...
123 biochemistry - Jordan University of Science and Technology
... Jordan University of Science and Technology Faculty of Medicine Department of Biochemistry and Molecular Biology Biochemistry M123; Course Description and Objectives: This course deals with structure and properties of biomolecules, such as amino acids, proteins, carbohydrates, lipids, and nucleic ac ...
... Jordan University of Science and Technology Faculty of Medicine Department of Biochemistry and Molecular Biology Biochemistry M123; Course Description and Objectives: This course deals with structure and properties of biomolecules, such as amino acids, proteins, carbohydrates, lipids, and nucleic ac ...
Unit 2 The Chemistry of Life
... leg to digesting your pizza. Protein is the polymer made up of monomers called amino acids. Amino acids are referred to as the “building blocks” of proteins. We use 20 different amino acids to build proteins in our bodies. Your body makes 12 and the others need to be ingested through meat, b ...
... leg to digesting your pizza. Protein is the polymer made up of monomers called amino acids. Amino acids are referred to as the “building blocks” of proteins. We use 20 different amino acids to build proteins in our bodies. Your body makes 12 and the others need to be ingested through meat, b ...
How life works
... genetic material, and ribosomes. This is all prokaryotic cells contain, whilst eukaryotic cells are far more advanced and contain internal structures called organelles. Ribosomes synthasise proteins from a set of 20 amino acids using information encoded on DNA or RNA via messenger RNA. The complex s ...
... genetic material, and ribosomes. This is all prokaryotic cells contain, whilst eukaryotic cells are far more advanced and contain internal structures called organelles. Ribosomes synthasise proteins from a set of 20 amino acids using information encoded on DNA or RNA via messenger RNA. The complex s ...
Cells Ch 1 Sec 3 Chemical Compounds in Cells
... form thousands of words. The letters you use and their order determine the words you form. Even a change in one letter, for example, from rice to mice, creates a new word. Similarly, changes in the type or order of amino acids result in a different protein. Enzymes An enzyme is a type of protein tha ...
... form thousands of words. The letters you use and their order determine the words you form. Even a change in one letter, for example, from rice to mice, creates a new word. Similarly, changes in the type or order of amino acids result in a different protein. Enzymes An enzyme is a type of protein tha ...
Aim and Scope of Organic Chemistry, Composition
... 66. The hormones which controls the process of burning of fats, proteins and carbohydrates to liberate energy in the body is a) Cortisone ...
... 66. The hormones which controls the process of burning of fats, proteins and carbohydrates to liberate energy in the body is a) Cortisone ...
Spotlight on Metabolism
... •Recurrent episodes of binge eating characterized by both: • Eating, in a discrete period of time (within a 2-hr period), an amount of food definitely larger than most people would eat during a similar period of time under similar circumstances • A sense of lack of control over eating during the epi ...
... •Recurrent episodes of binge eating characterized by both: • Eating, in a discrete period of time (within a 2-hr period), an amount of food definitely larger than most people would eat during a similar period of time under similar circumstances • A sense of lack of control over eating during the epi ...
Chapter 2
... unstructured regions of the polypeptide chain into a three-dimensional conformation. This configuration of the molecule is held together by bonds between side groups (R) of some amino acids in the polypeptide. Cysteine is a unique amino acid in that it is capable of disulfide bonding to enforce a te ...
... unstructured regions of the polypeptide chain into a three-dimensional conformation. This configuration of the molecule is held together by bonds between side groups (R) of some amino acids in the polypeptide. Cysteine is a unique amino acid in that it is capable of disulfide bonding to enforce a te ...
Ch 9 chapter summary
... Fermentation releases energy from food molecules by producing ATP without oxygen. Cells convert NADH to the electron carrier NAD+. This allows glycolysis to produce a steady stream of ATP. There are two forms of fermentation. Both start with the reactants pyruvic acid and NADH. • alcoholic fermentat ...
... Fermentation releases energy from food molecules by producing ATP without oxygen. Cells convert NADH to the electron carrier NAD+. This allows glycolysis to produce a steady stream of ATP. There are two forms of fermentation. Both start with the reactants pyruvic acid and NADH. • alcoholic fermentat ...
Regulation of metabolism by PPARs and Angiopoietin like proteins
... Division of Human Nutrition, Wageningen University The Netherlads ...
... Division of Human Nutrition, Wageningen University The Netherlads ...
Protein Structure
... • The tertiary structure is the final specific geometric shape that a protein assumes. • This final shape is determined and stabilized by a variety of bonding interactions between the side chains of the amino acids • These bonding interactions between side chains may cause a number of folds, bends, ...
... • The tertiary structure is the final specific geometric shape that a protein assumes. • This final shape is determined and stabilized by a variety of bonding interactions between the side chains of the amino acids • These bonding interactions between side chains may cause a number of folds, bends, ...
Introduction to Metabolism
... oxidation, amino acid breakdown) Cytosol (glycolysis, fatty acid ...
... oxidation, amino acid breakdown) Cytosol (glycolysis, fatty acid ...
Chapter 29 Biosynthetic Pathways 308 29.1 Your text states in
... The enzyme is inhibited by cholesterol or some derivative of cholesterol in order to control the amount of cholesterol in cells. The enzyme is a key target for the cholesterol-lowering drugs like lovastatin and lipitor that inhibit the enzyme. 29.29 All of the carbons in cholesterol orginate in acet ...
... The enzyme is inhibited by cholesterol or some derivative of cholesterol in order to control the amount of cholesterol in cells. The enzyme is a key target for the cholesterol-lowering drugs like lovastatin and lipitor that inhibit the enzyme. 29.29 All of the carbons in cholesterol orginate in acet ...
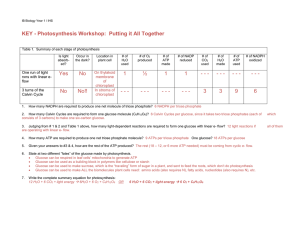
KEY - Photosynthesis Workshop: Putting it All Together
... Given your answers to #3 & 4, how are the rest of the ATP produced? The rest (18 – 12, or 6 more ATP needed) must be coming from cyclic e- flow. ...
... Given your answers to #3 & 4, how are the rest of the ATP produced? The rest (18 – 12, or 6 more ATP needed) must be coming from cyclic e- flow. ...
Chemistry of Life
... 1. What are the 4 major macromolecules? 2. A short polymer and a monomer are linked, what is the by-product and term for this process? 3. How many molecules of water are needed to completely hydrolyze a polymer that is ten monomers long? 4. Why are human sex hormones considered lipids? 5. Identify t ...
... 1. What are the 4 major macromolecules? 2. A short polymer and a monomer are linked, what is the by-product and term for this process? 3. How many molecules of water are needed to completely hydrolyze a polymer that is ten monomers long? 4. Why are human sex hormones considered lipids? 5. Identify t ...
Document
... membrane). And yet, cyanide fairly rapidly shuts down all of oxidative phosphorylation. Why? The electrons that should have gone to the oxygen stay in cytochrome C, such that all of the cytochrome c becomes reduced. When all the cytochrome c is reduced, it can no longer oxidize complex III and thus ...
... membrane). And yet, cyanide fairly rapidly shuts down all of oxidative phosphorylation. Why? The electrons that should have gone to the oxygen stay in cytochrome C, such that all of the cytochrome c becomes reduced. When all the cytochrome c is reduced, it can no longer oxidize complex III and thus ...
Chemical constituents
... a coenzyme. Polynucleotides : RNA (ribonucleic acid) and DNA (deoxyribonucleic acid). Inorganic components - the presence of inorganic ions in cells. The biological significance of water in relation to its properties. ...
... a coenzyme. Polynucleotides : RNA (ribonucleic acid) and DNA (deoxyribonucleic acid). Inorganic components - the presence of inorganic ions in cells. The biological significance of water in relation to its properties. ...
What Do Enzymes Do
... release of energy. Through catabolic pathways, polymers such as proteins, nucleic acids, and polysaccharides are reduced to their constituent parts: amino acids, nucleotides, and sugars, respectively. In contrast, the synthesis of new macromolecules occurs via anabolic pathways that require energy i ...
... release of energy. Through catabolic pathways, polymers such as proteins, nucleic acids, and polysaccharides are reduced to their constituent parts: amino acids, nucleotides, and sugars, respectively. In contrast, the synthesis of new macromolecules occurs via anabolic pathways that require energy i ...
221_exam_2_2003
... In the first section of this class we discussed stromatolites which are fossilized microbial mat communities. The microbial mats consisted of layers of different prokaryotic phototrophs. Based on what you have learned about the properties of the different kinds of phototrophs in this section of the ...
... In the first section of this class we discussed stromatolites which are fossilized microbial mat communities. The microbial mats consisted of layers of different prokaryotic phototrophs. Based on what you have learned about the properties of the different kinds of phototrophs in this section of the ...
Amino acid lecture(1) by Prof.Dr.Moaed Al
... transformation: Arginine – deamination occurs after transfomation to ornithin, lysine – transamination follows the transformation to α-aminoadipate, methionine – deamination of homoserine, proline – deamination after conversion to glutamate, tryptophan – after its transformation to kynurenine, alani ...
... transformation: Arginine – deamination occurs after transfomation to ornithin, lysine – transamination follows the transformation to α-aminoadipate, methionine – deamination of homoserine, proline – deamination after conversion to glutamate, tryptophan – after its transformation to kynurenine, alani ...
Cell Respiration Student Notes
... • Energy of activation (Ea) - the energy that must _____________ to cause molecules to react with one another • Enzyme ____________the amount of energy required for reaction to occur • Enzymes allow reactions to take place at ________________ – otherwise, reactions would not be able to occur at ____ ...
... • Energy of activation (Ea) - the energy that must _____________ to cause molecules to react with one another • Enzyme ____________the amount of energy required for reaction to occur • Enzymes allow reactions to take place at ________________ – otherwise, reactions would not be able to occur at ____ ...
RNA and protein synthesis
... the cytoplasm • Ribosome binds to mRNA and tRNA brings in amino acids which bond together to form a protein. • Codon and anticodon must be complementary ...
... the cytoplasm • Ribosome binds to mRNA and tRNA brings in amino acids which bond together to form a protein. • Codon and anticodon must be complementary ...
Ch 5 Activity List File
... biologically important. 7. Describe the role of symbiosis in cellulose digestion. 8. Describe the building-block molecules, structure, and biological importance of fats, phospholipids, and steroids. 9. Identify an ester linkage and describe how it is formed. 10. Distinguish between saturated and uns ...
... biologically important. 7. Describe the role of symbiosis in cellulose digestion. 8. Describe the building-block molecules, structure, and biological importance of fats, phospholipids, and steroids. 9. Identify an ester linkage and describe how it is formed. 10. Distinguish between saturated and uns ...
Chemistry, Bonds, Phospholipids, triglycerides, proteins, ATP
... ENERGY it’s essential to discuss chemical reactions. • WORK- Movement of an object or change in the physical structure of matter ( walking or running) ( converting water to water vapor) • ENERGY- Capacity to perform work. Need energy to do work. • Kinetic energy- energy in motion. Falling off ladder ...
... ENERGY it’s essential to discuss chemical reactions. • WORK- Movement of an object or change in the physical structure of matter ( walking or running) ( converting water to water vapor) • ENERGY- Capacity to perform work. Need energy to do work. • Kinetic energy- energy in motion. Falling off ladder ...
Biochemistry
_and_Carl_Ferdinand_Cori.jpg?width=300)
Biochemistry, sometimes called biological chemistry, is the study of chemical processes within and relating to living organisms. By controlling information flow through biochemical signaling and the flow of chemical energy through metabolism, biochemical processes give rise to the complexity of life. Over the last decades of the 20th century, biochemistry has become so successful at explaining living processes that now almost all areas of the life sciences from botany to medicine to genetics are engaged in biochemical research. Today, the main focus of pure biochemistry is in understanding how biological molecules give rise to the processes that occur within living cells, which in turn relates greatly to the study and understanding of whole organisms.Biochemistry is closely related to molecular biology, the study of the molecular mechanisms by which genetic information encoded in DNA is able to result in the processes of life. Depending on the exact definition of the terms used, molecular biology can be thought of as a branch of biochemistry, or biochemistry as a tool with which to investigate and study molecular biology.Much of biochemistry deals with the structures, functions and interactions of biological macromolecules, such as proteins, nucleic acids, carbohydrates and lipids, which provide the structure of cells and perform many of the functions associated with life. The chemistry of the cell also depends on the reactions of smaller molecules and ions. These can be inorganic, for example water and metal ions, or organic, for example the amino acids which are used to synthesize proteins. The mechanisms by which cells harness energy from their environment via chemical reactions are known as metabolism. The findings of biochemistry are applied primarily in medicine, nutrition, and agriculture. In medicine, biochemists investigate the causes and cures of disease. In nutrition, they study how to maintain health and study the effects of nutritional deficiencies. In agriculture, biochemists investigate soil and fertilizers, and try to discover ways to improve crop cultivation, crop storage and pest control.