SUPPORTED LIGANDS FOR METAL CATALYZED REACTIONS Rocío Marcos Escartín ISBN:
... Asymmetric Allylic Amination Reaction..............................................................35 PAPER A: Towards the Continuous Flow, Highly Enantioselective Allylic Amination: Ligand Design, Optimization and Supporting...................63 PAPER B: Homogeneous and Polymer-Supported Phosphinoi ...
... Asymmetric Allylic Amination Reaction..............................................................35 PAPER A: Towards the Continuous Flow, Highly Enantioselective Allylic Amination: Ligand Design, Optimization and Supporting...................63 PAPER B: Homogeneous and Polymer-Supported Phosphinoi ...
CH 3 - bYTEBoss
... That are primary (−NH2) or secondary (−NH−) form hydrogen bonds. That are primary have higher melting points than secondary. That are tertiary (no H on N) do not form hydrogen bonds and have lower melting points. All form hydrogen bonds with water. With 1-5 carbon atoms are soluble in wate ...
... That are primary (−NH2) or secondary (−NH−) form hydrogen bonds. That are primary have higher melting points than secondary. That are tertiary (no H on N) do not form hydrogen bonds and have lower melting points. All form hydrogen bonds with water. With 1-5 carbon atoms are soluble in wate ...
Organic Chemistry - hrsbstaff.ednet.ns.ca
... id you know that you have bark from a willow tree in your medicine cabinet at home? The model at the bottom right of this page shows a compound that is found naturally in willow bark. This chemical is called salicin. It is a source of pain relief for moose, deer, and other animals that chew the bark ...
... id you know that you have bark from a willow tree in your medicine cabinet at home? The model at the bottom right of this page shows a compound that is found naturally in willow bark. This chemical is called salicin. It is a source of pain relief for moose, deer, and other animals that chew the bark ...
Carboxylic Acids Esters, Amines and Amides
... hydrogen bonds. That are primary have higher melting points than secondary. That are tertiary (no H on N) do not form hydrogen bonds and have lower melting points. All form hydrogen bonds with water. With 1-5 carbon atoms are soluble in water. ...
... hydrogen bonds. That are primary have higher melting points than secondary. That are tertiary (no H on N) do not form hydrogen bonds and have lower melting points. All form hydrogen bonds with water. With 1-5 carbon atoms are soluble in water. ...
(omit), and Epoxides
... Name the OR group as an alkoxy substituent. • Common names: name the groups bonded to oxygen in alphabetical order followed by the word ...
... Name the OR group as an alkoxy substituent. • Common names: name the groups bonded to oxygen in alphabetical order followed by the word ...
Chapter 3 Alcohols, Phenols, and Ethers
... CHEM 2353 Fundamentals of Organic Chemistry Organic and Biochemistry for Today (Seager & Slabaugh) ...
... CHEM 2353 Fundamentals of Organic Chemistry Organic and Biochemistry for Today (Seager & Slabaugh) ...
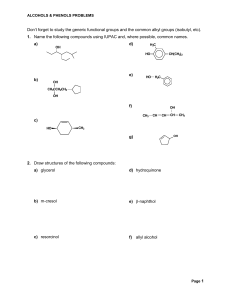
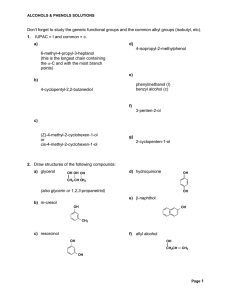
Don`t forget to study the generic functional groups and the common
... 11. Write equations to show how the following transformation can be carried out. More than one step may be necessary. There are marks assigned for each intermediate product (not charged transition states) and there are marks for the reagents used; so list them all. No marks are assigned for mechanis ...
... 11. Write equations to show how the following transformation can be carried out. More than one step may be necessary. There are marks assigned for each intermediate product (not charged transition states) and there are marks for the reagents used; so list them all. No marks are assigned for mechanis ...
Lecture 7_Amines and Amides
... That are tertiary (no H on N) do not form hydrogen bonds and have lower melting points. All form hydrogen bonds with water. With 1-5 carbon atoms are soluble in water. ...
... That are tertiary (no H on N) do not form hydrogen bonds and have lower melting points. All form hydrogen bonds with water. With 1-5 carbon atoms are soluble in water. ...
chapter 22 organic and biological molecules
... polymer. These double bonds can react with sulfur to form crosslinks (bonds) between individual polymer chains. The crosslinked polymer is stronger. c. The longer the chain of polystyrene, the stronger are the London dispersion forces between polymer chains. ...
... polymer. These double bonds can react with sulfur to form crosslinks (bonds) between individual polymer chains. The crosslinked polymer is stronger. c. The longer the chain of polystyrene, the stronger are the London dispersion forces between polymer chains. ...
Course Notes
... useful for determining molecular weight, presence of specific atoms and also certain molecular fragments an organic molecule can be ionized by a number of methods such as bombardment by electron s or other high energy species. usually the ionization results from loss of a single electron and the pro ...
... useful for determining molecular weight, presence of specific atoms and also certain molecular fragments an organic molecule can be ionized by a number of methods such as bombardment by electron s or other high energy species. usually the ionization results from loss of a single electron and the pro ...
Alcohols - Angelo State University
... CHEM 2353 Fundamentals of Organic Chemistry Organic and Biochemistry for Today (Seager & Slabaugh) www.angelo.edu/faculty/kboudrea ...
... CHEM 2353 Fundamentals of Organic Chemistry Organic and Biochemistry for Today (Seager & Slabaugh) www.angelo.edu/faculty/kboudrea ...
NAME - HCC Learning Web
... The conjugate acid to (CH3)3N: is (CH3)3NH+. An electron pair donor substance considers being a Lewis acid. The stronger the acid, the weaker the conjugate base. ...
... The conjugate acid to (CH3)3N: is (CH3)3NH+. An electron pair donor substance considers being a Lewis acid. The stronger the acid, the weaker the conjugate base. ...
Reactions of Alkenes
... carbocation character; more stable transition state (left) has positive charge on more highly substituted carbon ...
... carbocation character; more stable transition state (left) has positive charge on more highly substituted carbon ...
Chem. Soc. Rev., 2015, 44, 2202--2220 - RSC Publishing
... fundamentally different activation modes exerted by BF3 (CQO group) and B(C6F5)3 (Si–H bond) were mainly attributed to the electronic nature of their boron centres. Just recently, Piers, Tuononen, and co-workers finally presented the long-awaited experimental evidence for the missing link of the cat ...
... fundamentally different activation modes exerted by BF3 (CQO group) and B(C6F5)3 (Si–H bond) were mainly attributed to the electronic nature of their boron centres. Just recently, Piers, Tuononen, and co-workers finally presented the long-awaited experimental evidence for the missing link of the cat ...
Acid-Catalyzed Hydration of Alkenes
... transition state for attack of water on bromonium ion has carbocation character; more stable transition state (left) has positive charge on more highly substituted carbon ...
... transition state for attack of water on bromonium ion has carbocation character; more stable transition state (left) has positive charge on more highly substituted carbon ...
Complete Solution Manual
... carboxylic acid part of the ester gives the base name using the suffix –oate. In the common nomenclature rules, the carboxylic acid part is named using common names for the carboxyclic acid ending in the suffix –ate. The bonding and bond angles are the same as discussed previously with carboxylic ac ...
... carboxylic acid part of the ester gives the base name using the suffix –oate. In the common nomenclature rules, the carboxylic acid part is named using common names for the carboxyclic acid ending in the suffix –ate. The bonding and bond angles are the same as discussed previously with carboxylic ac ...
CHAPTER 22 ORGANIC AND BIOLOGICAL MOLECULES 1
... carboxylic acid part of the ester gives the base name using the suffix –oate. In the common nomenclature rules, the carboxylic acid part is named using common names for the carboxyclic acid ending in the suffix –ate. The bonding and bond angles are the same as discussed previously with carboxylic ac ...
... carboxylic acid part of the ester gives the base name using the suffix –oate. In the common nomenclature rules, the carboxylic acid part is named using common names for the carboxyclic acid ending in the suffix –ate. The bonding and bond angles are the same as discussed previously with carboxylic ac ...
Investigation of the steric and electronic properties of 3
... formation. Diversification of the hydroamination reaction’s substrate scope will provide key tools for organic chemists to make C-N bond containing products with greater ease and variety, allowing for the use of less reagents, time, and energy, therefore effecting a cost savings in these processes. ...
... formation. Diversification of the hydroamination reaction’s substrate scope will provide key tools for organic chemists to make C-N bond containing products with greater ease and variety, allowing for the use of less reagents, time, and energy, therefore effecting a cost savings in these processes. ...
Petasis reaction

The Petasis reaction (alternatively called the Petasis borono–Mannich (PBM) reaction) is the chemical reaction of an amine, aldehyde, and vinyl- or aryl-boronic acid to form substituted amines.Reported in 1993 by Nicos Petasis as a practical method towards the synthesis of a geometrically pure antifungal agent, naftifine, the Petasis reaction can be described as a variation of the Mannich reaction. Rather than generating an enolate to form the substituted amine product, in the Petasis reaction, the vinyl group of the organoboronic acid serves as the nucleophile. In comparison to other methods of generating allyl amines, the Petasis reaction tolerates a multifunctional scaffold, with a variety of amines and organoboronic acids as potential starting materials. Additionally, the reaction does not require anhydrous or inert conditions. As a mild, selective synthesis, the Petasis reaction is useful in generating α-amino acids, and is utilized in combinatorial chemistry and drug discovery.