- EdShare - University of Southampton
... Alkenes are unsaturated compounds that can be used in organic synthesis. They can be formed in elimination reactions of halogenoalkanes. An example of this is the reaction between 2-bromopentane and hot ethanolic KOH. Using your knowledge of reaction mechanisms, draw appropriate curly arrows to comp ...
... Alkenes are unsaturated compounds that can be used in organic synthesis. They can be formed in elimination reactions of halogenoalkanes. An example of this is the reaction between 2-bromopentane and hot ethanolic KOH. Using your knowledge of reaction mechanisms, draw appropriate curly arrows to comp ...
- EdShare - University of Southampton
... Alkenes are unsaturated compounds that can be used in organic synthesis. They can be formed in elimination reactions of halogenoalkanes. An example of this is the reaction between 2-bromopentane and hot ethanolic KOH. Using your knowledge of reaction mechanisms, draw appropriate curly arrows to comp ...
... Alkenes are unsaturated compounds that can be used in organic synthesis. They can be formed in elimination reactions of halogenoalkanes. An example of this is the reaction between 2-bromopentane and hot ethanolic KOH. Using your knowledge of reaction mechanisms, draw appropriate curly arrows to comp ...
CfE Higher Chemistry Homework Unit 2: Natures Chemistry Soaps
... 2. Synthetic perfumes are cheaper and easier to produce than natural perfumes. a. Cinnamyl alchol smells pleasantly of hyacinths; it can be described as aromatic. ...
... 2. Synthetic perfumes are cheaper and easier to produce than natural perfumes. a. Cinnamyl alchol smells pleasantly of hyacinths; it can be described as aromatic. ...
6.1.3 revision guide carboxylic acids and esters
... CH3CH2CO2CH3 + NaOH CH3CH2CO2- Na+ + CH3OH methyl propanoate sodium propanoate methanol The carboxylic acid salt product is the anion of the carboxylic acid. The anion is resistant to attack by weak nucleophiles such as alcohols, so the reaction is not reversible. ...
... CH3CH2CO2CH3 + NaOH CH3CH2CO2- Na+ + CH3OH methyl propanoate sodium propanoate methanol The carboxylic acid salt product is the anion of the carboxylic acid. The anion is resistant to attack by weak nucleophiles such as alcohols, so the reaction is not reversible. ...
SCH4U Unit Test Name
... The correct name for the compound given above is which of the following? a. 1-propyl-3-ethyl-4-methylbenzene b. 4-cyclopropyl-2-ethyl-1-methylbenzene c. p,σ - methyl, ethylcyclopropylbenzene d. 1-propyl-3-ethyl-4-methylcyclohexane ...
... The correct name for the compound given above is which of the following? a. 1-propyl-3-ethyl-4-methylbenzene b. 4-cyclopropyl-2-ethyl-1-methylbenzene c. p,σ - methyl, ethylcyclopropylbenzene d. 1-propyl-3-ethyl-4-methylcyclohexane ...
Problem: How would you prepare the following esters? a) Butyl
... The aldehyde intermediate can be isolated by using a gentler reducing agent such as lithium tri-tert-butoxyaluminum hydride. O CCl 1. LiAlH(OC)CH3) 3, ether ...
... The aldehyde intermediate can be isolated by using a gentler reducing agent such as lithium tri-tert-butoxyaluminum hydride. O CCl 1. LiAlH(OC)CH3) 3, ether ...
Soaps, Fragrances and Skin Care 1. In which line of the table are fat
... 2. Synthetic perfumes are cheaper and easier to produce than natural perfumes. a. Cinnamyl alchol smells pleasantly of hyacinths; it can be described as aromatic. ...
... 2. Synthetic perfumes are cheaper and easier to produce than natural perfumes. a. Cinnamyl alchol smells pleasantly of hyacinths; it can be described as aromatic. ...
Exam - Chemistry With BT
... all the stereoisomers (in a way they can be distinguished from one another). ...
... all the stereoisomers (in a way they can be distinguished from one another). ...
Going Bananas Over Isoamyl Acetate
... This Fischer esterification reaction reaches equilibrium after a few hours of refluxing. The position of the equilibrium can be shifted by adding more of the acid or of the alcohol, depending on cost or availability. The mechanism of the reaction involves initial protonation of the carboxyl group, a ...
... This Fischer esterification reaction reaches equilibrium after a few hours of refluxing. The position of the equilibrium can be shifted by adding more of the acid or of the alcohol, depending on cost or availability. The mechanism of the reaction involves initial protonation of the carboxyl group, a ...
Going Bananas Over Isoamyl Acetate
... This Fischer esterification reaction reaches equilibrium after a few hours of refluxing. The position of the equilibrium can be shifted by adding more of the acid or of the alcohol, depending on cost or availability. The mechanism of the reaction involves initial protonation of the carboxyl group, a ...
... This Fischer esterification reaction reaches equilibrium after a few hours of refluxing. The position of the equilibrium can be shifted by adding more of the acid or of the alcohol, depending on cost or availability. The mechanism of the reaction involves initial protonation of the carboxyl group, a ...
In the preparation of the esters given in this experiment
... 6. When tert-pentyl bromide is treated with 80% ethanol, the amounts of alkene products indicated in your lab textbook on pg. 218 (#6.57) are detected on analysis. Explain why Compound I is formed in far greater amount than the terminal alkene. 7. Why is sulfuric acid, rather than hydrochloric acid, ...
... 6. When tert-pentyl bromide is treated with 80% ethanol, the amounts of alkene products indicated in your lab textbook on pg. 218 (#6.57) are detected on analysis. Explain why Compound I is formed in far greater amount than the terminal alkene. 7. Why is sulfuric acid, rather than hydrochloric acid, ...
nitrogen compounds
... To make a primary amine in this reaction, excess ammonia is needed. Otherwise, with excess chloroethane, the reaction can continue and will produce a mixture of products CH3Br + CH3NH2 (CH3)2NH + HBr CH3Br + (CH3)2NH (CH3)3N + HBr CH3Br + (CH3)3N (CH3)4N+ + Br- ...
... To make a primary amine in this reaction, excess ammonia is needed. Otherwise, with excess chloroethane, the reaction can continue and will produce a mixture of products CH3Br + CH3NH2 (CH3)2NH + HBr CH3Br + (CH3)2NH (CH3)3N + HBr CH3Br + (CH3)3N (CH3)4N+ + Br- ...
Lecture Resource ()
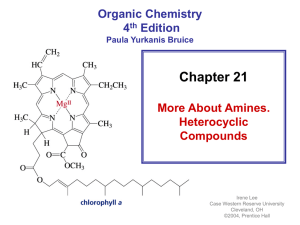
... • Some amines are heterocyclic compounds (or heterocycles) • Most drugs, vitamins, and many other natural products are heterocycles • A natural product is a compound synthesized by a plant or an animal ...
... • Some amines are heterocyclic compounds (or heterocycles) • Most drugs, vitamins, and many other natural products are heterocycles • A natural product is a compound synthesized by a plant or an animal ...
Level 3: Organics Part I
... pans, chloroform – CHCl3 – used as a solvent and in movies as an anaesthetic, CCl4 – solvent used in drycleaning fluid until it was found to ...
... pans, chloroform – CHCl3 – used as a solvent and in movies as an anaesthetic, CCl4 – solvent used in drycleaning fluid until it was found to ...
Chapter 25 & 26 Notes, part II
... carbons in the main chain and use the prefix, then end it in –ene. If there is more than one spot where a double bond could occur, number it just like you would number a substituent. ...
... carbons in the main chain and use the prefix, then end it in –ene. If there is more than one spot where a double bond could occur, number it just like you would number a substituent. ...
Organic Synthesis
... these is likely to have the best chance of success. Even the most experienced chemists develop routes, which work well on paper but fail miserably in the lab. But, that having been said, there are some guidelines, which are helpful in designing a synthesis. What follows is an attempt to lay out thos ...
... these is likely to have the best chance of success. Even the most experienced chemists develop routes, which work well on paper but fail miserably in the lab. But, that having been said, there are some guidelines, which are helpful in designing a synthesis. What follows is an attempt to lay out thos ...
File - Kheriaty Chemistry
... 22. a. Balance the reaction between sulfuric acid and ammonia (NH3) to yield ammonium sulfate. ...
... 22. a. Balance the reaction between sulfuric acid and ammonia (NH3) to yield ammonium sulfate. ...
Discussion Problem Set 1. C483 Spring 2014
... 8. Do you expect the pKa of the side chains in Glu and Asp to be higher or lower than the alpha carboxyl group. Why? 9. What amino acids are used by muscles as a significant source of energy? What is the byproduct formed by this process? 10. Collagen is thought to form triple helices in which H-bond ...
... 8. Do you expect the pKa of the side chains in Glu and Asp to be higher or lower than the alpha carboxyl group. Why? 9. What amino acids are used by muscles as a significant source of energy? What is the byproduct formed by this process? 10. Collagen is thought to form triple helices in which H-bond ...
Carboxylic Acids
... 4. Draw and name the isomers of C5H8 ( some may not be alkynes) 5. Construct a model of an acetylene lamp that could have been used as a lantern for early automobiles. Explain how your model works to make the light and direct the light down the road. 6. Write the formula equations for each of the fo ...
... 4. Draw and name the isomers of C5H8 ( some may not be alkynes) 5. Construct a model of an acetylene lamp that could have been used as a lantern for early automobiles. Explain how your model works to make the light and direct the light down the road. 6. Write the formula equations for each of the fo ...
3.4 How do we use the Activity Series
... ADPHS Instructor Ms. Kasia Room 109 UNIT 3 CHEMICAL REACTIONS 3.4 How do we use the Activity Series to Predict if a reaction will take place? AIM: ...
... ADPHS Instructor Ms. Kasia Room 109 UNIT 3 CHEMICAL REACTIONS 3.4 How do we use the Activity Series to Predict if a reaction will take place? AIM: ...
Petasis reaction

The Petasis reaction (alternatively called the Petasis borono–Mannich (PBM) reaction) is the chemical reaction of an amine, aldehyde, and vinyl- or aryl-boronic acid to form substituted amines.Reported in 1993 by Nicos Petasis as a practical method towards the synthesis of a geometrically pure antifungal agent, naftifine, the Petasis reaction can be described as a variation of the Mannich reaction. Rather than generating an enolate to form the substituted amine product, in the Petasis reaction, the vinyl group of the organoboronic acid serves as the nucleophile. In comparison to other methods of generating allyl amines, the Petasis reaction tolerates a multifunctional scaffold, with a variety of amines and organoboronic acids as potential starting materials. Additionally, the reaction does not require anhydrous or inert conditions. As a mild, selective synthesis, the Petasis reaction is useful in generating α-amino acids, and is utilized in combinatorial chemistry and drug discovery.