chapter-15
... • aldehydes and ketones are polar compounds and interact in the pure state by dipole-dipole interactions • they have higher boiling points and are more soluble in water than nonpolar compounds of comparable ...
... • aldehydes and ketones are polar compounds and interact in the pure state by dipole-dipole interactions • they have higher boiling points and are more soluble in water than nonpolar compounds of comparable ...
7: Reactions of Haloalkanes, Alcohols, and Amines. Nucleophilic
... S N1 mechanism (see Figure7.13). These changes in SN1 rates result from the effect of alkyl groups such as CH3 on the stability of R3 C+ that forms in the first step of the SN1 mechanism. Carbocation Stability. The relative stability of simple methyl substituted carbocations is (CH3 )3 C+ > (CH3 )2 ...
... S N1 mechanism (see Figure7.13). These changes in SN1 rates result from the effect of alkyl groups such as CH3 on the stability of R3 C+ that forms in the first step of the SN1 mechanism. Carbocation Stability. The relative stability of simple methyl substituted carbocations is (CH3 )3 C+ > (CH3 )2 ...
Chem E2b - Organic Chemistry II What is Organic Chemistry?
... • Strong reacts to give weak • Weaker base = stronger conjugate acid • Stable bases (anions) are weak bases ...
... • Strong reacts to give weak • Weaker base = stronger conjugate acid • Stable bases (anions) are weak bases ...
boehm_rl
... ethyl methylchloroacetate, ethyl phenylchloroacetate, and ethyl diphenylchloroacetate from the corresponding alcohols. Bissinger and Kung (32) studied the effects of varying experimental conditions upon the reactions of propyl alcohols and thionyl chloride . In order to prepare unstable propyl sulfi ...
... ethyl methylchloroacetate, ethyl phenylchloroacetate, and ethyl diphenylchloroacetate from the corresponding alcohols. Bissinger and Kung (32) studied the effects of varying experimental conditions upon the reactions of propyl alcohols and thionyl chloride . In order to prepare unstable propyl sulfi ...
Document
... vinyl radicals on the oxygen atom. Taking these observations into account, Professor Bartoli showed that the alkyl Grignard reaction behaved as a process analogous to the Friedel-Crafts reaction proceeding with reversed polarity. Contemporarily but independently from Prof. Makosza’s work on VNS Prof ...
... vinyl radicals on the oxygen atom. Taking these observations into account, Professor Bartoli showed that the alkyl Grignard reaction behaved as a process analogous to the Friedel-Crafts reaction proceeding with reversed polarity. Contemporarily but independently from Prof. Makosza’s work on VNS Prof ...
PDF - Nanyang Technological University
... We used a chiral amine combined with CuBr2 as a cooperative catalytic system to develop the enantioselective reaction between 1 a and 2 a. The reaction catalyzed by (S)proline/CuBr2 in DMF gave the desired product in 63 % yield and in a 1:1 diastereomeric ratio (d.r.), with virtually no enantioselec ...
... We used a chiral amine combined with CuBr2 as a cooperative catalytic system to develop the enantioselective reaction between 1 a and 2 a. The reaction catalyzed by (S)proline/CuBr2 in DMF gave the desired product in 63 % yield and in a 1:1 diastereomeric ratio (d.r.), with virtually no enantioselec ...
physicochemical properties of organic medicinal agents
... Because of their atomic composition, alkenes and alkenyl groups are classified as “non-polar compounds” and the only significant intermolecular bonding possible are relatively weak van der Waals interactions (VDWs), or “induced dipolar” interactions created by temporary distortions in the electron d ...
... Because of their atomic composition, alkenes and alkenyl groups are classified as “non-polar compounds” and the only significant intermolecular bonding possible are relatively weak van der Waals interactions (VDWs), or “induced dipolar” interactions created by temporary distortions in the electron d ...
chm121 tutorial kit - Covenant University
... The following are common drying agent for organic solutions but which of them has high capacity, fast speed, and good efficiency as its chemical property. (a) CaSO4 (b) CaCl 2 (c) K2CO3 (d) MgSO4 All except ------------- is not employed as an adsorbents in column chromatography (a) Starch (b) Silica ...
... The following are common drying agent for organic solutions but which of them has high capacity, fast speed, and good efficiency as its chemical property. (a) CaSO4 (b) CaCl 2 (c) K2CO3 (d) MgSO4 All except ------------- is not employed as an adsorbents in column chromatography (a) Starch (b) Silica ...
Synthetic Organic Chemistry - Name
... R1COR2 RMgX / ether R1CR2 HOH Due to the presence of bulky groups in both or OH either of the reactants in Grignard reagent the extent of addition is reduced or the reaction not take place or some abnormal product is formed. If Grignard reagent has β-hydrogen atom, then in some hindred ketone also s ...
... R1COR2 RMgX / ether R1CR2 HOH Due to the presence of bulky groups in both or OH either of the reactants in Grignard reagent the extent of addition is reduced or the reaction not take place or some abnormal product is formed. If Grignard reagent has β-hydrogen atom, then in some hindred ketone also s ...
Class Notes
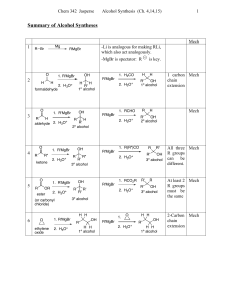
... • 3º alcohol, all three attachments different ß Ketone Precursor • 3º alcohol, two (or more) of the attachments identical ß Ester • 2º alcohol ß Aldehyde • 1º alcohol ß Formaldehyde or ethylene oxide ...
... • 3º alcohol, all three attachments different ß Ketone Precursor • 3º alcohol, two (or more) of the attachments identical ß Ester • 2º alcohol ß Aldehyde • 1º alcohol ß Formaldehyde or ethylene oxide ...
Document
... Now let us apply this background to the study of the organic chemistry of metabolism We study two key metabolic pathways • -oxidation of fatty acids • glycolysis ...
... Now let us apply this background to the study of the organic chemistry of metabolism We study two key metabolic pathways • -oxidation of fatty acids • glycolysis ...
$doc.title
... First I would like to start by taking the opportunity to thank my supervisor, Dr. Crudden. Over the course of my graduate career I have no doubt that you worked not only to make m ...
... First I would like to start by taking the opportunity to thank my supervisor, Dr. Crudden. Over the course of my graduate career I have no doubt that you worked not only to make m ...
HMDS+TMCS+Pyridine - Sigma
... sample), following the same procedure as used for the sample. 1. Weigh 1-10mg of sample into a 5mL reaction vessel. If appropriate, dissolve sample in solvent (see below). If sample is in aqueous solution, evaporate to dryness, then use neat or add solvent. 2. Add excess silylating reagent. HMDS+TMC ...
... sample), following the same procedure as used for the sample. 1. Weigh 1-10mg of sample into a 5mL reaction vessel. If appropriate, dissolve sample in solvent (see below). If sample is in aqueous solution, evaporate to dryness, then use neat or add solvent. 2. Add excess silylating reagent. HMDS+TMC ...
Reactions
... It is a milder reagent and if you use it carefully you can stop the reaction at the intermediate aldehyde step. ...
... It is a milder reagent and if you use it carefully you can stop the reaction at the intermediate aldehyde step. ...
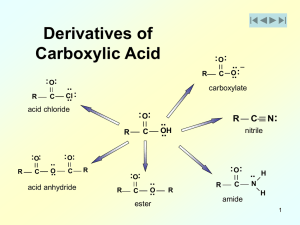
Nucleophilic Acyl Substitution
... and aromatic hydrocarbons. Like alcohols and ethers, carbonyl compounds with fewer than four carbons are soluble in water. Esters, N,N-disubstituted amides, and nitriles are often used as solvents because they are polar, but do not have reactive hydroxyl or amino groups. We have seen that dimethylfo ...
... and aromatic hydrocarbons. Like alcohols and ethers, carbonyl compounds with fewer than four carbons are soluble in water. Esters, N,N-disubstituted amides, and nitriles are often used as solvents because they are polar, but do not have reactive hydroxyl or amino groups. We have seen that dimethylfo ...
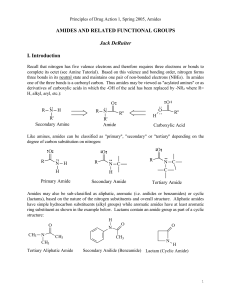
AMIDES AND RELATED FUNCTIONAL GROUPS
... Note the basic structural difference between an ester and amide at the site of reaction is a single atom! Amides have a nitrogen atom where esters have an (ether) oxygen. The difference in electrophilic and hydrolytic reactivity between amides and esters is directly related to the different electron ...
... Note the basic structural difference between an ester and amide at the site of reaction is a single atom! Amides have a nitrogen atom where esters have an (ether) oxygen. The difference in electrophilic and hydrolytic reactivity between amides and esters is directly related to the different electron ...
BIORANSFORMATION
... Glucoronic acid, Sulfate, Glycine to either unchanged drugs or Phase I product having suitable functional groups as COOH,-OH,-NH2,- SH. • Thus is called as Conjugation reactions. • Since the product formed is having high molecular weight so called as synthetic reactions. • The product formed is hydr ...
... Glucoronic acid, Sulfate, Glycine to either unchanged drugs or Phase I product having suitable functional groups as COOH,-OH,-NH2,- SH. • Thus is called as Conjugation reactions. • Since the product formed is having high molecular weight so called as synthetic reactions. • The product formed is hydr ...
BSA + TMCS + TMSI - Sigma
... sample), following the same procedure as used for the sample. 1. Weigh 1-10mg of sample into a 5mL reaction vessel. If appropriate, dissolve sample in solvent (see below). If sample is in aqueous solution, evaporate to dryness, then use neat or add solvent. 2. Add excess silylating reagent (BSA+TMCS ...
... sample), following the same procedure as used for the sample. 1. Weigh 1-10mg of sample into a 5mL reaction vessel. If appropriate, dissolve sample in solvent (see below). If sample is in aqueous solution, evaporate to dryness, then use neat or add solvent. 2. Add excess silylating reagent (BSA+TMCS ...
Slide 1
... equilibrium constant is not large (Keq ~ 1) but high yields can be obtained by adding a large excess of one of the reactants and removing the H2O formed. The reaction is reversible. A large excess of H2O favors the reverse reaction. Bulky (sterically hindered) reagents reduce yields. Since alcohols ...
... equilibrium constant is not large (Keq ~ 1) but high yields can be obtained by adding a large excess of one of the reactants and removing the H2O formed. The reaction is reversible. A large excess of H2O favors the reverse reaction. Bulky (sterically hindered) reagents reduce yields. Since alcohols ...
10 Haloalkanes and Haloarenes
... The reaction of alkenes (except ethylene) with Cl2 or Br2 at higher temperature (about 773 K) gives substitution reaction product instead of addition reaction product. This is because at higher temperature, the addition reaction is reversible and the substitution reaction is irreversible. The hydrog ...
... The reaction of alkenes (except ethylene) with Cl2 or Br2 at higher temperature (about 773 K) gives substitution reaction product instead of addition reaction product. This is because at higher temperature, the addition reaction is reversible and the substitution reaction is irreversible. The hydrog ...
Catalytic Asymmetric Induction. Highly Enantioselective Addition of
... asymmetric alkylation. The enantiomeric excess was determined carefully by HPLC analysis using a chiral stationary phase. Enantioselectivity of the reaction of p-substituted benzaldehydes is generally high. Certain a,@unsaturated or aliphatic aldehydes can be also alkylated with a high degree of ena ...
... asymmetric alkylation. The enantiomeric excess was determined carefully by HPLC analysis using a chiral stationary phase. Enantioselectivity of the reaction of p-substituted benzaldehydes is generally high. Certain a,@unsaturated or aliphatic aldehydes can be also alkylated with a high degree of ena ...
1 Q. What are Saturated and unsaturated hydrocarbons?imp
... each of these possible arrangement represents a conformations. However for the sake of convenience each rotation is done in the instalment of 600. Thus we will obtain six no. of different conformations of ethane. Out of these six, extreme two are worth to study. Because they are different from each ...
... each of these possible arrangement represents a conformations. However for the sake of convenience each rotation is done in the instalment of 600. Thus we will obtain six no. of different conformations of ethane. Out of these six, extreme two are worth to study. Because they are different from each ...
View/Open
... group is similar to the way they affect the reactivity of an aromatic ring toward electrophilic substitution (Section 16.5). A chlorine substituent, for example, inductively withdraws electrons from an acyl group in the same way that it withdraws electrons from and thus deactivates an aromatic ring. ...
... group is similar to the way they affect the reactivity of an aromatic ring toward electrophilic substitution (Section 16.5). A chlorine substituent, for example, inductively withdraws electrons from an acyl group in the same way that it withdraws electrons from and thus deactivates an aromatic ring. ...
12_chemistry_impq_CH10_haloalkanes_and_haloarenes_02
... Ans. It is because HI formed will get oxidized to I2 by concentrated Sulphuric acid which is an oxidizing agent. Q10. p- dichlorobenzene has highest m.p. than those of ortho and m-isomers.? Ans. p- dichlorobenzene is symmetrical, fits into crystal lattice more readily and has higher melting point. ...
... Ans. It is because HI formed will get oxidized to I2 by concentrated Sulphuric acid which is an oxidizing agent. Q10. p- dichlorobenzene has highest m.p. than those of ortho and m-isomers.? Ans. p- dichlorobenzene is symmetrical, fits into crystal lattice more readily and has higher melting point. ...
Full Text
... Protection of functional groups in multistep organic syntheses is one of the key factors in the success of the synthesis. The protecting group should selectively react in good yield to give a protected substrate and should be selectively removed in good yield by readily available, preferably nontoxi ...
... Protection of functional groups in multistep organic syntheses is one of the key factors in the success of the synthesis. The protecting group should selectively react in good yield to give a protected substrate and should be selectively removed in good yield by readily available, preferably nontoxi ...
Wolff–Kishner reduction

The Wolff–Kishner reduction is a reaction used in organic chemistry to convert carbonyl functionalities into methylene groups. In the context of complex molecule synthesis, it is most frequently employed to remove a carbonyl group after it has served its synthetic purpose of activating an intermediate in a preceding step. As such, there is no obvious retron for this reaction. Originally reported by Nikolai Kischner in 1911 and Ludwig Wolff in 1912, it has been applied to the total synthesis of scopadulcic acid B, aspidospermidine and dysidiolide.In general, the reaction mechanism first involves the in situ generation of a hydrazone by condensation of hydrazine with the ketone or aldehyde substrate. Sometimes it is however advantageous to use a pre-formed hydrazone as substrate (see modifications). The hydrazone is deprotonated by alkoxide base followed by a concerted, rate-determining step in which a diimide anion is formed. Collapse of this alkyldiimde with loss of N2 leads to formation of an alkylanion which can be protonated by solvent to give the desired product.Because the Wolff–Kishner reduction requires highly basic conditions, it is unsuitable for base-sensitive substrates. However, this method can be superior over the related Clemmensen reduction for acid-sensitive compounds such as pyrroles and for high-molecular weight compounds.