Week 8
... The exact mechanism of C-H activation/functionalization is unclear. Two possibilities are likely: 1. oxidative addition followed by reductive elimination, 2. σ-bond metathesis. The first possibility would require loss of a second CO or Cp* slippage to create a site of electronic unsaturation at the ...
... The exact mechanism of C-H activation/functionalization is unclear. Two possibilities are likely: 1. oxidative addition followed by reductive elimination, 2. σ-bond metathesis. The first possibility would require loss of a second CO or Cp* slippage to create a site of electronic unsaturation at the ...
Chemistry 360 - Athabasca University
... What are the hazards of working with concentrated acids like glacial acetic acid and sulfuric acid? a) They are both extremely flammable b) They are only mildly corrosive and no significant precautions are needed c) Boiling these highly corrosive acids increases the danger to the experimenter, espec ...
... What are the hazards of working with concentrated acids like glacial acetic acid and sulfuric acid? a) They are both extremely flammable b) They are only mildly corrosive and no significant precautions are needed c) Boiling these highly corrosive acids increases the danger to the experimenter, espec ...
Topic 10 SL Mark Scheme Past exam paper questions
... chains of up to 6 carbon atoms with one of the following functional groups: (alkane), alkene, alcohol, aldehyde, ketone, carboxylic acid and halide. Examples of compounds containing amino, benzene ring (phenyl) and ester functional groups. Describe the volatility and solubility in water of compounds ...
... chains of up to 6 carbon atoms with one of the following functional groups: (alkane), alkene, alcohol, aldehyde, ketone, carboxylic acid and halide. Examples of compounds containing amino, benzene ring (phenyl) and ester functional groups. Describe the volatility and solubility in water of compounds ...
LECTURE 7 REDUCTIVE ELIMINATIONSa
... • Indeed, Halpern showed that RhCl(PPh3)2, formed by loss of a PPh3 group from RhCl(PPh3)3, gives oxidative addition with hydrogen at a rate at least 104 times faster than the 4‐ coordinate complex. ...
... • Indeed, Halpern showed that RhCl(PPh3)2, formed by loss of a PPh3 group from RhCl(PPh3)3, gives oxidative addition with hydrogen at a rate at least 104 times faster than the 4‐ coordinate complex. ...
IOSR Journal of Applied Chemistry (IOSR-JAC)
... affluent societies, are also obtained by above means. It is the biggest challenge in the modern society to maintain good supply line of these products by applying the advanced research, at the same time creating strategies to meet the critical needs of growing population of the world. Chemists have ...
... affluent societies, are also obtained by above means. It is the biggest challenge in the modern society to maintain good supply line of these products by applying the advanced research, at the same time creating strategies to meet the critical needs of growing population of the world. Chemists have ...
nucleophilic addition on ketones and ketimines - ISI
... which will in turn allow the transfer of the allyl group to the copper atom. The addition of La(Oi Pr)3 as a Lewis acid accelerates the reaction, presumably by participation in the transmetallation step that leads to the active allylcopper species. This catalytic system was also applied to the first ...
... which will in turn allow the transfer of the allyl group to the copper atom. The addition of La(Oi Pr)3 as a Lewis acid accelerates the reaction, presumably by participation in the transmetallation step that leads to the active allylcopper species. This catalytic system was also applied to the first ...
Preparation of d, l-Phenylalanine by Amidocarbonylation of Benzyl
... catalyst, Co2(CO)8, are used which would require an efficient recycle process to be viable. Good yields of product (up to 79%) are obtained only if a two- to threefold excess of acetamide is used, but this might hamper the purification procedure. Also, the selectivity with respect to acetamide is ra ...
... catalyst, Co2(CO)8, are used which would require an efficient recycle process to be viable. Good yields of product (up to 79%) are obtained only if a two- to threefold excess of acetamide is used, but this might hamper the purification procedure. Also, the selectivity with respect to acetamide is ra ...
Chapter 19 Amines
... In the presence of a strong base, primary amides react with chlorine or bromine to form shortened amines, with the loss of the carbonyl carbon atom. This reaction, called the Hofmann rearrangement, is used to synthesize primary and aryl amines. Chapter 19 ...
... In the presence of a strong base, primary amides react with chlorine or bromine to form shortened amines, with the loss of the carbonyl carbon atom. This reaction, called the Hofmann rearrangement, is used to synthesize primary and aryl amines. Chapter 19 ...
4.8 Preparation of Alkyl Halides from Alcohols and Hydrogen
... a carbocation. The overall reaction mechanism involves three elementary steps; the first two steps lead to the carbocation intermediate, the third step is the conversion of this carbocation to the alkyl halide. ...
... a carbocation. The overall reaction mechanism involves three elementary steps; the first two steps lead to the carbocation intermediate, the third step is the conversion of this carbocation to the alkyl halide. ...
+ ∂ - CHEM171 – Lecture Series Seven : 2012/05
... Compound A, C4H8, reacts with O3 to give methanal and compound B. Compound B reacts with CrO3 to give compound C. Compound B also reacts with LiAlH4 to give D. Upon treatment with 95% H2SO4 at 170oC compound D gives rise to compound E. E, when treated with HCl gives us the major product F and also a ...
... Compound A, C4H8, reacts with O3 to give methanal and compound B. Compound B reacts with CrO3 to give compound C. Compound B also reacts with LiAlH4 to give D. Upon treatment with 95% H2SO4 at 170oC compound D gives rise to compound E. E, when treated with HCl gives us the major product F and also a ...
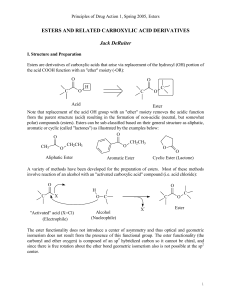
esters and related carboxylic acid derivatives
... Thus the esters carbonyl carbon is susceptible to "attack" by electron rich atoms (nucleophiles) including the oxygen of water and the nucleophilic residues at the active sites of esterase enzymes. When in the presence of a nucleophile, an ester may undergo reaction leading to cleavage of the carbon ...
... Thus the esters carbonyl carbon is susceptible to "attack" by electron rich atoms (nucleophiles) including the oxygen of water and the nucleophilic residues at the active sites of esterase enzymes. When in the presence of a nucleophile, an ester may undergo reaction leading to cleavage of the carbon ...
New Applications for Sulfur-Based Leaving Groups in Synthesis
... chemistry within his group and his stress-free approach to the subject has allowed me to form many of my own ideas and not be afraid to suggest them, no matter how absurd they seem. Towards the end of my PhD, EPR spectroscopy became an invaluable tool and I’d like to thank Dr. Chris Kay and Dr. Enri ...
... chemistry within his group and his stress-free approach to the subject has allowed me to form many of my own ideas and not be afraid to suggest them, no matter how absurd they seem. Towards the end of my PhD, EPR spectroscopy became an invaluable tool and I’d like to thank Dr. Chris Kay and Dr. Enri ...
Aromatic Substitution Reactions
... ost of the reactions discussed in this chapter involve the attack of an electrophile on an aromatic compound. Although the initial step of the mechanism resembles that of the electrophilic addition reactions of carbon–carbon double bonds discussed in Chapter 11, the final product here results from s ...
... ost of the reactions discussed in this chapter involve the attack of an electrophile on an aromatic compound. Although the initial step of the mechanism resembles that of the electrophilic addition reactions of carbon–carbon double bonds discussed in Chapter 11, the final product here results from s ...
© John Congleton, Orange Coast College Organic Chemistry 220
... Be able to predict whether a reaction will proceed via o SN1 and E1 o S N2 o SN2 and E2 o E2 What makes a good nucleophile? What makes a good base? What makes a good leaving group? What is meant by high and low polarizability? Allylic bromination Understand, be able to predict, and be able to comple ...
... Be able to predict whether a reaction will proceed via o SN1 and E1 o S N2 o SN2 and E2 o E2 What makes a good nucleophile? What makes a good base? What makes a good leaving group? What is meant by high and low polarizability? Allylic bromination Understand, be able to predict, and be able to comple ...
Synthesis of Amide Bond Isosteres Incorporated
... amide bond (Figure 8). Free rotation around the C-N bond in the methylene amine (ψ[CH2NH]) isostere also confers increased flexibility. The amino group is ionized at physiological pH which can protect from enzymatic degradation. However, introducing a charge in the binding site may also have implica ...
... amide bond (Figure 8). Free rotation around the C-N bond in the methylene amine (ψ[CH2NH]) isostere also confers increased flexibility. The amino group is ionized at physiological pH which can protect from enzymatic degradation. However, introducing a charge in the binding site may also have implica ...
HMDS - Sigma
... of the sample at selected time intervals until no further increase in product peak(s) is observed. Derivatization times vary widely, depending upon the specific compound(s) being derivatized. Many compounds are completely derivatized as soon as they dissolve in the reagent. Compounds with poor solub ...
... of the sample at selected time intervals until no further increase in product peak(s) is observed. Derivatization times vary widely, depending upon the specific compound(s) being derivatized. Many compounds are completely derivatized as soon as they dissolve in the reagent. Compounds with poor solub ...
Lectures 4-6
... Collins Oxidation (CrO3 • 2pyridine) TL 1969, 336 - CrO3 (anhydrous) + pyridine (anhydrous) ...
... Collins Oxidation (CrO3 • 2pyridine) TL 1969, 336 - CrO3 (anhydrous) + pyridine (anhydrous) ...
Organometallic Methods for Forming and Cleaving Carbon
... the PhD. ChemClub for the organizing of several events. I deeply appreciate Prof. Brian Stoltz for allowing me to be a part of his research group during my external stay at CalTech. I appreciate Dr. Scott Virgil for being able to use his equipment, David VanderVelde for the use of the NMR spectromet ...
... the PhD. ChemClub for the organizing of several events. I deeply appreciate Prof. Brian Stoltz for allowing me to be a part of his research group during my external stay at CalTech. I appreciate Dr. Scott Virgil for being able to use his equipment, David VanderVelde for the use of the NMR spectromet ...
Synthesis of Alcohols Using Grignard Reagents Grignard reagents
... O What remains is the combination of Grignard reagent and carbonyl compound that can be used to prepare the alcohol. ...
... O What remains is the combination of Grignard reagent and carbonyl compound that can be used to prepare the alcohol. ...
Chemistry 0310 - Organic Chemistry 1 Chapter 12. Reactions of
... - Syn-Hydroxylations of alkenes are most conveniently performed with catalytic OsO4 and NMO (N-methylmorpholine N-oxide) as co-oxidant. Attack occurs from the less-hindered face of the alkene, and a vicinal syn-diol is isolated after reductive workup. - Oxidative cleavage of 1,2-disubstituted alken ...
... - Syn-Hydroxylations of alkenes are most conveniently performed with catalytic OsO4 and NMO (N-methylmorpholine N-oxide) as co-oxidant. Attack occurs from the less-hindered face of the alkene, and a vicinal syn-diol is isolated after reductive workup. - Oxidative cleavage of 1,2-disubstituted alken ...
Isoindolone Formation via Intramolecular Diels
... synthesis, the disconnection of which is shown in Scheme 1. The isoindolone ring was constructed by transformation of 4bromo-2,5-dimethylaniline (3) to the corresponding acid 9 over three steps, followed by bromination, ester formation, and reaction with 4-trifluoromethoxybenzylamine (4) to give 12. ...
... synthesis, the disconnection of which is shown in Scheme 1. The isoindolone ring was constructed by transformation of 4bromo-2,5-dimethylaniline (3) to the corresponding acid 9 over three steps, followed by bromination, ester formation, and reaction with 4-trifluoromethoxybenzylamine (4) to give 12. ...
Scope and Limitations - Organic Reactions Wiki
... smaller size of the OsO4•quinuclidine complex compared to the OsO4•TMEDA system, increased levels of selectivity are obtained in the directed oxidation of sterically demanding substrates.19 Replacing QNO•H2O, which needs to be prepared beforehand, with commercially available Me3NO•2H2O makes the dih ...
... smaller size of the OsO4•quinuclidine complex compared to the OsO4•TMEDA system, increased levels of selectivity are obtained in the directed oxidation of sterically demanding substrates.19 Replacing QNO•H2O, which needs to be prepared beforehand, with commercially available Me3NO•2H2O makes the dih ...
Ch 7 - Practice problem (Answers)
... C) E1 for tertiary halides, E2 for primary and secondary halides. D) E2 for tertiary halides, E1 for primary and secondary halides. Ans: B 25. Which of the following sets of conditions most favors the E1 mechanism? A) when the alkyl halide is tertiary and the base is a weak base B) when the alkyl ha ...
... C) E1 for tertiary halides, E2 for primary and secondary halides. D) E2 for tertiary halides, E1 for primary and secondary halides. Ans: B 25. Which of the following sets of conditions most favors the E1 mechanism? A) when the alkyl halide is tertiary and the base is a weak base B) when the alkyl ha ...
Microsoft Word
... While compound 71 was characterized, the organomercuric bromides resulting from other cyclopropylcarbinols were not characterized and directly subjected to demercuration using n– tributyltin hydride in the presence of catalytic amounts of triethylborane in an oxygen atmosphere. Demercuration using L ...
... While compound 71 was characterized, the organomercuric bromides resulting from other cyclopropylcarbinols were not characterized and directly subjected to demercuration using n– tributyltin hydride in the presence of catalytic amounts of triethylborane in an oxygen atmosphere. Demercuration using L ...
Wolff–Kishner reduction

The Wolff–Kishner reduction is a reaction used in organic chemistry to convert carbonyl functionalities into methylene groups. In the context of complex molecule synthesis, it is most frequently employed to remove a carbonyl group after it has served its synthetic purpose of activating an intermediate in a preceding step. As such, there is no obvious retron for this reaction. Originally reported by Nikolai Kischner in 1911 and Ludwig Wolff in 1912, it has been applied to the total synthesis of scopadulcic acid B, aspidospermidine and dysidiolide.In general, the reaction mechanism first involves the in situ generation of a hydrazone by condensation of hydrazine with the ketone or aldehyde substrate. Sometimes it is however advantageous to use a pre-formed hydrazone as substrate (see modifications). The hydrazone is deprotonated by alkoxide base followed by a concerted, rate-determining step in which a diimide anion is formed. Collapse of this alkyldiimde with loss of N2 leads to formation of an alkylanion which can be protonated by solvent to give the desired product.Because the Wolff–Kishner reduction requires highly basic conditions, it is unsuitable for base-sensitive substrates. However, this method can be superior over the related Clemmensen reduction for acid-sensitive compounds such as pyrroles and for high-molecular weight compounds.