Ch-9-Carboxylic Acids and their derivatives new
... Carboxylic acids are the most acidic simple organic compounds (they are stronger acids by over ten powers of ten compared to alcohols of comparable weights); also they are more acidic than phenols However, they are weak acids compared to inorganic acids (HCl or H2SO4) Adjacent electron withdrawing s ...
... Carboxylic acids are the most acidic simple organic compounds (they are stronger acids by over ten powers of ten compared to alcohols of comparable weights); also they are more acidic than phenols However, they are weak acids compared to inorganic acids (HCl or H2SO4) Adjacent electron withdrawing s ...
Organic Chemistry Introduction
... – Organometallic Reagents – Nucleophilic Substitution Reactions – Elimination Reactions ...
... – Organometallic Reagents – Nucleophilic Substitution Reactions – Elimination Reactions ...
Studies toward the Stereoselective Synthesis of the
... In the dissertation the retrosynhetic analysis of this 3,7-dimethylundecane-1,5,6-triol building block identifies a simple precursor, ethyl 2-heptenoate, as the starting material for the proposed synthetic route toward this target. The Sharpless asymmetric epoxidation reaction plays a pivotal role i ...
... In the dissertation the retrosynhetic analysis of this 3,7-dimethylundecane-1,5,6-triol building block identifies a simple precursor, ethyl 2-heptenoate, as the starting material for the proposed synthetic route toward this target. The Sharpless asymmetric epoxidation reaction plays a pivotal role i ...
Cl + CH3OH * HCl + CH2OH
... experimental data if we assign an uncertainty of a factor 4. The large uncertainty associated with use of a generic rate constant, kb, and also use of potentially inappropriate values of H and Dl is taken into account by assigning a large uncertainty to kb. Uptake to multicomponent organic mixtures ...
... experimental data if we assign an uncertainty of a factor 4. The large uncertainty associated with use of a generic rate constant, kb, and also use of potentially inappropriate values of H and Dl is taken into account by assigning a large uncertainty to kb. Uptake to multicomponent organic mixtures ...
Experiments for an Introduction to Organic Chemistry
... indicator colour just appears. (Shake thoroughly after any drop, and avoid strictly to add more NaOH solution than is absolutely necessary.) Put the reaction vessel into a warm water bath (40 °C) and shake every minute. If the sample contains an ester, the pink solution becomes colourless within som ...
... indicator colour just appears. (Shake thoroughly after any drop, and avoid strictly to add more NaOH solution than is absolutely necessary.) Put the reaction vessel into a warm water bath (40 °C) and shake every minute. If the sample contains an ester, the pink solution becomes colourless within som ...
Aldehydes and Ketones The Carbonyl Group
... Other Nomenclature Rules • In cyclic ketones, the carbonyl group is always numbered “1”; this does not need to be included in the name. The numbering continues clockwise or counterclockwise to give the lowest number for the next substituent. • Molecules with more than one ketone group are named by p ...
... Other Nomenclature Rules • In cyclic ketones, the carbonyl group is always numbered “1”; this does not need to be included in the name. The numbering continues clockwise or counterclockwise to give the lowest number for the next substituent. • Molecules with more than one ketone group are named by p ...
Ethers and Epoxides
... • Specific to allyl aryl ethers, ArOCH2CH=CH2 • Heating to 200–250°C leads to an o-allylphenol • Result is alkylation of the phenol in an ortho position ...
... • Specific to allyl aryl ethers, ArOCH2CH=CH2 • Heating to 200–250°C leads to an o-allylphenol • Result is alkylation of the phenol in an ortho position ...
II. Chemistry of Sugars
... (anion, scavenges proton) plus sodium hydride (catalyst; very strong base) to produce methyl ether (-OCH3; -OH's react as strongly nucleophilic -O -). The ether link is very stable. The oxygen at C1 also reacts (it is a hemi acetal which will be clarified soon) but the -OCH3 at C1 is atypical and hy ...
... (anion, scavenges proton) plus sodium hydride (catalyst; very strong base) to produce methyl ether (-OCH3; -OH's react as strongly nucleophilic -O -). The ether link is very stable. The oxygen at C1 also reacts (it is a hemi acetal which will be clarified soon) but the -OCH3 at C1 is atypical and hy ...
Learning Guide for Chapter 22 - Carboxylic Acids
... How could this compound be distinguished from the one shown below? O H ...
... How could this compound be distinguished from the one shown below? O H ...
Chapter 18
... Esters are named according to the parent carboxylic acid and the –ic acid is replaced with an –ate suffix, the alkyl ester substituent is named as a separate alkyl group with the name in front of the alkanoate name ...
... Esters are named according to the parent carboxylic acid and the –ic acid is replaced with an –ate suffix, the alkyl ester substituent is named as a separate alkyl group with the name in front of the alkanoate name ...
A Simple and Advantageous Protocol for the Oxidation of Alcohols
... between solvents are their stability to IBX and the slight solubility (or lack thereof) of the IBX-derived byproducts. We regard EtOAc and DCE as the solvents of choice because they are inert and all byproducts are insoluble at room temperature, such that no purification is required beyond simple fi ...
... between solvents are their stability to IBX and the slight solubility (or lack thereof) of the IBX-derived byproducts. We regard EtOAc and DCE as the solvents of choice because they are inert and all byproducts are insoluble at room temperature, such that no purification is required beyond simple fi ...
No Slide Title
... You cannot put an OH group directly onto a benzene ring by electrophilic substitution, so phenol has to be synthesised in ...
... You cannot put an OH group directly onto a benzene ring by electrophilic substitution, so phenol has to be synthesised in ...
lecture 11 catalysis_hydrogenation of alkenes
... The protons of a dihydrogen ligand are known to be more acidic than those of free H2, and many H2 complexes can be deprotonated by NEt3. ...
... The protons of a dihydrogen ligand are known to be more acidic than those of free H2, and many H2 complexes can be deprotonated by NEt3. ...
Organic Chemistry Fifth Edition
... The Baeyer-Villiger Oxidation is the oxidative cleavage of a carbon-carbon bond adjacent to a carbonyl, which converts ketones to esters and cyclic ketones to lactones. The Baeyer-Villiger can be carried out with peracids, such as MCBPA, or with hydrogen peroxide and a Lewis acid. ...
... The Baeyer-Villiger Oxidation is the oxidative cleavage of a carbon-carbon bond adjacent to a carbonyl, which converts ketones to esters and cyclic ketones to lactones. The Baeyer-Villiger can be carried out with peracids, such as MCBPA, or with hydrogen peroxide and a Lewis acid. ...
Document
... • Problem: draw condensed structural formulas for the two aldehydes of molecular formula C4H8O • Solution: • first draw the functional group of an aldehyde and add the remaining three carbons; these may be bonded in two ways. • then add the seven hydrogens necessary to complete the four bonds of eac ...
... • Problem: draw condensed structural formulas for the two aldehydes of molecular formula C4H8O • Solution: • first draw the functional group of an aldehyde and add the remaining three carbons; these may be bonded in two ways. • then add the seven hydrogens necessary to complete the four bonds of eac ...
Classes and Nomenclature of Halogen Compounds
... Reactions of Halogen Compounds C) Formation of organometallic compounds. Most organic chlorides, bromides, and iodides react with certain metals to give organometallic compounds, molecules with carbon-metal bonds. Grignard reagents are obtained by the reaction of alkyl or aryl halides with meta ...
... Reactions of Halogen Compounds C) Formation of organometallic compounds. Most organic chlorides, bromides, and iodides react with certain metals to give organometallic compounds, molecules with carbon-metal bonds. Grignard reagents are obtained by the reaction of alkyl or aryl halides with meta ...
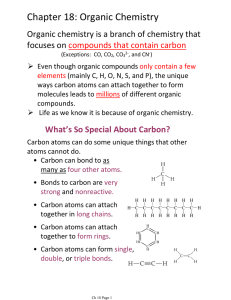
Chapter 18: Organic Chemistry
... 1. The names of esters are derived from the name of the carboxylic acid from which they are formed. Drop the 'ic acid' ending of the carboylic acid name and add the ending 'ate'. (This gives the ester base name). 2. The group attached to the oxygen of the ester is then named as an alkyl group. This ...
... 1. The names of esters are derived from the name of the carboxylic acid from which they are formed. Drop the 'ic acid' ending of the carboylic acid name and add the ending 'ate'. (This gives the ester base name). 2. The group attached to the oxygen of the ester is then named as an alkyl group. This ...
The Grignard Reagent
... – Be careful not to overheat the reaction. Remember ether boils at 35°C. Do not confuse the reaction starting with the ether boiling. You will see a cloudiness and change in color when the reaction has started. – Use your time wisely. While one partner monitors reaction 1 the other should prepare th ...
... – Be careful not to overheat the reaction. Remember ether boils at 35°C. Do not confuse the reaction starting with the ether boiling. You will see a cloudiness and change in color when the reaction has started. – Use your time wisely. While one partner monitors reaction 1 the other should prepare th ...
An Overview of Carbonyl Compound Chemistry
... as the final products. For example, when organometallic compounds, Grignard or organolithium reagents, are used as the carbanion species, they can react with the ketone or aldehyde intermediates very rapidly to form a second tetrahedral intermediate, in which no good leaving groups are present. Over ...
... as the final products. For example, when organometallic compounds, Grignard or organolithium reagents, are used as the carbanion species, they can react with the ketone or aldehyde intermediates very rapidly to form a second tetrahedral intermediate, in which no good leaving groups are present. Over ...
Microwave-Assisted Esterification of N -Acetyl-L-Phenylalanine Using Modified Mukaiyama s Reagents: A New Approach Involving Ionic Liquids
... (such as p-toulenesulfonic acid [4-5] or benzenesulfonic acid [6]). These reactions are equilibriumdriven and generate huge amounts of salts such as NaCl or Na2SO4 during large-scale productions [7]. An improvement made by Wegman et al. [7] was the utilization of acid form of ultrastable zeolite Y ( ...
... (such as p-toulenesulfonic acid [4-5] or benzenesulfonic acid [6]). These reactions are equilibriumdriven and generate huge amounts of salts such as NaCl or Na2SO4 during large-scale productions [7]. An improvement made by Wegman et al. [7] was the utilization of acid form of ultrastable zeolite Y ( ...
Carboxylic acid-Group A
... An amide has an NH2, NHR, or NHR2 group in place of OH group. Amides are named by replacing “oic acid”, “ic acid” or “ylic acid” of the acid name with “amide” ...
... An amide has an NH2, NHR, or NHR2 group in place of OH group. Amides are named by replacing “oic acid”, “ic acid” or “ylic acid” of the acid name with “amide” ...
Alcohol

In chemistry, an alcohol is any organic compound in which the hydroxyl functional group (–OH) is bound to a saturated carbon atom. The term alcohol originally referred to the primary alcohol ethyl alcohol (ethanol), the predominant alcohol in alcoholic beverages.The suffix -ol appears in the IUPAC chemical name of all substances where the hydroxyl group is the functional group with the highest priority; in substances where a higher priority group is present the prefix hydroxy- will appear in the IUPAC name. The suffix -ol in non-systematic names (such as paracetamol or cholesterol) also typically indicates that the substance includes a hydroxyl functional group and, so, can be termed an alcohol. But many substances, particularly sugars (examples glucose and sucrose) contain hydroxyl functional groups without using the suffix. An important class of alcohols, of which methanol and ethanol are the simplest members is the saturated straight chain alcohols, the general formula for which is CnH2n+1OH.