Aldehydes and Ketones
... Some ketones can also be prepared from acyl halides and organo-copper reagents (called lithium dialkylcuprates), as shown below: O O CuBr R Cl Li R Li Cu CuLi ...
... Some ketones can also be prepared from acyl halides and organo-copper reagents (called lithium dialkylcuprates), as shown below: O O CuBr R Cl Li R Li Cu CuLi ...
SAMPL E PA GES
... This is much lower than the value given in the Data Booklet. The reasons for this include: • heat loss to the surroundings, including the air, the copper can and the thermometer • the ethanol is burning in air rather than in pure oxygen and so undergoes incomplete combustion • the mass of ethanol ...
... This is much lower than the value given in the Data Booklet. The reasons for this include: • heat loss to the surroundings, including the air, the copper can and the thermometer • the ethanol is burning in air rather than in pure oxygen and so undergoes incomplete combustion • the mass of ethanol ...
Arenes HW
... Salicylic acid is used in the manufacture of aspirin tablets. In the UK around 3500 tonnes of salicylic acid are manufactured per year. ...
... Salicylic acid is used in the manufacture of aspirin tablets. In the UK around 3500 tonnes of salicylic acid are manufactured per year. ...
Chromatography Spectroscopy HW
... Salicylic acid is used in the manufacture of aspirin tablets. In the UK around 3500 tonnes of salicylic acid are manufactured per year. ...
... Salicylic acid is used in the manufacture of aspirin tablets. In the UK around 3500 tonnes of salicylic acid are manufactured per year. ...
Carboxylic Acids - BSAK Chemistry weebly
... temperature producing an ester and misty fumes of hydrogen chloride. • Add the liquid ethanoyl chloride to ethanol, you get a burst of hydrogen chloride produced together with the liquid ester ethyl ethanoate. ...
... temperature producing an ester and misty fumes of hydrogen chloride. • Add the liquid ethanoyl chloride to ethanol, you get a burst of hydrogen chloride produced together with the liquid ester ethyl ethanoate. ...
Unit F322 - Chains, energy and resources - Visually impaired
... very little carbon monoxide, CO, is formed but a significant amount of nitrogen monoxide, NO, is formed. (i) Construct the equation to show the COMPLETE combustion of C10H22. _____________________________________ [2] (ii) Suggest, with the aid of an equation, how NO is formed within an aeroplane eng ...
... very little carbon monoxide, CO, is formed but a significant amount of nitrogen monoxide, NO, is formed. (i) Construct the equation to show the COMPLETE combustion of C10H22. _____________________________________ [2] (ii) Suggest, with the aid of an equation, how NO is formed within an aeroplane eng ...
N.b. A catalyst is a species which speeds up a chemical reaction but
... Common oxidising agents [O] are typically metal based oxidants in which the metal is in a high oxidation state. e.g. i) Chromium(vi) in acid: Chromium is reduced from Cr(vi) to Cr(iii). ii) Manganese (vii) in permanganate (MnO4-) Manganese (vi) in manganate (MnO42-) Manganese is typically reduced to ...
... Common oxidising agents [O] are typically metal based oxidants in which the metal is in a high oxidation state. e.g. i) Chromium(vi) in acid: Chromium is reduced from Cr(vi) to Cr(iii). ii) Manganese (vii) in permanganate (MnO4-) Manganese (vi) in manganate (MnO42-) Manganese is typically reduced to ...
3.5 revision guide alcohols
... At higher temperatures the yeast dies and the enzymes denature. Fermentation is done in an absence of air because the presence of air can cause extra reactions to occur. It oxidises the ethanol produced to ethanoic acid (vinegar). ...
... At higher temperatures the yeast dies and the enzymes denature. Fermentation is done in an absence of air because the presence of air can cause extra reactions to occur. It oxidises the ethanol produced to ethanoic acid (vinegar). ...
File - Garbally Chemistry
... • For the initiation step 1. The reaction will not occur in the dark at room temperature. It will occur at room temperature if ultraviolet light is shone on the reactants. ...
... • For the initiation step 1. The reaction will not occur in the dark at room temperature. It will occur at room temperature if ultraviolet light is shone on the reactants. ...
Chapter 24. Amines
... Most simple alkylammmonium ions have pKa's of 10 to 11 Arylamines and heterocyclic aromatic amines are considerably less basic than alkylamines (conjugate acid pKa 5 or less) ...
... Most simple alkylammmonium ions have pKa's of 10 to 11 Arylamines and heterocyclic aromatic amines are considerably less basic than alkylamines (conjugate acid pKa 5 or less) ...
Synthesis of (−)-Epibatidine - David A. Evans
... conformational effects play an important role in the stereochemical outcome of this reduction. The completion of the synthesis required inverting the equatorial alcohol 16 in order to facilitate subsequent closure to the 7-azabicylco[2.2.1]heptane system. Accordingly, alcohols 16/17 were converted i ...
... conformational effects play an important role in the stereochemical outcome of this reduction. The completion of the synthesis required inverting the equatorial alcohol 16 in order to facilitate subsequent closure to the 7-azabicylco[2.2.1]heptane system. Accordingly, alcohols 16/17 were converted i ...
Document
... Spectra of Aldehydes and Ketones: The 1H chemical shift range for the aldehyde proton is 9-10 ppm The aldehyde proton will couple to the protons on the -carbon with a typical coupling constant of J 2 Hz A carbonyl will slightly deshield the protons on the -carbon; typical chemical shift range ...
... Spectra of Aldehydes and Ketones: The 1H chemical shift range for the aldehyde proton is 9-10 ppm The aldehyde proton will couple to the protons on the -carbon with a typical coupling constant of J 2 Hz A carbonyl will slightly deshield the protons on the -carbon; typical chemical shift range ...
nomenclature continued… - Turner Fenton Secondary School
... reacting a carboxylic acid with an amine group (ammonia or an amine). Naming: 1. First name the chain that contains that carbonyl. Replace –oic acid with an AMIDE. C=O group is number one on the chain. (BASE) 2. Naming of the alkyl chain depends on whether the amine is primary, secondary or tertiary ...
... reacting a carboxylic acid with an amine group (ammonia or an amine). Naming: 1. First name the chain that contains that carbonyl. Replace –oic acid with an AMIDE. C=O group is number one on the chain. (BASE) 2. Naming of the alkyl chain depends on whether the amine is primary, secondary or tertiary ...
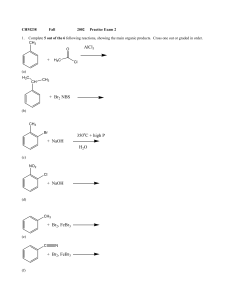
+ NaOH 350 C + high P H2O + H3C AlCl3 + NaOH + Br2, FeBr3
... processes can be accomplished with 2 steps, but there is more than one correct answer for each. Assume that ortho and para isomers can be separated. OH O CH3 ...
... processes can be accomplished with 2 steps, but there is more than one correct answer for each. Assume that ortho and para isomers can be separated. OH O CH3 ...
Article Summaries
... that bpym is a -acid. Using the bypm ligand prevents precipitation of insoluble platinum salts from solution. This system is catalytic in the Pt complex, with SO3 as the formal oxidant. The mechanism for this reaction is quite similar to that of the original Shilov reaction, although the details of ...
... that bpym is a -acid. Using the bypm ligand prevents precipitation of insoluble platinum salts from solution. This system is catalytic in the Pt complex, with SO3 as the formal oxidant. The mechanism for this reaction is quite similar to that of the original Shilov reaction, although the details of ...
Aluminum Utilization for Hydrogen Production
... • Arose during studies on the synthesis of aluminum alkoxides; • The hydrogen is produced by the reaction of an alcohol with aluminum activated by I2, HgCl2 or SnCl4, under reflux conditions: 2Al + 6 ROH (excess) = 2Al(OR)3 + 3H2 ...
... • Arose during studies on the synthesis of aluminum alkoxides; • The hydrogen is produced by the reaction of an alcohol with aluminum activated by I2, HgCl2 or SnCl4, under reflux conditions: 2Al + 6 ROH (excess) = 2Al(OR)3 + 3H2 ...
Synthesis of Benzyl Acetate from Acetic Anhydride
... equilibrium can be shifted by adding more of the acid or of the alcohol, depending on cost or availability. The mechanism of the reaction involves initial protonation of the carboxyl group, attack by the nucleophilic hydroxyl, a proton transfer, and loss of water followed by loss of the catalyzing p ...
... equilibrium can be shifted by adding more of the acid or of the alcohol, depending on cost or availability. The mechanism of the reaction involves initial protonation of the carboxyl group, attack by the nucleophilic hydroxyl, a proton transfer, and loss of water followed by loss of the catalyzing p ...
Organic compounds containing Nitrogen
... *8. i. Why are amines less acidic than alcohols of comparable molecular masses? ii. Compare the basicity of the following in gaseous and aqueous state and arrange then in increasing order of basicity. CH3NH2, (CH3)2NH, (CH3)3N and NH3 Ans. i. Amines are less acidic than alcohols of comparable molecu ...
... *8. i. Why are amines less acidic than alcohols of comparable molecular masses? ii. Compare the basicity of the following in gaseous and aqueous state and arrange then in increasing order of basicity. CH3NH2, (CH3)2NH, (CH3)3N and NH3 Ans. i. Amines are less acidic than alcohols of comparable molecu ...
Chapter 20: Carboxylic Acids and Nitriles
... Most amines that have 3 different substituents on N are not resolved because the molecules interconvert by pyramidal inversion ...
... Most amines that have 3 different substituents on N are not resolved because the molecules interconvert by pyramidal inversion ...
Alcohols, Ethers, and Epoxides
... Synthesis of alkyl halides from alcohols: via strong acids: Reactivity order = 3o > 2o > 1o SN1 pathway for 2o and 3o -carbocations are intermediates ...
... Synthesis of alkyl halides from alcohols: via strong acids: Reactivity order = 3o > 2o > 1o SN1 pathway for 2o and 3o -carbocations are intermediates ...
AlCl3 in modern chemistry of polyfluoroarenes
... AlCl3 application in the chemistry of polyfluoroarenes beginning since 1995y. As substrates or reagents there are given fluorinated compounds containing two and more fluorine atoms in one aromatic ring and the transformations are systematized according to the type of the reactions taking place. 1. F ...
... AlCl3 application in the chemistry of polyfluoroarenes beginning since 1995y. As substrates or reagents there are given fluorinated compounds containing two and more fluorine atoms in one aromatic ring and the transformations are systematized according to the type of the reactions taking place. 1. F ...
Sn1 and Sn2 Chemtivity
... Because the rate determining step of an SN1 reaction is the dissociation of the alkyl halide to form a carbocation, two factors affect the rate of an SN1 reaction – the ease with which the leaving group dissociates from the carbon and the stability of the carbocation that is formed. In the previous ...
... Because the rate determining step of an SN1 reaction is the dissociation of the alkyl halide to form a carbocation, two factors affect the rate of an SN1 reaction – the ease with which the leaving group dissociates from the carbon and the stability of the carbocation that is formed. In the previous ...
Chapter 18 - Aldehydes and Ketones
... carbonyl by protonation. This reaction is not a very useful reaction in the forward process since the carbonyl hydrate will rearrange to the carbonyl. However, when a carbonyl hydrate is formed during a reaction, the resulting product will be the ketone or aldehyde. ...
... carbonyl by protonation. This reaction is not a very useful reaction in the forward process since the carbonyl hydrate will rearrange to the carbonyl. However, when a carbonyl hydrate is formed during a reaction, the resulting product will be the ketone or aldehyde. ...
Hofmann–Löffler reaction

The Hofmann–Löffler reaction (also referred to as Hofmann–Löffler–Freytag reaction, Löffler–Freytag reaction, Löffler–Hofmann reaction, as well as Löffler's method) is an organic reaction in which a cyclic amine 2 (pyrrolidine or, in some cases, piperidine) is generated by thermal or photochemical decomposition of N-halogenated amine 1 in the presence of a strong acid (concentrated sulfuric acid or concentrated CF3CO2H). The Hofmann–Löffler–Freytag reaction proceeds via an intramolecular hydrogen atom transfer to a nitrogen-centered radical and is an example of a remote intramolecular free radical C–H functionalization.