Electrochemical Investigations of W(CO) (L) and W(CO) (L) Complexes:
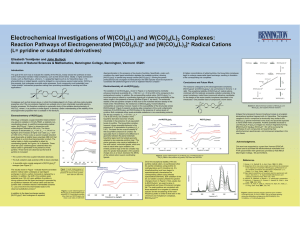
... irreversible in the presence of sub-equivalent levels of pyridine. The bis-pyridine complex, however, shows reversible behavior in the presence of 10 equivalents of pyridine (ic/ia = 0.87). The basis for the unusual stability of the bis-pyridine compound is unclear; we speculate that it is related t ...
... irreversible in the presence of sub-equivalent levels of pyridine. The bis-pyridine complex, however, shows reversible behavior in the presence of 10 equivalents of pyridine (ic/ia = 0.87). The basis for the unusual stability of the bis-pyridine compound is unclear; we speculate that it is related t ...
10. Alkyl Halides
... Reaction of RX with Mg in ether or THF Product is RMgX – an organometallic compound ...
... Reaction of RX with Mg in ether or THF Product is RMgX – an organometallic compound ...
Developments in Synthetic Application of Selenium(IV) Oxide and
... Selenium(IV) oxide allylic hydroxylations are highly regiospecific and occur at the α-position to the more substituted carbon of the double bond with a reactivity order CH2 > CH3 > CH. When the double bond is inside a ring, oxidation occurs in the ring when possible, and in the α-position to the mor ...
... Selenium(IV) oxide allylic hydroxylations are highly regiospecific and occur at the α-position to the more substituted carbon of the double bond with a reactivity order CH2 > CH3 > CH. When the double bond is inside a ring, oxidation occurs in the ring when possible, and in the α-position to the mor ...
If the substance is soluble in dil. NaOH, add a solution of NaHCO3 to
... 4.1 Introduction: Spectroscopy is the study of interaction of light with atoms and molecules. The usual range of an infrared spectrum is between 2.5-16µm (1µm = 10-4 cm). The most common method of expressing infrared spectra is to use wave number (1/π) units ranging from ...
... 4.1 Introduction: Spectroscopy is the study of interaction of light with atoms and molecules. The usual range of an infrared spectrum is between 2.5-16µm (1µm = 10-4 cm). The most common method of expressing infrared spectra is to use wave number (1/π) units ranging from ...
AP Chemistry Review Preparing for the AP
... Review your incorrect MC from the Practice Exam and understand the concepts. Know the 6 strong acids HCl, HI, HBr, H2SO4, HClO4, HNO3 and the one weak by formula acetic acid CH3COOH, everything else is weak. Remember that strong acids/bases don’t make buffers!!! You should be 100% confident what ion ...
... Review your incorrect MC from the Practice Exam and understand the concepts. Know the 6 strong acids HCl, HI, HBr, H2SO4, HClO4, HNO3 and the one weak by formula acetic acid CH3COOH, everything else is weak. Remember that strong acids/bases don’t make buffers!!! You should be 100% confident what ion ...
Oxidation-Reduction Reactions - An Introduction to Chemistry
... Phosphates, like ammonium phosphate, are important components of fertilizers used to stimulate the growth of agricultural crops and to make our gardens green. Their commercial synthesis requires elemental phosphorus, which can be acquired by heating phosphate rock (containing calcium phosphate) with ...
... Phosphates, like ammonium phosphate, are important components of fertilizers used to stimulate the growth of agricultural crops and to make our gardens green. Their commercial synthesis requires elemental phosphorus, which can be acquired by heating phosphate rock (containing calcium phosphate) with ...
Copy of Acids, bases, salts answer key
... Limitations of Arrhenius theory : Arhhenius’ theory became quite popular and was widely accepted yet it had the following limitations: This theory was applicable only to aqueous solutions. Substances like Ammonia (NH3) do not contain hydroxide (OH) ion, even then its aqueous solution acts as a ...
... Limitations of Arrhenius theory : Arhhenius’ theory became quite popular and was widely accepted yet it had the following limitations: This theory was applicable only to aqueous solutions. Substances like Ammonia (NH3) do not contain hydroxide (OH) ion, even then its aqueous solution acts as a ...
Structures and Bonding
... The Answer: 1) Scientists discovered 50 years later that the Earth generates massive amounts of heat through radioactive decay in the core. This heat generated convection currents in the mantle causing the crust to move 2) We also now know that the sea floor is spreading outwards from plate ...
... The Answer: 1) Scientists discovered 50 years later that the Earth generates massive amounts of heat through radioactive decay in the core. This heat generated convection currents in the mantle causing the crust to move 2) We also now know that the sea floor is spreading outwards from plate ...
Alkanes - MsReenChemistry
... hydrocarbons contain double or triple carbon-carbon bonds.] • Alkanes are also aliphatic, meaning the carbon atoms form open chains. [In contrast to aromatic compounds which contain benzene rings.] Alkanes contain only strong C-C (348 kJ/mol) and C-H (412 kJ/mol) bonds. Alkanes are non-polar. They h ...
... hydrocarbons contain double or triple carbon-carbon bonds.] • Alkanes are also aliphatic, meaning the carbon atoms form open chains. [In contrast to aromatic compounds which contain benzene rings.] Alkanes contain only strong C-C (348 kJ/mol) and C-H (412 kJ/mol) bonds. Alkanes are non-polar. They h ...
CHAPTER 19
... either atom has totally lost or totally gained any electrons. In the case of the formation of hydrogen chloride, for example, hydrogen simply has donated a share of its bonding electron to the chlorine; it has not completely transferred that electron. The assignment of oxidation numbers allows an ap ...
... either atom has totally lost or totally gained any electrons. In the case of the formation of hydrogen chloride, for example, hydrogen simply has donated a share of its bonding electron to the chlorine; it has not completely transferred that electron. The assignment of oxidation numbers allows an ap ...
SUPPLEMENTAL PROBLEMS FOR CHEM 110
... are found to contain the same number of protons, the same number of neutrons, and different numbers of electrons. Which of the following statements about X and Y is correct? ...
... are found to contain the same number of protons, the same number of neutrons, and different numbers of electrons. Which of the following statements about X and Y is correct? ...
Aromatic Compounds
... Rings: The Friedel-Crafts Reaction Alkylation • The introduction of an alkyl group onto the benzene ring • Called the Friedel-Crafts reaction after its discoverers • Among the most useful electrophilic aromatic substitution ...
... Rings: The Friedel-Crafts Reaction Alkylation • The introduction of an alkyl group onto the benzene ring • Called the Friedel-Crafts reaction after its discoverers • Among the most useful electrophilic aromatic substitution ...
Chemistry 209 - Experiment #4
... reaction that you will carry out in this lab. The acetone product will be purified using a distillation. CH3 ...
... reaction that you will carry out in this lab. The acetone product will be purified using a distillation. CH3 ...
Organic Chemistry Organic Chemistry
... almost any size and can form combinations of single, double, and triple covalent bonds. This versatility allows the formation of a huge variety of very large organic molecules. In this chapter, we will examine the characteristic physical properties of families of organic molecules, and relate these ...
... almost any size and can form combinations of single, double, and triple covalent bonds. This versatility allows the formation of a huge variety of very large organic molecules. In this chapter, we will examine the characteristic physical properties of families of organic molecules, and relate these ...
Materials Chemistry Prof. S. Sunder Manoharan Department of
... Now, when we think about material synthesis first we need to understand making materials is a big word, and the group of people who are engaged in making solids are host of groups, who involved in variety of solid state materials chemistry synthesis. Now, here I have listed some of the portfolio of ...
... Now, when we think about material synthesis first we need to understand making materials is a big word, and the group of people who are engaged in making solids are host of groups, who involved in variety of solid state materials chemistry synthesis. Now, here I have listed some of the portfolio of ...
kinetic characterisation of catalysts for methanol synthesis
... where rMexp,i , rMcal,i is the methanol formation rate determined experimentally and calculated from Eq. (4), respectively, and M is the number of experimental data obtained at different temperatures and reagent ...
... where rMexp,i , rMcal,i is the methanol formation rate determined experimentally and calculated from Eq. (4), respectively, and M is the number of experimental data obtained at different temperatures and reagent ...
Chabot College
... reactions involving hydrocarbons, alkyl halides, alcohols and ethers; 3. explain physical and chemical properties of groups studied based on structural analysis; 4. use spectroscopic data from infrared spectroscopy, 1H nuclear magnetic spectroscopy to elucidate structures for organic compounds; 5. i ...
... reactions involving hydrocarbons, alkyl halides, alcohols and ethers; 3. explain physical and chemical properties of groups studied based on structural analysis; 4. use spectroscopic data from infrared spectroscopy, 1H nuclear magnetic spectroscopy to elucidate structures for organic compounds; 5. i ...
Visible light photooxidation of nitrate: the dawn of
... to explore the synthetic application of this new method and to compare it with the previously reported methods. The results are compiled in Table 1. Under photocatalytic conditions using 5 mol% of Acr+-Mes (1), 0.25 mmol of alkyne 2 and 2 eq. of LiNO3, diketone 3 and ketone 4 were obtained after 2 h ...
... to explore the synthetic application of this new method and to compare it with the previously reported methods. The results are compiled in Table 1. Under photocatalytic conditions using 5 mol% of Acr+-Mes (1), 0.25 mmol of alkyne 2 and 2 eq. of LiNO3, diketone 3 and ketone 4 were obtained after 2 h ...
Slide 1 - Alfred State College intranet site
... Formaldehyde (CH2═O) is the simplest aldehyde: •It is sold as formalin, a 37% aqueous solution used to preserve biological specimens. ...
... Formaldehyde (CH2═O) is the simplest aldehyde: •It is sold as formalin, a 37% aqueous solution used to preserve biological specimens. ...
+ Br2, FeBr3 + Br2, FeBr3
... (c) If we used only pure (fuming) sulfuric acid, what would be the product(s)? mostly sulfonation of Cl benzene, both o and p, because SO3H+ becomes the superelectrophile and there is not as much protons for the dehydration of nitric acid. (d) Chlorine is o,p directing group but chlorobenzene is slo ...
... (c) If we used only pure (fuming) sulfuric acid, what would be the product(s)? mostly sulfonation of Cl benzene, both o and p, because SO3H+ becomes the superelectrophile and there is not as much protons for the dehydration of nitric acid. (d) Chlorine is o,p directing group but chlorobenzene is slo ...
Preparation and Physical Properties of Chitosan Benzoic Acid
... Chitin, the second abundant natural polymer in Nature, and chitosan, a partially deacetylated form of chitin, have recently received much of attention owing to their applicability in wide range of fields such as pharmaceuticals, cosmetics, agriculture, foods, and material sciences [1–5]. Their film ...
... Chitin, the second abundant natural polymer in Nature, and chitosan, a partially deacetylated form of chitin, have recently received much of attention owing to their applicability in wide range of fields such as pharmaceuticals, cosmetics, agriculture, foods, and material sciences [1–5]. Their film ...
Carbon Bond - Rutgers Chemistry
... of a functionalized aromatic compound, such as an aryl halide, triflate, or boron compound, before it can be used in the carbon-carbon coupling reaction. A major step forward in efficiency and atom economy would be achieved if direct coupling between an organic substrate and a carbon atom at a C-H p ...
... of a functionalized aromatic compound, such as an aryl halide, triflate, or boron compound, before it can be used in the carbon-carbon coupling reaction. A major step forward in efficiency and atom economy would be achieved if direct coupling between an organic substrate and a carbon atom at a C-H p ...
Strychnine total synthesis

Strychnine total synthesis in chemistry describes the total synthesis of the complex biomolecule strychnine. The first reported method by the group of Robert Burns Woodward in 1954 is considered a classic in this research field. At the time it formed the natural conclusion to an elaborate process of molecular structure elucidation that started with the isolation of strychnine from the beans of Strychnos ignatii by Pierre Joseph Pelletier and Joseph Bienaimé Caventou in 1818. Major contributors to the entire effort were Sir Robert Robinson with over 250 publications and Hermann Leuchs with another 125 papers in a time span of 40 years. Robinson was awarded the Nobel Prize in Chemistry in 1947 for his work on alkaloids, strychnine included. The process of chemical identification was completed with publications in 1946 by Robinson and later confirmed by Woodward in 1947. X-ray structures establishing the absolute configuration became available between 1947 and 1951 with publications from J. M. Bijvoet and J.H. Robertson .Woodward published a very brief account on the strychnine synthesis in 1954 (just 3 pages) and a lengthy one (42 pages) in 1963.Many more methods exist and reported by the research groups of Magnus, Overman, Kuehne, Rawal, Bosch, Vollhardt, Mori, Shibasaki, Li, Fukuyama Vanderwal and MacMillan. Synthetic (+)-strychnine is also known. Racemic synthesises were published by Padwa in 2007 and in 2010 by Andrade and by Reissig.In his 1963 publication Woodward quoted Sir Robert Robinson who said for its molecular size it is the most complex substance known.