Essential oils - Uddingston Grammar School
... Terpenes are components in a wide variety of fruit and floral flavours and aromas. Terpenes can be oxidised within plants to produce some of the compounds responsible for the distinctive aroma of spices. Ultraviolet radiation (UV) is a high-energy form of light present in sunlight. Exposure to UV li ...
... Terpenes are components in a wide variety of fruit and floral flavours and aromas. Terpenes can be oxidised within plants to produce some of the compounds responsible for the distinctive aroma of spices. Ultraviolet radiation (UV) is a high-energy form of light present in sunlight. Exposure to UV li ...
1.24 calculations and chemical reactions
... The magnesium oxide, produced was reacted with hydrochloric acid. MgO + 2HCl MgCl2 + H2O This sample of magnesium oxide required 33.2cm3 of hydrochloric acid for complete reaction. Calculate the concentration, in mol dm–3, of the hydrochloric acid. 5.2) Sodium carbonate is manufactured in a two-stag ...
... The magnesium oxide, produced was reacted with hydrochloric acid. MgO + 2HCl MgCl2 + H2O This sample of magnesium oxide required 33.2cm3 of hydrochloric acid for complete reaction. Calculate the concentration, in mol dm–3, of the hydrochloric acid. 5.2) Sodium carbonate is manufactured in a two-stag ...
3.0 Properties of Phosgene
... shock-sensitive compound. Isopropyl Alcohol - The reaction between isopropyl alcohol and phosgene forms isopropyl chloroformate and hydrogen chloride. At temperatures slightly above ambient isopropyl, chloroformate can decompose explosively in the presence of iron salts. Secondary Amines - Phosgene ...
... shock-sensitive compound. Isopropyl Alcohol - The reaction between isopropyl alcohol and phosgene forms isopropyl chloroformate and hydrogen chloride. At temperatures slightly above ambient isopropyl, chloroformate can decompose explosively in the presence of iron salts. Secondary Amines - Phosgene ...
幻灯片 1
... • Like the SN2 reaction, the E2 reaction takes place in one step without intermediate. As the attacking base begins to abstract H+ from a carbon next the leaving group, the C-H bond begins to break, a C=C bond begins to form, and the leaving group begins to depart, taking with it the electron pair f ...
... • Like the SN2 reaction, the E2 reaction takes place in one step without intermediate. As the attacking base begins to abstract H+ from a carbon next the leaving group, the C-H bond begins to break, a C=C bond begins to form, and the leaving group begins to depart, taking with it the electron pair f ...
SUMHAKT The thesis consists of two parts composed
... oil has been determined by a combination of methods involving TLC, GLC and lipolysis. Presence of acetic acid in the fractionated oil was confirmed by IR and NMR spectroscopic analyses. On the basis of these results it was confirmed that in the monoacetotriacylglycerols fraction the acetate group is ...
... oil has been determined by a combination of methods involving TLC, GLC and lipolysis. Presence of acetic acid in the fractionated oil was confirmed by IR and NMR spectroscopic analyses. On the basis of these results it was confirmed that in the monoacetotriacylglycerols fraction the acetate group is ...
COMPOUNDS OF CARBON CONTAINING NITROGEN
... Similarly, amides are reduced by LiAlH4 to primary amines having same number of carbon atoms as in the starting amide. For example, ethanamide gives ethanamine on reduction. O ...
... Similarly, amides are reduced by LiAlH4 to primary amines having same number of carbon atoms as in the starting amide. For example, ethanamide gives ethanamine on reduction. O ...
Chapter 8 I. Nucleophilic Substitution
... Physicist Roland Wester and his team in Matthias Weidemüller's group at the University of Freiburg, in Germany, in collaboration with William L. Hase's group at Texas Tech University, provide direct evidence for this mechanism in the gas phase. However, they also detected an additional, unexpected m ...
... Physicist Roland Wester and his team in Matthias Weidemüller's group at the University of Freiburg, in Germany, in collaboration with William L. Hase's group at Texas Tech University, provide direct evidence for this mechanism in the gas phase. However, they also detected an additional, unexpected m ...
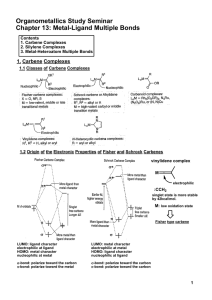
Metal-Ligand Multiple Bonds
... In general, multiple bonds to silicon and other second-row main group elements are less stable than multiple bonds to carbon, oxygen and nitrogen. ex) Si=O, M=Si: unstable ...
... In general, multiple bonds to silicon and other second-row main group elements are less stable than multiple bonds to carbon, oxygen and nitrogen. ex) Si=O, M=Si: unstable ...
7. Organic halides
... Conformation and reactivity of cycloalkanes Experimental observations show that cyclopropane (and, to a lesser extent, cyclobutane) differ in reactivity from the larger cycloalkanes and acyclic alkanes. Cyclopropane exhibits easy ring opening (see p. 20) instead of substitution characteristic of alk ...
... Conformation and reactivity of cycloalkanes Experimental observations show that cyclopropane (and, to a lesser extent, cyclobutane) differ in reactivity from the larger cycloalkanes and acyclic alkanes. Cyclopropane exhibits easy ring opening (see p. 20) instead of substitution characteristic of alk ...
992_4th_ Exam_1000615
... C) 3-methylbutene D) 2-methyl-2-butene and 3-methylbutene E) 2-methyl-2-butene Answer: D 36) Choose the INCORRECT statement. A) Salts of fatty acids are called soaps. B) Glycerides can be hydrolyzed in alkaline solution to glycerol and salts of fatty acids. C) Saturated fatty acids have multiple dou ...
... C) 3-methylbutene D) 2-methyl-2-butene and 3-methylbutene E) 2-methyl-2-butene Answer: D 36) Choose the INCORRECT statement. A) Salts of fatty acids are called soaps. B) Glycerides can be hydrolyzed in alkaline solution to glycerol and salts of fatty acids. C) Saturated fatty acids have multiple dou ...
Lipid Hydroperoxide Activation of N-Hydroxy-N
... Chart 1 demonstrates the free radical signal obtained with methemoglobin- and hematin-catalyzed oxidation of N-OH AAF by LAHP. We compute a g value of 2.0063 forthe free ...
... Chart 1 demonstrates the free radical signal obtained with methemoglobin- and hematin-catalyzed oxidation of N-OH AAF by LAHP. We compute a g value of 2.0063 forthe free ...
Naming organic compounds
... The functional group in the alcohols is the hydroxyl group (-OH). Alcohols end in the letters -ol ...
... The functional group in the alcohols is the hydroxyl group (-OH). Alcohols end in the letters -ol ...
Reactions of Alcohol
... When the positive end of one of the dipoles is a hydrogen atom bonded to O or N (atoms of high electronegativity) and the negative end of the other dipole is an O or N atom, the attractive interaction between dipoles is particularly strong and is given the special name of hydrogen bonding. ...
... When the positive end of one of the dipoles is a hydrogen atom bonded to O or N (atoms of high electronegativity) and the negative end of the other dipole is an O or N atom, the attractive interaction between dipoles is particularly strong and is given the special name of hydrogen bonding. ...
ΔG - Lemon Bay High School
... the cooler water. The final temperature, after the metal and water achieve the same temperature (thermal equilibrium), will be somewhere between the initial temperatures of the metal and the water. (b) Experience tells us that this process is not spontaneous—we certainly have never seen hydrogen and ...
... the cooler water. The final temperature, after the metal and water achieve the same temperature (thermal equilibrium), will be somewhere between the initial temperatures of the metal and the water. (b) Experience tells us that this process is not spontaneous—we certainly have never seen hydrogen and ...
chapter 12_13_14_16_17 Organic Nomenclature
... 13.2 Naming Alkenes and Alkynes ► Very similar rules as for alkanes ► Additional rules: ►Choose longest chain that contains the double or triple bond ►Suffix for alkenes: -ene ►Suffix for alkynes: -yne ►Include a number with the parent to indicate which carbon the double/triple bond starts on. ...
... 13.2 Naming Alkenes and Alkynes ► Very similar rules as for alkanes ► Additional rules: ►Choose longest chain that contains the double or triple bond ►Suffix for alkenes: -ene ►Suffix for alkynes: -yne ►Include a number with the parent to indicate which carbon the double/triple bond starts on. ...
Slide 1
... the cooler water. The final temperature, after the metal and water achieve the same temperature (thermal equilibrium), will be somewhere between the initial temperatures of the metal and the water. (b) Experience tells us that this process is not spontaneous—we certainly have never seen hydrogen and ...
... the cooler water. The final temperature, after the metal and water achieve the same temperature (thermal equilibrium), will be somewhere between the initial temperatures of the metal and the water. (b) Experience tells us that this process is not spontaneous—we certainly have never seen hydrogen and ...
HOMOLOGATION OF HETEROCYCLES BY A SEQUENTIAL REDUCTIVE OPENING LITHIATION – S
... In sharp contrast to the behaviour of epoxides and oxetanes, tetrahydrofuran (26a) do not undergo reductive opening by means of lithium metal itself and in the presence of arenes as electron carriers at low temperatures. However, it is also possible to carry out this process at low temperature but n ...
... In sharp contrast to the behaviour of epoxides and oxetanes, tetrahydrofuran (26a) do not undergo reductive opening by means of lithium metal itself and in the presence of arenes as electron carriers at low temperatures. However, it is also possible to carry out this process at low temperature but n ...
Handout V
... While comparing cyclohexanamine and aniline, aniline is a weak base because the lone pair can be delocalized into the benzene ring (Figure 7). In order for the lone pair to be fully conjugated with the benzene ring, the nitrogen would have to be sp2 hybridized with the lone pair in the p-orbital. Th ...
... While comparing cyclohexanamine and aniline, aniline is a weak base because the lone pair can be delocalized into the benzene ring (Figure 7). In order for the lone pair to be fully conjugated with the benzene ring, the nitrogen would have to be sp2 hybridized with the lone pair in the p-orbital. Th ...
Teaching to Standards: Science
... set them next to the packages (e.g., place the plastic bag with salt next to the container of salt) in your work area. Using a permanent marker, write S, B, or F on the cups so each student will have a set of each. ...
... set them next to the packages (e.g., place the plastic bag with salt next to the container of salt) in your work area. Using a permanent marker, write S, B, or F on the cups so each student will have a set of each. ...
chm121 tutorial kit - Covenant University
... Markovnikov rule obeys addition reactions with ….. alkyne (a)symmetrical (b)similar (c) parallel (d)unsymmetrical Carboxylic acid reacts with alcohol under acid catalysis to produce (a) ether (b)ester(c) mineral acid (d) coke The molecular formular for 2-pentyne is (a) CH3CH2C CCH3 (b) CH3CH2C CH (c ...
... Markovnikov rule obeys addition reactions with ….. alkyne (a)symmetrical (b)similar (c) parallel (d)unsymmetrical Carboxylic acid reacts with alcohol under acid catalysis to produce (a) ether (b)ester(c) mineral acid (d) coke The molecular formular for 2-pentyne is (a) CH3CH2C CCH3 (b) CH3CH2C CH (c ...
Influence of alkyl chain length on sulfated zirconia catalysed batch
... their acidity and thereby improve oil stability.6 Similar pretreatments are required to remove C12–C20 fatty acids from waste cooking, non-edible plant and algal oils to facilitate biodiesel production.3 Biodiesel typically comprises fatty acid methyl esters (FAMEs) formed via the esterification and ...
... their acidity and thereby improve oil stability.6 Similar pretreatments are required to remove C12–C20 fatty acids from waste cooking, non-edible plant and algal oils to facilitate biodiesel production.3 Biodiesel typically comprises fatty acid methyl esters (FAMEs) formed via the esterification and ...
Synthesis of PbS Nanoclusters within Block Copolymer Nanoreactors
... solution. Since much faster loading takes place using organometallic reagents, tetraethyllead was chosen as the reagent for loading lead. Organometallic reagents such as dimethylcadmium, and diethylzinc have previously been used by Yue et al.29 to load diblock copolymer nanoreactors. Loading of lead ...
... solution. Since much faster loading takes place using organometallic reagents, tetraethyllead was chosen as the reagent for loading lead. Organometallic reagents such as dimethylcadmium, and diethylzinc have previously been used by Yue et al.29 to load diblock copolymer nanoreactors. Loading of lead ...
Strychnine total synthesis

Strychnine total synthesis in chemistry describes the total synthesis of the complex biomolecule strychnine. The first reported method by the group of Robert Burns Woodward in 1954 is considered a classic in this research field. At the time it formed the natural conclusion to an elaborate process of molecular structure elucidation that started with the isolation of strychnine from the beans of Strychnos ignatii by Pierre Joseph Pelletier and Joseph Bienaimé Caventou in 1818. Major contributors to the entire effort were Sir Robert Robinson with over 250 publications and Hermann Leuchs with another 125 papers in a time span of 40 years. Robinson was awarded the Nobel Prize in Chemistry in 1947 for his work on alkaloids, strychnine included. The process of chemical identification was completed with publications in 1946 by Robinson and later confirmed by Woodward in 1947. X-ray structures establishing the absolute configuration became available between 1947 and 1951 with publications from J. M. Bijvoet and J.H. Robertson .Woodward published a very brief account on the strychnine synthesis in 1954 (just 3 pages) and a lengthy one (42 pages) in 1963.Many more methods exist and reported by the research groups of Magnus, Overman, Kuehne, Rawal, Bosch, Vollhardt, Mori, Shibasaki, Li, Fukuyama Vanderwal and MacMillan. Synthetic (+)-strychnine is also known. Racemic synthesises were published by Padwa in 2007 and in 2010 by Andrade and by Reissig.In his 1963 publication Woodward quoted Sir Robert Robinson who said for its molecular size it is the most complex substance known.