Sulfuric Acid
... impurities, the molten material has to be filtered to avoid poisoning the catalyst and forming water from burning hydrogen. Step 2. When using sulfur from sources 1 and 2, purification of the SO 2 gas is normally not needed. Other sources of SO2 require wet scrubbing followed by treatment of the gas ...
... impurities, the molten material has to be filtered to avoid poisoning the catalyst and forming water from burning hydrogen. Step 2. When using sulfur from sources 1 and 2, purification of the SO 2 gas is normally not needed. Other sources of SO2 require wet scrubbing followed by treatment of the gas ...
Chapter 16, Amines
... ¾Amines are polar, hydrogen bond with water, and 1Eand 2E with each other, but more weakly than alcohols ¾Amines have lower boiling points than alcohols of similar molecular weight, they are also soluble in water if the substituent group part is not too large and non-polar. C1-C7 = miscible to sligh ...
... ¾Amines are polar, hydrogen bond with water, and 1Eand 2E with each other, but more weakly than alcohols ¾Amines have lower boiling points than alcohols of similar molecular weight, they are also soluble in water if the substituent group part is not too large and non-polar. C1-C7 = miscible to sligh ...
alcohols - A-Level Chemistry
... Identify the organic product or products formed by the following dehydration reactions and write an equation for the reaction: (i) ...
... Identify the organic product or products formed by the following dehydration reactions and write an equation for the reaction: (i) ...
Document
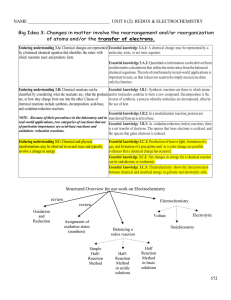
... important to note, so that it does not seem to be simply an exercise done only by chemists. Enduring understanding 3.B: Chemical reactions can be Essential knowledge 3.B.1: Synthesis reactions are those in which atoms classified by considering what the reactants are, what the products and/or molecul ...
... important to note, so that it does not seem to be simply an exercise done only by chemists. Enduring understanding 3.B: Chemical reactions can be Essential knowledge 3.B.1: Synthesis reactions are those in which atoms classified by considering what the reactants are, what the products and/or molecul ...
Full-Text PDF
... aromatic ring, as Calderazzo and co-workers proposed [32]. Indeed, another interesting point was the observed dimerization of two aromatic rings, affording diphenylmethanol compounds, as the proposed mechanism indicates (Scheme 3). ...
... aromatic ring, as Calderazzo and co-workers proposed [32]. Indeed, another interesting point was the observed dimerization of two aromatic rings, affording diphenylmethanol compounds, as the proposed mechanism indicates (Scheme 3). ...
17_fermentation-and-tca
... mitochondria • Some tumor cells overproduce several glycolytic enzymes due to the presence of HIF-1 • HIF-1 acts at the level of mRNA synthesis to stimulate the production of at least 8 glycolytic enzymes ...
... mitochondria • Some tumor cells overproduce several glycolytic enzymes due to the presence of HIF-1 • HIF-1 acts at the level of mRNA synthesis to stimulate the production of at least 8 glycolytic enzymes ...
Document
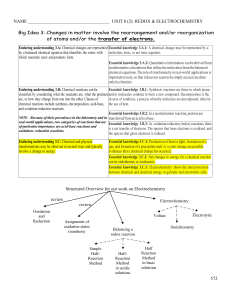
... important to note, so that it does not seem to be simply an exercise done only by chemists. Enduring understanding 3.B: Chemical reactions can be Essential knowledge 3.B.1: Synthesis reactions are those in which atoms classified by considering what the reactants are, what the products and/or molecul ...
... important to note, so that it does not seem to be simply an exercise done only by chemists. Enduring understanding 3.B: Chemical reactions can be Essential knowledge 3.B.1: Synthesis reactions are those in which atoms classified by considering what the reactants are, what the products and/or molecul ...
Metabolism Phase-I
... NH2, SH) into the molecule to produce a more water soluble compound. The compound now either be polar enough to be excreted or may undergo phase-II reactions In this step drugs undergoes functionalization reaction of oxidation, reduction or hydrolysis. Phase-I oxidation reactions are catalyzed ...
... NH2, SH) into the molecule to produce a more water soluble compound. The compound now either be polar enough to be excreted or may undergo phase-II reactions In this step drugs undergoes functionalization reaction of oxidation, reduction or hydrolysis. Phase-I oxidation reactions are catalyzed ...
Spontaniety Worked Examples
... metal to the cooler water. The final temperature, after the metal and water achieve the same temperature (thermal equilibrium), will be somewhere between the initial temperatures of the metal and the water. (b) Experience tells us that this process is not spontaneous— we certainly have never seen hy ...
... metal to the cooler water. The final temperature, after the metal and water achieve the same temperature (thermal equilibrium), will be somewhere between the initial temperatures of the metal and the water. (b) Experience tells us that this process is not spontaneous— we certainly have never seen hy ...
The reaction pathways of hydrogen peroxide in
... enthalpy, entropy and free energy of the transition states of the formation and breakdown of the intermediate have been calculated. The metal-catalyzed pathway of hydrogen peroxide is dealing with the effect of hydroxyl radicals created by the Fenton reaction and their potential to oxidize the disul ...
... enthalpy, entropy and free energy of the transition states of the formation and breakdown of the intermediate have been calculated. The metal-catalyzed pathway of hydrogen peroxide is dealing with the effect of hydroxyl radicals created by the Fenton reaction and their potential to oxidize the disul ...
Kinetics
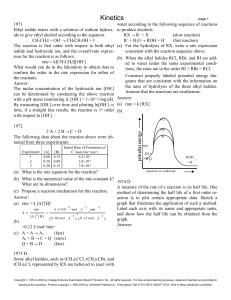
... Ethyl iodide reacts with a solution of sodium hydroxide to give ethyl alcohol according to the equation. CH3CH2I + OH- CH3CH2OH + IThe reaction is first order with respect to both ethyl iodide and hydroxide ion, and the overall-rate expression for the reaction is as follows: rate = k[CH3CH2I][OH-] ...
... Ethyl iodide reacts with a solution of sodium hydroxide to give ethyl alcohol according to the equation. CH3CH2I + OH- CH3CH2OH + IThe reaction is first order with respect to both ethyl iodide and hydroxide ion, and the overall-rate expression for the reaction is as follows: rate = k[CH3CH2I][OH-] ...
C:\SUBJECTS\SUBJECTS\Chemistry
... yeast and in the absence of air X is converted to compound Y in the absence of air, X is converted to compound Y and colourless gas. Compound Y reacts with sodium metal to produce a gas Z which gives a ‘pop’ sound with a glowing splint. Y also reacts with ethanoic acid to give a sweet smelling compo ...
... yeast and in the absence of air X is converted to compound Y in the absence of air, X is converted to compound Y and colourless gas. Compound Y reacts with sodium metal to produce a gas Z which gives a ‘pop’ sound with a glowing splint. Y also reacts with ethanoic acid to give a sweet smelling compo ...
enthalpy changes
... The amount of energy released from a chemical reaction is affected to the number of moles of reactant or product. If given the mass of reactant or product, the enthalpy change (∆H in kJ) ...
... The amount of energy released from a chemical reaction is affected to the number of moles of reactant or product. If given the mass of reactant or product, the enthalpy change (∆H in kJ) ...
103. Oxalates as Activating Groups for Alcohols in Visible Light Photoredox Catalysis: Formation of Quaternary Centers by Redox-Neutral Fragment Coupling
... with aqueous CsOH allowed pure cesium oxalate 44 to be isolated in one step and high yield from alcohol 69 without the use of chromatography. Coupling of oxalate 44 (1.5 equiv) with commercially available 4-vinylfuran-2-one (1.0 equiv) proceeded with perfect diastereo- and regioselectivity in 98% yi ...
... with aqueous CsOH allowed pure cesium oxalate 44 to be isolated in one step and high yield from alcohol 69 without the use of chromatography. Coupling of oxalate 44 (1.5 equiv) with commercially available 4-vinylfuran-2-one (1.0 equiv) proceeded with perfect diastereo- and regioselectivity in 98% yi ...
Summer Study Assignment – Honors Chem 2/AP Chemistry
... 31. Write the electron configuration using the Noble Gas core method for californium. 32. Write a balanced equation for the following double replacement reactions: a. Calcium hydroxide (aq) + nitric acid (aq) b. Chromium (III) sulfite (aq) + sulfuric acid (aq) c. Zinc chloride (aq) + ammonium su ...
... 31. Write the electron configuration using the Noble Gas core method for californium. 32. Write a balanced equation for the following double replacement reactions: a. Calcium hydroxide (aq) + nitric acid (aq) b. Chromium (III) sulfite (aq) + sulfuric acid (aq) c. Zinc chloride (aq) + ammonium su ...
Chemistry JAMB Past Questions
... A jet plane carrying 3,000 kg of ethane burns off all the gas forming water and carbondioxide. If all the carbondioxide is expelled and the water formed is condensed and kept on board the plane, then the gain in weight is A. ...
... A jet plane carrying 3,000 kg of ethane burns off all the gas forming water and carbondioxide. If all the carbondioxide is expelled and the water formed is condensed and kept on board the plane, then the gain in weight is A. ...
New Exp8
... Competition can occur with SN1 reaction if reaction conditions are not ‘controlled’ (when protic solvents, non-basic nucleophiles are used). Mixtures of products form with the E1 reaction (also SN1). Unsymmetrical reagents and rearrangements possible (hydride and methyl shifts).This mechanism works ...
... Competition can occur with SN1 reaction if reaction conditions are not ‘controlled’ (when protic solvents, non-basic nucleophiles are used). Mixtures of products form with the E1 reaction (also SN1). Unsymmetrical reagents and rearrangements possible (hydride and methyl shifts).This mechanism works ...
Main Group Organometallic Compounds
... • All organometallic compounds are thermodynamically unstable to oxidation – Due to large negative free energies of formation of metal oxide, carbon dioxide and water ...
... • All organometallic compounds are thermodynamically unstable to oxidation – Due to large negative free energies of formation of metal oxide, carbon dioxide and water ...
Alcohols - SAVE MY EXAMS!
... ALLOW correct bonds shown by the appropriate absorption on the IR spectrum IGNORE reference to C—O bond ALLOW methylpropan-2-ol OR tertiarybutanol ...
... ALLOW correct bonds shown by the appropriate absorption on the IR spectrum IGNORE reference to C—O bond ALLOW methylpropan-2-ol OR tertiarybutanol ...
Strychnine total synthesis

Strychnine total synthesis in chemistry describes the total synthesis of the complex biomolecule strychnine. The first reported method by the group of Robert Burns Woodward in 1954 is considered a classic in this research field. At the time it formed the natural conclusion to an elaborate process of molecular structure elucidation that started with the isolation of strychnine from the beans of Strychnos ignatii by Pierre Joseph Pelletier and Joseph Bienaimé Caventou in 1818. Major contributors to the entire effort were Sir Robert Robinson with over 250 publications and Hermann Leuchs with another 125 papers in a time span of 40 years. Robinson was awarded the Nobel Prize in Chemistry in 1947 for his work on alkaloids, strychnine included. The process of chemical identification was completed with publications in 1946 by Robinson and later confirmed by Woodward in 1947. X-ray structures establishing the absolute configuration became available between 1947 and 1951 with publications from J. M. Bijvoet and J.H. Robertson .Woodward published a very brief account on the strychnine synthesis in 1954 (just 3 pages) and a lengthy one (42 pages) in 1963.Many more methods exist and reported by the research groups of Magnus, Overman, Kuehne, Rawal, Bosch, Vollhardt, Mori, Shibasaki, Li, Fukuyama Vanderwal and MacMillan. Synthetic (+)-strychnine is also known. Racemic synthesises were published by Padwa in 2007 and in 2010 by Andrade and by Reissig.In his 1963 publication Woodward quoted Sir Robert Robinson who said for its molecular size it is the most complex substance known.