CHAPTER 12. DECODING ORGANIC STRUCTURES: THE
... Since life on this planet is based on compounds of the element carbon, the study of these compounds, called organic chemistry, is an important area of chemistry. As we have seen in Chapter 10, the ability of carbon to form single or multiple covalent bonds with itself to form chains of varying lengt ...
... Since life on this planet is based on compounds of the element carbon, the study of these compounds, called organic chemistry, is an important area of chemistry. As we have seen in Chapter 10, the ability of carbon to form single or multiple covalent bonds with itself to form chains of varying lengt ...
Unit 8 Note Packet
... 2. Balance a chemical equation based upon the law of conservation of matter. 3. Analyze or draw a graph for the energy change of a chemical reaction. 4. Calculate the heat of solution for a given compound. We are looking for: 1a. Given the word equation/sentence, translate it into a formula chemical ...
... 2. Balance a chemical equation based upon the law of conservation of matter. 3. Analyze or draw a graph for the energy change of a chemical reaction. 4. Calculate the heat of solution for a given compound. We are looking for: 1a. Given the word equation/sentence, translate it into a formula chemical ...
ENZYME MIMIC ASYMMETRIC ALDOL REACTIONS
... reactions with α-unsubstituted aldehydes result in modest ee’s along with product contamination by selfaldol condensation product (entry 5 and 6).18 This is presumably due to the difficulty in differentiating the α-protons of the aldehyde and ketone. The mechanism of proline-catalyzed intermolecular ...
... reactions with α-unsubstituted aldehydes result in modest ee’s along with product contamination by selfaldol condensation product (entry 5 and 6).18 This is presumably due to the difficulty in differentiating the α-protons of the aldehyde and ketone. The mechanism of proline-catalyzed intermolecular ...
Jordan University of Science and Technology
... Reactions of alkenes, addition of HCl, addition of water, addition of halogens, oxidation,Hydroboration- oxidation, reduction, polymerization, addition reactions in dienes Alkynes, structure, naming, addition of HX, hydration (ketone formation ), addition of X2 , acidity of terminal alkynes, structu ...
... Reactions of alkenes, addition of HCl, addition of water, addition of halogens, oxidation,Hydroboration- oxidation, reduction, polymerization, addition reactions in dienes Alkynes, structure, naming, addition of HX, hydration (ketone formation ), addition of X2 , acidity of terminal alkynes, structu ...
2 - mrstorie
... EQUILIBRIUM PROBLEMS 6. At 773 K, the reaction 2 NO (g) + O2 (g) 2 NO2 (g) produces the following concentrations: [NO] = 3.49 X 10-4M; [O2] = 0.80M; [NO2] = 0.25M. a. What is the equilibrium constant expression for this reaction? Kc = [NO]2/[NO]2[O2] b. What is the equilibrium constant for the re ...
... EQUILIBRIUM PROBLEMS 6. At 773 K, the reaction 2 NO (g) + O2 (g) 2 NO2 (g) produces the following concentrations: [NO] = 3.49 X 10-4M; [O2] = 0.80M; [NO2] = 0.25M. a. What is the equilibrium constant expression for this reaction? Kc = [NO]2/[NO]2[O2] b. What is the equilibrium constant for the re ...
Enzymes
... - With exception of coenz. Q, all members of chain are proteins. These may act as enz’s as is the case with dehydrogenases, they may contain iron as part of an iron-sulfur center, they may be coordinated with a porphyrin ring as in cytochromes, or they may contain copper, as does the cytochrome a + ...
... - With exception of coenz. Q, all members of chain are proteins. These may act as enz’s as is the case with dehydrogenases, they may contain iron as part of an iron-sulfur center, they may be coordinated with a porphyrin ring as in cytochromes, or they may contain copper, as does the cytochrome a + ...
Inorganic and organic chemistry 2
... This Answers document provides suggestions for some of the possible answers that might be given for the questions asked in the workbook. They are not exhaustive and other answers may be acceptable, but they are intended as a guide to give teachers and students feedback. ...
... This Answers document provides suggestions for some of the possible answers that might be given for the questions asked in the workbook. They are not exhaustive and other answers may be acceptable, but they are intended as a guide to give teachers and students feedback. ...
Document
... 7. The separating process of the components of a mixture, based on the differences between the boiling points of the components, is possible by: A. extraction B. distillation C. sublimation D. decantation E. crystallization 8. Which of the following substances: (I) naphthalene, (II) ethanol, (III) ...
... 7. The separating process of the components of a mixture, based on the differences between the boiling points of the components, is possible by: A. extraction B. distillation C. sublimation D. decantation E. crystallization 8. Which of the following substances: (I) naphthalene, (II) ethanol, (III) ...
Click for Section 2.9 notes
... Methane has one C atom (CH4) Ethane has two C atoms (CH3CH3) Propane has three C atoms (CH3CH2CH3) Butane has four C atoms (CH3CH2CH2CH3), etc. ...
... Methane has one C atom (CH4) Ethane has two C atoms (CH3CH3) Propane has three C atoms (CH3CH2CH3) Butane has four C atoms (CH3CH2CH2CH3), etc. ...
Alcohol responsive 2D coordination network of 3
... Coordination network / Guest exchange / Thermal analysis / Alcohol sorption Abstract. The solvothermal reaction of 3-(4-pyridyl)benzoic acid and zinc nitrate yielded a 2D network with an sql topology. Both elemental analysis and single crystal X-ray diffraction studies confirmed the formula of the c ...
... Coordination network / Guest exchange / Thermal analysis / Alcohol sorption Abstract. The solvothermal reaction of 3-(4-pyridyl)benzoic acid and zinc nitrate yielded a 2D network with an sql topology. Both elemental analysis and single crystal X-ray diffraction studies confirmed the formula of the c ...
A Straightforward Route to Enantiopure Pyrrolizidines and
... those of the calcined ones. One or two exothermic peaks were generally observed for the dried catalysts, while there was no endothermic peak corresponding to the stepwise dehydration and/or the decomposition of the AHM (or the decomposition of KNO3). The reason might be that the exothermic peak(s) c ...
... those of the calcined ones. One or two exothermic peaks were generally observed for the dried catalysts, while there was no endothermic peak corresponding to the stepwise dehydration and/or the decomposition of the AHM (or the decomposition of KNO3). The reason might be that the exothermic peak(s) c ...
1b-Redox FIB notes and practice
... Rules for determining oxidation numbers: 1. The oxidation number of a monatomic ion is equal to the charge on the ion. Ie. The oxidation number of Br1- is ____, iron (III) has an oxidation number of __ Ex) Na3N(s) --> 3Na+(aq) + N3-(aq) 2. The oxidation number for metals in an ionic compound is jus ...
... Rules for determining oxidation numbers: 1. The oxidation number of a monatomic ion is equal to the charge on the ion. Ie. The oxidation number of Br1- is ____, iron (III) has an oxidation number of __ Ex) Na3N(s) --> 3Na+(aq) + N3-(aq) 2. The oxidation number for metals in an ionic compound is jus ...
Organic Molecules Lab
... HYDROLYSIS is the opposite reaction to dehydration synthesis. Hydrolysis means literally “breaking with water. In this case, the molecule being hydrolyzed is split AND a molecule of water is split. The –H from the water is added to one of the “broken” ends of the molecule and the –OH from the water ...
... HYDROLYSIS is the opposite reaction to dehydration synthesis. Hydrolysis means literally “breaking with water. In this case, the molecule being hydrolyzed is split AND a molecule of water is split. The –H from the water is added to one of the “broken” ends of the molecule and the –OH from the water ...
Organic Chemistry
... Carbon is unique in its ability to form long chains bonded to other carbon atoms, which results in a wide variety of chemical structures and unique organic compounds. With the exception of CH4, all hydrocarbons consist of a network of bonded C atoms. The structure of a hydrocarbon is often considere ...
... Carbon is unique in its ability to form long chains bonded to other carbon atoms, which results in a wide variety of chemical structures and unique organic compounds. With the exception of CH4, all hydrocarbons consist of a network of bonded C atoms. The structure of a hydrocarbon is often considere ...
Common Incompatible Chemicals
... chromic acid, ethylene glycol, nitric acid, hydroxyl compounds, perchloric acid, peroxides, permanganates ...
... chromic acid, ethylene glycol, nitric acid, hydroxyl compounds, perchloric acid, peroxides, permanganates ...
syllabus details - hrsbstaff.ednet.ns.ca
... The electron pair is attracted by both nuclei leading to a bond which is directional in nature. Both single and multiple bonds should be considered. Dative covalent bonds are not required. ...
... The electron pair is attracted by both nuclei leading to a bond which is directional in nature. Both single and multiple bonds should be considered. Dative covalent bonds are not required. ...
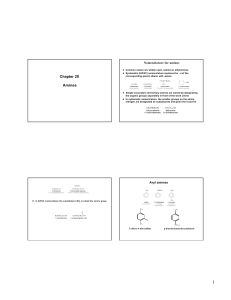
Amines
... t First step in the Gabriel synthesis is alkylation of potassium phthalimide t Reaction of the N-alkylphthalimide with hydrazine in boiling ethanol gives the primary amine ...
... t First step in the Gabriel synthesis is alkylation of potassium phthalimide t Reaction of the N-alkylphthalimide with hydrazine in boiling ethanol gives the primary amine ...
PowerPoint Lectures - Northwest ISD Moodle
... H2CO3, it is unstable and will decompose to give H2O(l) and CO2(g) ...
... H2CO3, it is unstable and will decompose to give H2O(l) and CO2(g) ...
ORGANIC CHEMISTRY
... Chains may contain single, double, or triple bonds. A saturated compound contains only single bond between carbons. These are alkanes. ...
... Chains may contain single, double, or triple bonds. A saturated compound contains only single bond between carbons. These are alkanes. ...
MHS Student Guide to Organic Chemistry
... Chemicals compounds that contain the element Carbon are known as organic compounds. “Organic” comes from the fact that until the mid 1800’s it was thought that these chemicals could only be derived from living plant or animal components. In 1828 Friedrich Woher converted the inorganic ammonium salt ...
... Chemicals compounds that contain the element Carbon are known as organic compounds. “Organic” comes from the fact that until the mid 1800’s it was thought that these chemicals could only be derived from living plant or animal components. In 1828 Friedrich Woher converted the inorganic ammonium salt ...
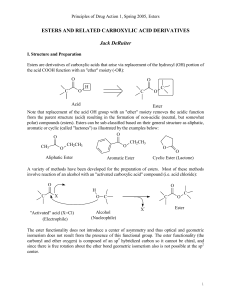
Nucleophilic Substitution on the Carbonyl Group
... acetic acid, and 100 mg of Amberlyst® 15 ion exchange resin. This resin provides a convenient source of acid catalyst. Add a boiling stone or magnetic stir bar. Reflux the reaction mixture for at least 1 hour. Cool to room temperature. Filter the cool mixture to remove the ion exchange resin. Prepar ...
... acetic acid, and 100 mg of Amberlyst® 15 ion exchange resin. This resin provides a convenient source of acid catalyst. Add a boiling stone or magnetic stir bar. Reflux the reaction mixture for at least 1 hour. Cool to room temperature. Filter the cool mixture to remove the ion exchange resin. Prepar ...
Strychnine total synthesis

Strychnine total synthesis in chemistry describes the total synthesis of the complex biomolecule strychnine. The first reported method by the group of Robert Burns Woodward in 1954 is considered a classic in this research field. At the time it formed the natural conclusion to an elaborate process of molecular structure elucidation that started with the isolation of strychnine from the beans of Strychnos ignatii by Pierre Joseph Pelletier and Joseph Bienaimé Caventou in 1818. Major contributors to the entire effort were Sir Robert Robinson with over 250 publications and Hermann Leuchs with another 125 papers in a time span of 40 years. Robinson was awarded the Nobel Prize in Chemistry in 1947 for his work on alkaloids, strychnine included. The process of chemical identification was completed with publications in 1946 by Robinson and later confirmed by Woodward in 1947. X-ray structures establishing the absolute configuration became available between 1947 and 1951 with publications from J. M. Bijvoet and J.H. Robertson .Woodward published a very brief account on the strychnine synthesis in 1954 (just 3 pages) and a lengthy one (42 pages) in 1963.Many more methods exist and reported by the research groups of Magnus, Overman, Kuehne, Rawal, Bosch, Vollhardt, Mori, Shibasaki, Li, Fukuyama Vanderwal and MacMillan. Synthetic (+)-strychnine is also known. Racemic synthesises were published by Padwa in 2007 and in 2010 by Andrade and by Reissig.In his 1963 publication Woodward quoted Sir Robert Robinson who said for its molecular size it is the most complex substance known.