HIGHLIGHT
... simple tectonic units in zeolites already allow for huge diversity in topologies. In the MOF domain, (often polynuclear) transition metal chemistry governs the self-assembly mechanism, and polynuclear transition metal chemistry is an area in which inorganic chemists, despite numerous attempts, have ...
... simple tectonic units in zeolites already allow for huge diversity in topologies. In the MOF domain, (often polynuclear) transition metal chemistry governs the self-assembly mechanism, and polynuclear transition metal chemistry is an area in which inorganic chemists, despite numerous attempts, have ...
reactivity of heterocyclic compounds
... Enzymes are proteins which participate in the chemical reactions associated with life processes by catalyzing them Enzymes can also be used in chemical synthesis (both in academic laboratories or in inductrial processes) Enzymes are fully substrate-selective catalysts and only work on aqueous buffer ...
... Enzymes are proteins which participate in the chemical reactions associated with life processes by catalyzing them Enzymes can also be used in chemical synthesis (both in academic laboratories or in inductrial processes) Enzymes are fully substrate-selective catalysts and only work on aqueous buffer ...
Stereochemistry - University Science Books
... ability of a collection of molecules to rotate plane polarized light (a phenomenon that we explore in detail in Section 6.1.3). In order for a sample to be optically active, it must have an excess of one enantiomer. Now comes the confusion. Optically active was generally used as a synonym for chiral ...
... ability of a collection of molecules to rotate plane polarized light (a phenomenon that we explore in detail in Section 6.1.3). In order for a sample to be optically active, it must have an excess of one enantiomer. Now comes the confusion. Optically active was generally used as a synonym for chiral ...
Co-ordination engineering: when can one speak of an
... Then one derives the structure of 9, ∞2[Ag(µ-NO3)(µ-L)], with solely bridging co-ordination. Otherwise, if an excess of silver was used in the case of the NO3⫺ anion, tetradentate coordination was achieved. The structure of 10, ∞3[Ag3(µ3-NO3)3(µ3-L)2], is the example. An obvious variable in the silv ...
... Then one derives the structure of 9, ∞2[Ag(µ-NO3)(µ-L)], with solely bridging co-ordination. Otherwise, if an excess of silver was used in the case of the NO3⫺ anion, tetradentate coordination was achieved. The structure of 10, ∞3[Ag3(µ3-NO3)3(µ3-L)2], is the example. An obvious variable in the silv ...
A Model For the Calculation of Solvent ... Reaction Rates for Process Design Purposes
... Solvents are used in chemical processes as separation and cleaning agents as well as reaction media. Their selection has a significant impact on the environmental impact, efficiency and profitability of a process. For this reason it would be desirable that solvents be chosen with respect not only to ...
... Solvents are used in chemical processes as separation and cleaning agents as well as reaction media. Their selection has a significant impact on the environmental impact, efficiency and profitability of a process. For this reason it would be desirable that solvents be chosen with respect not only to ...
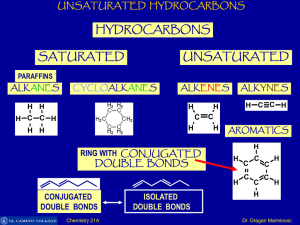
Unsaturated Hydrocarbons
... Benzene boils at 80°C - rather higher than other hydrocarbons of similar molecular size (pentane and hexane, for example). This is presumably due to the ease with which temporary dipoles can be set up involving the delocalized electrons. Methylbenzene (toluene) boils at 111°C. It is a bigger molecul ...
... Benzene boils at 80°C - rather higher than other hydrocarbons of similar molecular size (pentane and hexane, for example). This is presumably due to the ease with which temporary dipoles can be set up involving the delocalized electrons. Methylbenzene (toluene) boils at 111°C. It is a bigger molecul ...
Mathematical Model of Prediction of Nitrogen Pickup in Nitriding
... Copyright © 2014 Saeed Ghali. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Low carbon ferromanganese was nitrided through gas-soli ...
... Copyright © 2014 Saeed Ghali. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Low carbon ferromanganese was nitrided through gas-soli ...
Precious Metal Compounds and Catalysts
... shots, targets, powders, thermocouple wires, and many more. ...
... shots, targets, powders, thermocouple wires, and many more. ...
Mechanism of stibnite volatilization at high temperature
... a steam atmosphere from 923 to 1123 K (650 to 850 °C), and they proposed a complex gas phase reaction mechanism that Sb2S3 could be oxidized/decomposed by water vapor to Sb2O3 and metallic Sb at high temperature. Knowledge of the behaviors of Sb2S3 at high temperatures (i.e. rate of decomposition an ...
... a steam atmosphere from 923 to 1123 K (650 to 850 °C), and they proposed a complex gas phase reaction mechanism that Sb2S3 could be oxidized/decomposed by water vapor to Sb2O3 and metallic Sb at high temperature. Knowledge of the behaviors of Sb2S3 at high temperatures (i.e. rate of decomposition an ...
Chemistry 30 - SharpSchool
... if there is _____________________________________ hydroxyl group, use a prefix (_________________________________) to indicate the ___________________ of OH groups and place the numbers between the parent name and the suffix ***Note, if the suffix starts with a vowel, drop the “e” on the parent nam ...
... if there is _____________________________________ hydroxyl group, use a prefix (_________________________________) to indicate the ___________________ of OH groups and place the numbers between the parent name and the suffix ***Note, if the suffix starts with a vowel, drop the “e” on the parent nam ...
Study on Synthesis and Antibacterial Activity of Co(II) and NiC(II
... Thiosemicarbazone complexes of transition metals have attracted special attention due to their activity against virus, protozoa and certain kinds of tumor and also antitubercular activity(1-3). Recent years witness a growing interest in the chemistry of thiosemicarbazone owing to their ligational pr ...
... Thiosemicarbazone complexes of transition metals have attracted special attention due to their activity against virus, protozoa and certain kinds of tumor and also antitubercular activity(1-3). Recent years witness a growing interest in the chemistry of thiosemicarbazone owing to their ligational pr ...
Photoremovable Protecting Groups
... It was also Barltrop et al.19 who first reported the use of an o-nitrobenzyl group to release benzoic acid (see Eq. (69.2)). The poor yield stemmed from the subsequent conversion of 2-nitrosobenzaldehyde (3), the initial photoproduct, into azobenzene-2,2′-dicarboxylic acid (4),20 which then competed ...
... It was also Barltrop et al.19 who first reported the use of an o-nitrobenzyl group to release benzoic acid (see Eq. (69.2)). The poor yield stemmed from the subsequent conversion of 2-nitrosobenzaldehyde (3), the initial photoproduct, into azobenzene-2,2′-dicarboxylic acid (4),20 which then competed ...
Nano-sized manganese oxides as biomimetic catalysts for water
... use sunlight energy to make different useful materials or high-energy chemicals to store energy (scheme 1) ...
... use sunlight energy to make different useful materials or high-energy chemicals to store energy (scheme 1) ...
IIT-JEE - Brilliant Public School Sitamarhi
... Point defects: When ions or atoms do not hold the theoretical position, this is called point defect. Point defects are of two types: Stoichiometric defects: Schottky defect: Due to missing of ions from lattice point in pairs. Frenkel defect: It is caused due to the creation of lattice vacancy as a r ...
... Point defects: When ions or atoms do not hold the theoretical position, this is called point defect. Point defects are of two types: Stoichiometric defects: Schottky defect: Due to missing of ions from lattice point in pairs. Frenkel defect: It is caused due to the creation of lattice vacancy as a r ...
ALCOHOLS, ETHERS, PHENOLS, AND THIOLS
... All three compounds are alcohols. 1-pentanol has the lowest molar mass and hence the lowest boiling point. 1-octanol has a higher molar mass and therefore a higher boiling point than 1-pentanol. 1,2-pentanediol has two ¬ OH groups and therefore forms more hydrogen bonds than the other two alcohols w ...
... All three compounds are alcohols. 1-pentanol has the lowest molar mass and hence the lowest boiling point. 1-octanol has a higher molar mass and therefore a higher boiling point than 1-pentanol. 1,2-pentanediol has two ¬ OH groups and therefore forms more hydrogen bonds than the other two alcohols w ...
Structural Evaluation and Solution Integrity of Alkali Metal Salt
... multifunctional hydroxamic acid (e.g., salicylhydroxamic acid) or oxime is used to stabilize the metallacrown ring. Incorporation of transition metals into the metallacrowns affords properties not found in the simple organic crowns, such as redox activity, strong absorption spectra, and paramagnetis ...
... multifunctional hydroxamic acid (e.g., salicylhydroxamic acid) or oxime is used to stabilize the metallacrown ring. Incorporation of transition metals into the metallacrowns affords properties not found in the simple organic crowns, such as redox activity, strong absorption spectra, and paramagnetis ...
Hydroformylation

Hydroformylation, also known as oxo synthesis or oxo process, is an important homogeneously catalyzed industrial process for the production of aldehydes from alkenes. This chemical reaction entails the addition of a formyl group (CHO) and a hydrogen atom to a carbon-carbon double bond. This process has undergone continuous growth since its invention in 1938: Production capacity reached 6.6×106 tons in 1995. It is important because the resulting aldehydes are easily converted into many secondary products. For example, the resulting aldehydes are hydrogenated to alcohols that are converted to plasticizers or detergents. Hydroformylation is also used in specialty chemicals, relevant to the organic synthesis of fragrances and natural products. The development of hydroformylation, which originated within the German coal-based industry, is considered one of the premier achievements of 20th-century industrial chemistry.The process typically entails treatment of an alkene with high pressures (between 10 to 100 atmospheres) of carbon monoxide and hydrogen at temperatures between 40 and 200 °C. Transition metal catalysts are required.