
Dimethylsulphoxide Complexes of Vanadium(III) o
... reported here. the bands at 940 and 923 em-I for chloride and 940 and 930 cm- 1 for bromide are attributed to v(S=O) for the coordinated DMSO molecules. As these absorptions are found at lower frequencies than those of a free ligand, this shows that in both cases. the DMSO ligand is bonded to vanadi ...
... reported here. the bands at 940 and 923 em-I for chloride and 940 and 930 cm- 1 for bromide are attributed to v(S=O) for the coordinated DMSO molecules. As these absorptions are found at lower frequencies than those of a free ligand, this shows that in both cases. the DMSO ligand is bonded to vanadi ...
Structural Evaluation and Solution Integrity of Alkali Metal Salt
... multifunctional hydroxamic acid (e.g., salicylhydroxamic acid) or oxime is used to stabilize the metallacrown ring. Incorporation of transition metals into the metallacrowns affords properties not found in the simple organic crowns, such as redox activity, strong absorption spectra, and paramagnetis ...
... multifunctional hydroxamic acid (e.g., salicylhydroxamic acid) or oxime is used to stabilize the metallacrown ring. Incorporation of transition metals into the metallacrowns affords properties not found in the simple organic crowns, such as redox activity, strong absorption spectra, and paramagnetis ...
Module I Oxidation Reactions
... 2.1.3 Mn(IV) Reagents as an Oxidant MnO2 is a useful selective oxidizing reagent in organic synthesis. It is commercially available, and it can also be prepared by the reaction of MnSO4∙4H2O with KMnO4 in aqueous NaOH. G. Cahiez, M. Alami, Encyclopedia of Reagents for Organic Synthesis, John Wiley a ...
... 2.1.3 Mn(IV) Reagents as an Oxidant MnO2 is a useful selective oxidizing reagent in organic synthesis. It is commercially available, and it can also be prepared by the reaction of MnSO4∙4H2O with KMnO4 in aqueous NaOH. G. Cahiez, M. Alami, Encyclopedia of Reagents for Organic Synthesis, John Wiley a ...
Chemical Speciation of Sulfur and Metals in Biogas
... chemical speciation of the metals is regulated by hydrogen sulfide and the formation of metal sulfide precipitates, which in turn restrict the availability of metals for microorganisms. However, despite the limitations set by sulfide, aqueous concentrations of different Co and Ni species were shown ...
... chemical speciation of the metals is regulated by hydrogen sulfide and the formation of metal sulfide precipitates, which in turn restrict the availability of metals for microorganisms. However, despite the limitations set by sulfide, aqueous concentrations of different Co and Ni species were shown ...
Chapter #14 Newest CD
... CH3 CH3 H2 CH3 (d) CH3-CH2-CH-C-CH3 H2 H2 CH3 CH3 (e) CH3-CH2-CH=C-CH-CH3 CH2-CH3 CH3 Plan: For (a) to (c), we refer to Table 15.2. We first name the longest chain (root- + -ane). Then we find the lowest branch numbers by counting C atoms from the end closer to a branch. Finally, we name each branch ...
... CH3 CH3 H2 CH3 (d) CH3-CH2-CH-C-CH3 H2 H2 CH3 CH3 (e) CH3-CH2-CH=C-CH-CH3 CH2-CH3 CH3 Plan: For (a) to (c), we refer to Table 15.2. We first name the longest chain (root- + -ane). Then we find the lowest branch numbers by counting C atoms from the end closer to a branch. Finally, we name each branch ...
Stability Studies of Transition-Metal Linkage Isomers Using
... investigated [Co(DH)2(NCS)2]-, DH is the monoanion of dimethylglyoxime, using IR spectral methods and molecular orbital calculations; they observed small energy differences (less than 1 kcal/mol) between S- and N-bonded linkage isomers. Moreover, the authors stated that π-bonding effects will be mor ...
... investigated [Co(DH)2(NCS)2]-, DH is the monoanion of dimethylglyoxime, using IR spectral methods and molecular orbital calculations; they observed small energy differences (less than 1 kcal/mol) between S- and N-bonded linkage isomers. Moreover, the authors stated that π-bonding effects will be mor ...
Chem 535 Synthetic Organic Chemistry – Common Reducing
... Chem 535 Synthetic Organic Chemistry – Common Reducing Agents for the Conversion of Alcoholic, Carboxylic and Nitrogen-Containing Related Functionality ...
... Chem 535 Synthetic Organic Chemistry – Common Reducing Agents for the Conversion of Alcoholic, Carboxylic and Nitrogen-Containing Related Functionality ...
Answers to Midterm Practice Questions
... b. If A, B, C and D are all -OH groups, what is the R/S designation of the carbon atoms that these OH groups are attached to. ...
... b. If A, B, C and D are all -OH groups, what is the R/S designation of the carbon atoms that these OH groups are attached to. ...
Topic 22 Notes
... hydrogens have been replaced by an alkyl group (part of an alkane) or an aryl group (part of an aromatic ring) 3. Carbon-oxygen double bond a. Aldehydes – have a carbonyl group (“C=O”) with a hydrogen attached to one side of it and either an alkyl (part of a alkane) or aryl group (part of an aromati ...
... hydrogens have been replaced by an alkyl group (part of an alkane) or an aryl group (part of an aromatic ring) 3. Carbon-oxygen double bond a. Aldehydes – have a carbonyl group (“C=O”) with a hydrogen attached to one side of it and either an alkyl (part of a alkane) or aryl group (part of an aromati ...
Lecture - Ch 24
... Synthesis of Amines – Arylamines are prepared from nitration of an aromatic compound and reduction of the nitro group – Reduction by catalytic hydrogenation over platinum is suitable if no other groups can be reduced – Iron, zinc, tin, and tin(II) chloride are effective in ...
... Synthesis of Amines – Arylamines are prepared from nitration of an aromatic compound and reduction of the nitro group – Reduction by catalytic hydrogenation over platinum is suitable if no other groups can be reduced – Iron, zinc, tin, and tin(II) chloride are effective in ...
Relevant notes
... Some Indicators, and Where they Change pH • Phenolphthalein is by far the most famous and common • Most have a limited range over which they turn color. You’re either on the acid side (one color) or base side (different color). But you can’t tell exactly which pH you’re at. • “Universal Indicator” h ...
... Some Indicators, and Where they Change pH • Phenolphthalein is by far the most famous and common • Most have a limited range over which they turn color. You’re either on the acid side (one color) or base side (different color). But you can’t tell exactly which pH you’re at. • “Universal Indicator” h ...
Organic Compounds
... – Carbon: normally forms four covalent bonds and has no unshared pairs of electrons. C – Hydrogen: forms one covalent bond and no unshared pairs of electrons. H – Nitrogen: normally forms three covalent bonds and has one unshared pair of electrons. ...
... – Carbon: normally forms four covalent bonds and has no unshared pairs of electrons. C – Hydrogen: forms one covalent bond and no unshared pairs of electrons. H – Nitrogen: normally forms three covalent bonds and has one unshared pair of electrons. ...
View/Open - POLITesi - Politecnico di Milano
... below melting point) with 395 nm light. The initial crystal (1) melts into isotropic liquid (2) after ca. 30 s of illumination. The recrystallization (3) occurs through a partial nematic phase formation approximately 3 minutes after the UV light illumination. The recrystallization proceeds as a fron ...
... below melting point) with 395 nm light. The initial crystal (1) melts into isotropic liquid (2) after ca. 30 s of illumination. The recrystallization (3) occurs through a partial nematic phase formation approximately 3 minutes after the UV light illumination. The recrystallization proceeds as a fron ...
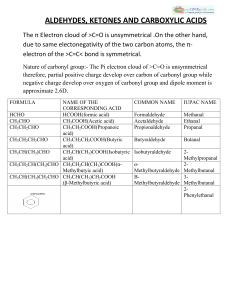
ALDEHYDES, KETONES AND CARBOXYLIC ACIDS
... hydrocarbons and weakly polar compounds such as ethers. :-Lower members are soluble in water because they can form H-bond with water. :-Higher members are insoluble in water due to large size of their hydrophobic group. :-Aldehydes are prepared bya. Dehydrogenation of primary alcohols b. Controlled ...
... hydrocarbons and weakly polar compounds such as ethers. :-Lower members are soluble in water because they can form H-bond with water. :-Higher members are insoluble in water due to large size of their hydrophobic group. :-Aldehydes are prepared bya. Dehydrogenation of primary alcohols b. Controlled ...
Nickel(II) and palladium(II) complexes with [alpha]
... molmetal 䡠 h. With the system B(C6F5)3/AlEt3, the activation process can be formulated as the following two-step reaction: (1) B(C6F5)3 and TEA lead to an aryl/alkyl group exchange and result in the formation of Al(C6F5)nEt3⫺n and B(C6F5)3⫺nEtn; and (2) Al(C6F5)nEt3⫺n will then react with the precat ...
... molmetal 䡠 h. With the system B(C6F5)3/AlEt3, the activation process can be formulated as the following two-step reaction: (1) B(C6F5)3 and TEA lead to an aryl/alkyl group exchange and result in the formation of Al(C6F5)nEt3⫺n and B(C6F5)3⫺nEtn; and (2) Al(C6F5)nEt3⫺n will then react with the precat ...
Naming Organic Compounds I
... The iso prefix can be applied to any branched alkyl group with a (CH3)2CH- group connected to a straight chain. Thus, (CH3)2CH2CH2CH2CH2- is the isohexyl group. ...
... The iso prefix can be applied to any branched alkyl group with a (CH3)2CH- group connected to a straight chain. Thus, (CH3)2CH2CH2CH2CH2- is the isohexyl group. ...
Interaction of saccharides with rare earth metal ions: synthesis and
... spectropolarimeter. Thermal analysis was performed on a Shimadzu DT-30 thermal analyser. Cyclic voltammetry was carried out on a BAS-100 electrochemical analyser. NMR studies were performed on a Bruker Advance DRX500 spectrometer. All the solution characterisations were performed with freshly prepar ...
... spectropolarimeter. Thermal analysis was performed on a Shimadzu DT-30 thermal analyser. Cyclic voltammetry was carried out on a BAS-100 electrochemical analyser. NMR studies were performed on a Bruker Advance DRX500 spectrometer. All the solution characterisations were performed with freshly prepar ...
Chemistry 360 - Athabasca University
... Report Book Structure and How to Prepare for the Labs: This CHEM360 Report Book is to be used in conjunction with the CHEM360 Organic Chemistry II Lab Manual. It consists of an Introduction, Experiment Report Forms, Table of Reagents and Unknown Spectra. The reports are to be completed one month aft ...
... Report Book Structure and How to Prepare for the Labs: This CHEM360 Report Book is to be used in conjunction with the CHEM360 Organic Chemistry II Lab Manual. It consists of an Introduction, Experiment Report Forms, Table of Reagents and Unknown Spectra. The reports are to be completed one month aft ...
Aromatic heterocycles 1: structures and reactions
... ring. This makes it an imine. Most of the imines you have met before (in Chapter 14, for example), have been unstable intermediates in carbonyl group reactions, but in pyridine we have a stable imine—stable because of its aromaticity. All imines are more weakly basic than saturated amines and pyridi ...
... ring. This makes it an imine. Most of the imines you have met before (in Chapter 14, for example), have been unstable intermediates in carbonyl group reactions, but in pyridine we have a stable imine—stable because of its aromaticity. All imines are more weakly basic than saturated amines and pyridi ...
Synthesis, characterization and applications of metal oxides with
... parameters and by the choice of the templates that are used to induce pore formation. The templating methods, which provide attractive routes to hierarchical materials with interconnected pores, tunable pore sizes and, in many cases, flexibility to introduce functional groups, they require post synt ...
... parameters and by the choice of the templates that are used to induce pore formation. The templating methods, which provide attractive routes to hierarchical materials with interconnected pores, tunable pore sizes and, in many cases, flexibility to introduce functional groups, they require post synt ...
Organomet-2009-Hartinger (pdf, 785 KiB) - Infoscience
... only a few Os(II)-arene complexes with anticancer activity were reported. Recently, Dyson et al. have compared the effect of replacing Ru by Os in RAPTA-C, a well-explored drug candidate, and found for Ru and Os only minor differences in terms of in vitro anticancer activity, while a structurally re ...
... only a few Os(II)-arene complexes with anticancer activity were reported. Recently, Dyson et al. have compared the effect of replacing Ru by Os in RAPTA-C, a well-explored drug candidate, and found for Ru and Os only minor differences in terms of in vitro anticancer activity, while a structurally re ...
Hydroformylation

Hydroformylation, also known as oxo synthesis or oxo process, is an important homogeneously catalyzed industrial process for the production of aldehydes from alkenes. This chemical reaction entails the addition of a formyl group (CHO) and a hydrogen atom to a carbon-carbon double bond. This process has undergone continuous growth since its invention in 1938: Production capacity reached 6.6×106 tons in 1995. It is important because the resulting aldehydes are easily converted into many secondary products. For example, the resulting aldehydes are hydrogenated to alcohols that are converted to plasticizers or detergents. Hydroformylation is also used in specialty chemicals, relevant to the organic synthesis of fragrances and natural products. The development of hydroformylation, which originated within the German coal-based industry, is considered one of the premier achievements of 20th-century industrial chemistry.The process typically entails treatment of an alkene with high pressures (between 10 to 100 atmospheres) of carbon monoxide and hydrogen at temperatures between 40 and 200 °C. Transition metal catalysts are required.












![Nickel(II) and palladium(II) complexes with [alpha]](http://s1.studyres.com/store/data/015369127_1-b90d900ca77cb0a51a4faa2871e88861-300x300.png)







